Abstract
Type 2 diabetes (T2D) is one of the major risk factors for developing cardiovascular disease and the resultant devastating morbidity and mortality. The key features of T2D are hyperglycemia, hyperlipidemia, insulin resistance, and impaired insulin secretion. Patients with diabetes and myocardial infarction have worse prognosis than those without T2D. Moreover, obesity and T2D are recognized risk factors in developing severe form of COVID-19 with higher mortality rate. The current lines of drug therapy are insufficient to control T2D and its serious cardiovascular complications. Phosphodiesterase 5 (PDE5) is a cGMP specific enzyme, which is the target of erectile dysfunction drugs including sildenafil, vardenafil, and tadalafil. Cardioprotective effects of PDE5 inhibitors against ischemia/reperfusion (I/R) injury were reported in normal and diabetic animals. Hydroxychloroquine (HCQ) is a widely used antimalarial and anti-inflammatory drug and its hyperglycemia-controlling effect in diabetic patients is also under investigation. This review provides our perspective of a potential use of combination therapy of PDE5 inhibitor with HCQ to reduce cardiovascular risk factors and myocardial I/R injury in T2D. We previously observed that diabetic mice treated with tadalafil and HCQ had significantly reduced fasting blood glucose and lipid levels, increased plasma insulin and insulin-like growth factor-1 levels, and improved insulin sensitivity, along with smaller myocardial infarct size following I/R. The combination treatment activated Akt/mTOR cellular survival pathway, which was likely responsible for the salutary effects. Therefore, pretreatment with PDE5 inhibitor and HCQ may be a potentially useful therapy not only for controlling T2D but also reducing the rate and severity of COVID-19 infection in the vulnerable population of diabetics.
Similar content being viewed by others
Introduction
Type 2 diabetes (T2D) is one of the major risk factors for developing cardiovascular (CV) disease and the resultant devastating morbidity and mortality in the world [1, 2]. The main features of T2D are hyperglycemia, hyperlipidemia, insulin resistance and reduced insulin production in the late stages of the disease. Patients with diabetes and myocardial infarction (MI) have worse prognosis than those without diabetic co-morbidity [3]. Moreover, individuals with diabetes are resistant to most of the cardioprotective modalities that are normally beneficial in the non-diabetic people [4, 5]. In the diabetic heart, reduced glucose uptake and increased circulating free fatty acids lead to shift of energy substrate from carbohydrates to fatty acids, resulting in less ATP production with more oxygen consumption, which makes cells more susceptible to myocardial ischemia [6]. It has been suggested that diabetic hearts retain metabolic flexibility to adapt to hypoxia and become more dependent on oxidative metabolism following hypoxia with 30% lower glycolytic rates and 36% higher fatty acid oxidation than non-diabetic controls, which lead to functional deficit in response to ischemic stress [7]. Thus, normalization of circulating glucose and lipid levels is an important therapeutic strategy for management of diabetic patients [2, 8, 9].
The pathology of myocardial ischemia/reperfusion (I/R) injury involves multiple pathways, which include calcium overload, pH paradox, generation of reactive oxygen species (ROS), inflammation, endothelial dysfunction, and altered myocardial metabolism [10]. Numerous therapeutic strategies targeting single mechanistic pathway have limited effect on human and animal models, indicating that myocardial I/R injury is a complex confluence of divergent biological signaling. Due to the pathogenic complexity, the current lines of drug therapy are insufficient to control diabetes and its serious cardiovascular complications. There is an ongoing search for new and more effective therapies for management of T2D and its CV consequences.
Phosphodiesterase 5 inhibitors and diabetes
Phosphodiesterases (PDEs) belong to a class of phosphohydrolytic enzymes that modulate the intensity of intracellular second messenger signaling by catalyzing the degradation of 3ʹ,5ʹ-cyclic adenosine monophosphate (cAMP) and 3ʹ,5ʹcyclic guanosine monophosphate (cGMP) molecules to their inactive 5ʹ AMP and 5ʹ GMP forms, respectively. PDEs are divided into 11 different families, which include 21 different genes and comprises more than 100 enzyme variants that are generated from multiple promoters and products of alternative splicing [11, 12]. Phosphodiesterase 5 (PDE5) inhibitors are a class of drugs widely used to treat erectile dysfunction (ED). Two of the PDE5 inhibitors sildenafil and tadalafil (TAD) are also approved by FDA for the treatment of pulmonary arterial hypertension [13,14,15]. Tadalafil is a long-acting drug with the average half-life of 17.5 h whereas the duration of actions of sildenafil and vardenafil is generally 4–8 h [16]. Tadalafil has the advantage of being more than 10,000-fold selective for PDE5 over PDE1 to PDE4 and nearly 600-selectivity over PDE6. Moreover, the activity of tadalafil is unaffected by food and has short time (nearly 16–17 min) to onset of action as compared to other PDE5 inhibitors. Several studies from our laboratory have shown that PDE5 inhibitors protect against myocardial I/R injury in both healthy and diabetic animals [17]. The underlying molecular mechanism of cardioprotection with PDE5 inhibitors involves induction of nitric oxide (NO) synthase [18, 19], which leads to subsequent NO-cGMP-protein kinase G (PKG) signaling cascade with promotion of hydrogen sulfide production [20], activation of extracellular signal-regulated kinase 1/2 (Erk1/2) pathway [21], and opening of mitochondrial KATP channels [22].
Metabolic syndrome is a complex systemic disorder, which is comprised of multiple inter-related factors including abdominal obesity, abnormal glucose metabolism, hypertension, insulin resistance, endothelial dysfunction, and inflammatory reaction [23, 24], and may lead to the development of CV diseases [25]. Long-term treatment with sildenafil reduced body weight gain and visceral fat in high fat-fed mice and rats by preventing diet-induced energy balance [26, 27]. In fact, PDE5 is involved in adipocyte physiology and it has been suggested that its inhibition may affect adipogenesis and ameliorate white adipose tissue quality. Chronic treatment with sildenafil had beneficial effects on subcutaneous adipose tissue plasticity in type 2 diabetic mice. Therefore, treatment with PDE5 inhibitors may reduce weight gain by different mechanisms than fat cell lipolysis. Clinical studies suggest a potential protective role of PDE5 inhibitors on endothelial function in short- and long-term assessments [28,29,30]. Diabetic patients with ED are at increased risk for silent coronary artery disease and ED is also is powerful predictor of CV morbidity and mortality [31]. TAD treatment in men with ED showed improved endothelial function, increased insulin levels and a robust decrease in the inflammatory marker, high sensitivity C-reactive protein (hs-CRP) [32]. Furthermore, both acute and chronic administration of sildenafil improved endothelial function in patients with T2D as shown by improved flow-mediated dilatation of the brachial artery [32, 33]. PDE5 inhibitors may have potentially reduced CV events in high risk populations such as men with T2D [34,35,36].
As indicated before, the diabetic myocardium is vulnerable to I/R injury, which has been confirmed in diabetic animal models with hyperinsulinemia [37, 38]. We first demonstrated that chronic treatment of db/db diabetic mice with TAD ameliorated circulating inflammatory cytokines and chemokines while improving fasting glucose levels and reduced infarct size following myocardial I/R injury [39]. Also, chronic treatment with TAD reduced blood glucose and triglyceride levels [39], indicating beneficial effects in maintaining homeostasis of whole body metabolism under conditions of T2D. Likewise, long-term treatment with TAD improved LV diastolic dysfunction (E/A ratio) in C57BL/6NCrl-Leprdb−lb/Crl strain mice, a model which fulfills all the criteria of metabolic syndrome (MetS) [40]. Also, TAD had positive effect on the metabolic health status by improving insulin sensitivity, lowering circulating lipids, and protecting against I/R injury via enhanced NO production in MetS mice. This study was particularly important since the favorable effects of TAD treatment in these mice could clinically benefit MetS patients who are at high risk for CV disease. While the mechanism of these antidiabetic effects is not fully clear, our studies showed that TAD treatment upregulated myocardial SIRT1, PGC-1α expression, and increased phosphorylation of Akt as well as AMPK in the diabetic hearts. In addition, TAD treatment normalized oxidative phosphorylation and attenuated reactive oxygen species production from complex I in the diabetic heart [41]. Using proteomic approaches, we further demonstrated increased expression of antioxidant mitochondrial enzyme glutathione S-transferase Kappa-1 (GSKT-1) and downregulation of redox regulatory chaperones like heat shock protein 8 (HSPA8), and 75 kD glucose regulatory protein (75GRP). Thus, it appears that chronic treatment with TAD in db/db mice modulates proteins involved in cytoskeletal rearrangement and redox signaling of the heart, which may explain the beneficial effects of PDE5 inhibition in diabetes [42, 43]. More recently, a relationship between PDE5 and β2 adrenergic receptor (β2AR) has been suggested which could be a mechanistic axis by which sildenafil improves symptoms of diabetic cardiomyopathy [44]. It was shown that PDE5 inhibition enhanced β2AR stimulation of cGMP and cAMP signals, as well as PKG-dependent excitation–contractile coupling in myocytes derived from high fat diet-fed mice (Fig. 1).
Protective mechanisms of phosphodiesterase 5 (PDE5) inhibitors and hydroxychloroquine (HCQ) against Type II diabetes and pathophysiology of COVID-19 infection. Type II diabetes leads to oxidative stress, inflammation, metabolic derangement, downregulation of NO/cGMP/PKG signaling resulting in endothelial dysfunction and vasoconstriction. These conditions predispose diabetic individuals extremely vulnerable to severe COVID-19 infection and “cytokine storm”, which may lead to high risk of vascular hyperpermeability, pulmonary dysfunction, increased risk of myocardial infarction, and sudden cardiac death. PDE5 inhibitors are known to activate NO/cGMP/PKG signaling pathway, which may improve pulmonary function and exert cardioprotective effect. HCQ can exert anti-diabetes actions via inhibiting insulin degradation as well as enhancing pancreatic insulin secretion, along with its well-known anti-inflammatory effects. Furthermore, PDE5 inhibitors and HCQ may also suppress SARS-CoV-2 cell entry/viral proliferation and alleviate the pathological events (e.g., cytokine storm) of COVID-19 in the vital organs, such as heart and lungs. eNOS endothelial nitric oxide synthase, HCQ hydroxychloroquine, NO nitric oxide, cGMP cyclic guanosine monophosphate, PKG protein kinase G, COVID-19 coronavirus disease 2019, SARS-CoV-2 severe acute respiratory syndrome coronavirus 2, ACE2 angiotensin-converting enzyme 2. The authors would like to acknowledge the use of software of BioRender.com for drawing this figure
In a clinical study, chronic treatment with sildenafil in diabetic men with non-ischemic, nonfailing diabetic cardiomyopathy had an anti-remodeling effect, resulting in improved cardiac kinetics and circulating markers [45]. This effect was reported to be independent of any other vasodilatory or endothelial effects and is apparently exerted through a direct intramyocardial action. More recently, TAD was shown to improve maladaptive cardiac kinetic changes in diabetic men as compared to women [46]. However, TAD treatment reduced hsa-miR-199-5p with increased α-Klotho and improved low-grade inflammation thereby halting renal disease progression in both sexes. Intriguingly, this trial uncovered new molecular pathways targeted by PDE5 inhibition, which may have a role in prevention of cardiac remodeling and renal complication in T2D [46]. Table 1 summarizes a series of completed and ongoing clinical trials using PDE5 inhibitors in management of T2D patients and may be referred for further information.
Hydroxychloroquine in treatment of diabetes
Hydroxychloroquine (HCQ), an analogue of the parent compound chloroquine, is a first-in-line anti-malarial drug widely used in the patients. HCQ has multiple therapeutic properties including anti-inflammatory, immunomodulating, anti-infective, and antithrombotic actions [47]. This drug has been routinely used to treat systemic lupus erythematosus and rheumatoid arthritis [48, 49]. Recent studies have suggested that chloroquine interferes with different metabolic pathways including cholesterol, glucose, amino acids, and mitochondria metabolism [50]. Chloroquine is reported to improve insulin sensitivity through the activation of Akt which increases glucose uptake and glycogen synthesis in L6 muscle cell lines [51]. A few clinical case reports indicated that both chloroquine and HCQ improved glycemic control in patients with type 1 diabetes, partially through the inhibition of insulin degradation [52, 53]. A 3-day oral treatment with chloroquine in 20 patients led to improved serum lipid profile along with increased fasting insulin levels in T2D patients [54]. In alloxan-induced diabetic rats, a high dose combination of HCQ (10 mg/kg) and lipid-lowering drug, atorvastatin (200 mg/kg) demonstrated 21% reduction in blood glucose levels than low dose combinations and individual treatments [55]. HCQ inhibits the degradation of insulin, which enhances the metabolic effects and improves insulin sensitivity [56]. HCQ users among rheumatoid arthritis patients had lower risk of developing diabetes [57] suggesting a potential protective effect of HCQ against insulin resistance.
It is noteworthy that the use of HCQ in diabetic patients appears to be safe, based on a prospective, randomized, placebo, double-blind 6-month trial conducted in 1990 on HCQ therapy (200 mg × 3 times per day) in 38 decompensated, treatment-refractory T2D patients [58]. After 6 months, 11 patients who received insulin and HCQ had a significant decrease in blood glucose (− 11.7 mmol/L) and glycated hemoglobin A1c (HbA1c, − 3.3%). No major side effects were detected. In addition, Gerstein et al. conducted a randomized trial in 2002 in 135 patients with obesity and T2D who were refractory to sulfonylureas [59]. The results showed that HCQ decreased HbA1c and improved glucose tolerance without significant adverse effects during the first 6 months of treatment [59]. Another randomized, double-blind, parallel-arm (placebo vs HCQ 400 mg/day) trial in non-diabetic individuals [60] reported that HCQ treatment significantly improved insulin sensitivity, β-cell function as well as fasting plasma glucose and HbA1c. Again, there were no serious or unexpected adverse effects in HCQ-treated participants. These authors also found that HCQ elevated adiponectin, which may mediate the HCQ-induced improvement in glucose metabolism. Table 2 provides a summary of the completed and ongoing clinical trials using HCQ in management of T2D patients.
Therapeutic effects of tadalafil and hydroxychloroquine against myocardial ischemia/reperfusion injury in type 2 diabetes
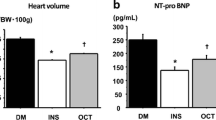
Based on the abovementioned cardioprotective of PDE5 inhibitors and anti-ischemic effects HCQ [61, 62], we contemplated that combination treatment with TAD and HCQ may synergistically protect type 2 diabetic db/db mice from I/R injury. Accordingly we conducted a study in adult male db/db mice with T2D (fasting glucose levels ranging 200–400 mg/dL). Oral dose of HCQ 50 mg/kg/day [63] was administered, which is compatible with the human dose of 250 mg/day for rheumatoid arthritis and systemic lupus erythematosus. The co-treatment with TAD and HCQ for 7 days significantly reduced myocardial infarct size following I/R injury and increased cardiac ATP production [64]. The cardioprotective effects of TAD and HCQ combination also improved lipid and glucose profiles and upregulated the levels of insulin and insulin-like growth factor-1 (IGF-1), which possibly lead to activation of PI3K/Akt/mTOR pathway in β-cells [64]. The mass of pancreas and insulin + β-cell area was increased in TAD- and HCQ-treated groups, suggesting their protective effect on β-cells [64]. Interestingly, previous clinical studies also reported improvement in β-cell function with HCQ or TAD in human subjects [60, 65].
IGF-1 system is interrelated with the insulin signaling [66] to control blood glucose levels [67]. Both insulin and IGF-1 bind to insulin receptors and IGF-1 receptors with different affinity, regulating cell survival through activation of the PI3K-Akt pathway [68, 69]. The co-treatment of TAD + HCQ showed higher plasma levels of IGF-1 as compared to the single drug treatment [64]. Treatment with TAD, HCQ or their combination significantly increased Akt phosphorylation at Thr308, suggesting enhanced cell survival signaling. It has been shown that insulin can protect against I/R injury via facilitating glucose transport [70], inhibition of apoptosis and inflammation [71], and suppression of reactive oxidative species [72]. Insulin and IGF-1 could also improve mitochondrial function through PI3K/Akt pathway [73]. In addition, IGF-1 exerts its indirect cardioprotective effect by increasing insulin sensitivity in various organs/tissues [74].
Potential anti-inflammatory properties of tadalafil and hydroxychloroquine
The anti-inflammatory properties of HCQ have been recognized and utilized for treatment of rheumatoid arthritis in patients around 1950s–1960s [75]. The immunomodulatory effects of HCQ have also been increasingly recognized and utilized as monotherapy or in conjunction with other drugs for the treatment of autoimmune diseases such as systemic lupus erythematosus (SLE), rheumatoid arthritis, Sjögren's syndrome, and antiphospholipid antibody syndrome [76]. HCQ is considered as a critical medication in SLE with a broad spectrum of disease-alleviating benefits including reduction of cutaneous disease and inflammatory arthralgias, thrombosis, and increased longevity, and better glycemic control [77]. In an in vivo mouse model of acute kidney I/R injury and in vitro cultured HK-2 human renal proximal tubule cells exposed to hypoxia/reoxygenation, HCQ pretreatment provided significant renal protection against I/R injury, along with reduction in macrophage and neutrophil infiltration, pro-inflammatory cytokine production, and Nod-like receptor (NLR), the family of innate immune cell sensors, such as the nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3 (NLRP3) inflammasome activation [78].
Many inflammatory mediators including tumor-necrosis factor (TNF), interleukins, and the cytokine-like proteins known as 'adipokines' have been linked to the development of various forms of metabolic disorders and T2D [79, 80]. IL-1β is a prominent proinflammatory cytokine because it generates other inflammatory mediators through signaling via IL-1 receptor, thus initiating a self-amplifying cytokine network [81]. It has been suggested that NLRP3 (also known as cryopyrin), inflammasome promotes autocatalytic activation of the cysteine protease caspase-1 and mediates the cleavage of inactive pro-IL-1β and IL-18, among other proteins, into their active forms. Cryopyrin functions as a danger sensor ultimately causing the autocatalytic cleavage and activation of caspase-1 [82]. Activation of the inflammasome is complex and involves the components ASC (apoptosis-associated speck-like protein containing a caspase recruitment domain), cryopyrin, and pro-caspase-1 [83]. Unlike other caspases of apoptotic pathway, caspase-1 is a key modulator of the inflammatory response to tissue injury [82, 84,85,86,87]. In the heart, genetic deletion of caspase-1 reduced early mortality and ventricular dilatation while its overexpression leads to heart failure [88, 89]. Inflammasome plays a role in the development of insulin resistance [90,91,92,93,94]. Genetic ablation of the cytoplasmic receptor NLRP3 (Nlrp3−/− mice) or NLRP3 inflammasome-associated ASC (PYCARD; Pycard−/− mice) and caspase-1 (Casp1−/− mice) leads to improved glucose tolerance and insulin sensitivity in high fat diet fed mice thereby linking NLRP3 inflammasome to insulin resistance. Moreover, ROS are important activators of cryopyrin, which trigger inflammasome formation [85, 87]. We reported that mitochondrial ROS (H2O2) in the hearts from db/db mice, which was significantly attenuated by treatment with TAD [41] suggesting the possibility that combination treatment with TAD and HCQ may exert even more powerful effect in the inhibition of inflammasome, which could contribute to significant reduction of infarct size as compared to monotherapy (Fig. 1).
PDE5 inhibitors and hydroxychloroquine treatment in diabetic patients—a prevention strategy for COVID-19?
Both obesity and T2D are recognized risk factors in developing severe form of COVID-19, with a higher death rate. In fact, the co-morbid conditions like diabetes, hypertension, and obesity have significant correlation with severity of COVID-19 with T2D alone contributing to 10–40% cases of severity and mortality [95, 96]. Fasting glucose level at the time of hospital admission predicted 28-day mortality even in those without a previous diagnosis of diabetes [97]. Glycemic control and body mass index along with older age, male sex, socio-economic deprivation, non-white ethnicity, and pre-existing renal and CV disease all independently increased mortality [98]. There is also significant endothelial damage in COVID-19, which appears to play key pathophysiological role in the onset and the progression of the disease [99]. The impaired vascular homeostasis secondary to the endothelial dysfunction contributes to the systemic proinflammatory state and multiorgan hypoxia and failure in COVID-19 patients [100].
COVID-19 is suggested to be associated with the imbalance in the generation of NO from the iNOS and eNOS enzymes. The NO generation from eNOS is generally protective of the tissue, whereas excessive formation of NO from iNOS is pro-inflammatory due to oxidative stress [101]. PDE5 inhibitors initiated NO-cGMP-PKG pathway could be potentially beneficial for COVID-19 patients. This is because eNOS activation is in part mediated by AMPK; a serine-threonine protein kinase [101], which may suppress inflammation through inhibition of iNOS. We previously showed that PDE5 inhibitor, TAD triggered AMPK/eNOS/NO signaling and protected against I/R injury in db/db diabetic mice through improvement of mitochondrial dysfunction [41] and suppressing inflammatory cytokines [39]. It has been reported that sildenafil treatment in patients with cerebellar demyelination caused downregulation of both the inactive AMPK and iNOS [102]. Thus, enhancing the AMPK/eNOS/NO/cGMP pathway can potentially counteract inflammation and thromboembolism in patients with T2D [101]. Moreover, because of the higher PDE5 expression in the lungs, treatment with the PDE5 inhibitors may improve the prognosis of pulmonary inflammation caused by SARS-CoV-2 infection.
It has also been suggested that PDE5 inhibitors may potentially block the replication of SARS-CoV-2 [103]. The polyproteins produced by coronaviruses are further processed by certain viral proteases, especially the 3CLpro, into the functional form which then mediates the replication of the virus. The protease inhibitors have been reported to lower mortality of patients with SARS [104]. Based on the crystal structure of the SARS-CoV-2 3CLpro [105], a computational study revealed that among other drugs, clinically approved PDE5 inhibitors such as TAD, sildenafil were potent inhibitors of 3CLpro suggesting that these drugs may block the replication of SARS-CoV-2 at clinically relevant doses [106]. However, a definite clinical evidence is currently unavailable.
The inhibitory effects of HCQ or chloroquine on SARS-CoV2 appear to be multi-factorial and remain incompletely understood [107]. The antiviral effects of HCQ against Zika, Chikungunya, HIV, as well as SARS-CoV2 are likely due to its annulation of endosomal/lysosomal acidification [76]. A recent study with in-silico approaches showed that in the presence of HCQ, the S (Spike) protein of SARS-CoV2 was no longer able to bind gangliosides and, in turn, HCQ would deactivate this new type of ganglioside-binding domain at the N-terminal domain of SARS-CoV-2 S protein and consequently disrupt the viral attachment to lipid rafts as well as its contact with human ACE2 receptors for host cell entry [108]. In addition, it was recently suggested that suppression of coronavirus replication could be attained by interrupting the autophagy pathway [109], since HCQ is a well-known inhibitor of autophagolysosome and it has been shown to potently inhibit SARS‐CoV-2 viral replication in vitro [109,110,111]. Collectively, the newly identified direct anti-SARS-CoV2 action of HCQ provided a potential utility in management of COVID-19 patients at the early stage of viral invasion to vital organs. However, a randomized controlled trial (NCT04321616) from Norway indicated that neither remdesivir nor HCQ affected viral clearance in the hospitalized patients with COVID-19 and also both drugs did not affect the degree of respiratory failure or circulating inflammatory markers of the patients [112]. These negative results suggest that the timing of early treatment/preventive intervention prior to the disease advancement to hospitalization stage may be critical, since early prescription of HCQ, prednisone or both to 717 consecutive SARS-CoV-2-positive Brazilian patients (> 40 years old) significantly reduced hospitalization risk by 50–60% [113]. Thus, based on the compelling evidence of anti-diabetic effects of the class of PDE5 inhibitors and HCQ (Fig. 1), we speculate that combination treatment with these clinically approved drugs may support added benefits including the alleviation of diabetes and reducing the severity of SARS-CoV-2 symptoms [108, 114].
Potential adverse effects of PDE5 inhibitors and hydroxychloroquine in diabetes
Potential dose- and duration-dependent cardio- and neuro-toxicities of HCQ have been recognized in the past decades [115]. It was summarized that HCQ is a generally well-tolerated medication with long-term (years) toxicities may include retinopathy, neuromyotoxicity, cardiac conduction abnormalities, and cardiomyopathy [115, 116]. Deaths from HCQ overdoses most often result from CV collapse [115]. The recent experimental emergency use of HCQ in COVID-19 patients has heightened the concerns on its cardiac toxicity. For example, a retrospective comparative observational study, using the French Pharmacovigilance network database between 1985 and May 2020, found that the estimated incidence of cardiac adverse drug reactions was significantly higher among COVID-19 patients exposed to off-label use of HCQ (2.90%) than those treated with its approved indications such as lupus and rheumatoid arthritis prior to COVID-19 pandemic (0.01%, P < 0.001) [117]. HCQ treatment may cause cardiac QT prolongation and increased dispersion may occur in patients with MI leading to higher susceptibility to ventricular tachyarrhythmias [118]. There is also concern that therapeutic doses of HCQ could trigger cardiac arrhythmic events, which caused sudden cardiac arrest in COVID-19 patients [119,120,121,122]. Furthermore, a population-based study reported a considerably high prevalence of increased QT prolongation and dispersion in T2D diabetic patients [123], indicating a possible excessive mortality risk in these patients.
Conversely, several observational analyses revealed efficacy of HCQ in treating COVID-19 in humans [124,125,126], including significant reduction of in-hospital mortality in the 2541 COVID-19 patients admitted to The Henry Ford Health System in United States [124]. A similar report by the CORIST Collaboration study in Italy revealed that the use of HCQ among 3451 hospitalized patients with COVID-19 was associated with 30% lower 35-day mortality [127]. Such analyses like the CORIST Collaboration can be useful to generate new hypotheses for treating COVID-19 at its earlier stage with a lower dose of HCQ than those of previously used in RECOVERY and SOLIDARITY trials and also need to be further confirmed by large-scale randomized trials [128]. Nevertheless, extra caution should be taken during the course of treatment with HCQ in COVID-19 patients, who need careful monitoring for CV adverse events [129].
Co-administration of PDE5 inhibitor(s) may potentially overcome the occurrence of HCQ-triggered QT prolongation and ventricular arrhythmias, considering that sildenafil treatment reduced ischemia-induced ventricular arrhythmias in dogs [130]. In fact, a more recent study in sheep model of dofetilide-induced QT prolongation demonstrated that treatment with sildenafil suppressed the occurrence of afterdepolarizations and ventricular arrhythmias within 90s of administration [131]. The protective effects of sildenafil were mediated by a PKG-dependent reduction of sarcoplasmic reticulum Ca2+ content. Thus, careful preclinical investigations need to be performed to demonstrate the anti-arrhythmic effects of the combination PDE5 inhibitors in combination with HCQ. Nevertheless, as recently suggested by Dutta et al. [132], HCQ should not be used in patients with diabetic maculopathy and retinopathy. Blood glucose levels need to be closely monitored to reduce the risk of hypoglycemia in the HCQ-treated patients T2D [129].
Conclusion and perspectives
Repositioning, reprofiling and repurposing for old drugs has become a highly beneficial strategy to bring new drugs into clinical reality. This is because the existing drugs have undergone extensive testing, allowing them to move quickly into clinical trials and accelerating their potential. We have provided promising evidence for the efficacy of PDE5 inhibitors and HCQ in protection of diabetic heart. Therefore, combination treatment with TAD and HCQ could potentially lead to a novel line of pharmacotherapy in diabetes to manage CV risk factors and improve clinical outcomes of diabetic patients with acute MI even more effectively. HCQ was considered as one of the repurposed early treatments of COVID-19 infection although with mixed clinical results. Because obesity and T2D are recognized risk factors in developing severe form of COVID-19 with higher mortality rate, we speculate that combination pretreatment therapy with PDE5 inhibitors as well as HCQ may provide additional benefits including the alleviation of diabetic and SARS-CoV2 symptoms (Fig. 1). Nevertheless, future well-designed clinical trials are needed to establish or refute the value of TAD and HCQ combination as potential prophylactics and/or treatments. This notion, in fact, was partially endorsed by the new results from a multicenter, randomized, double-blind, placebo-controlled OXI pilot trial among 125 patients with MI in Finland, which indicated that early administration of HCQ within 48 h after MI (300 mg once daily for 6 months) significantly reduced serum levels of interleukin-6 (IL-6), a key pro-inflammatory marker and did not cause any significant adverse events [133].
Data availability
Data sharing not applicable to this review article as no datasets were generated during the current review work. All data analyzed in this review article are based on the published journal articles assessable via PUBMED (https://pubmed.ncbi.nlm.nih.gov/) and the clinical trials summarized in Tables 1 and 2 were extracted from the information registered by the original trial investigators through https://clinicaltrials.gov/.
References
Ginter E, Simko V (2012) Type 2 diabetes mellitus, pandemic in 21st century. Adv Exp Med Biol 771:42–50
Nichols GA, Joshua-Gotlib S, Parasuraman S (2013) Glycemic control and risk of cardiovascular disease hospitalization and all-cause mortality. J Am Coll Cardiol 62:121–127
Miettinen H, Lehto S, Salomaa V, Mahonen M, Niemela M, Haffner SM, Pyorala K, Tuomilehto J (1998) Impact of diabetes on mortality after the first myocardial infarction. The FINMONICA Myocardial Infarction Register Study Group. Diabetes Care 21:69–75
Paneni F, Luscher TF (2017) Cardiovascular protection in the treatment of type 2 diabetes: a review of clinical trial results across drug classes. Am J Cardiol 120:S17–S27. https://doi.org/10.1016/j.amjcard.2017.05.015
Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Kober LV, Lawson FC, Ping L, Wei X, Lewis EF, Maggioni AP, McMurray JJ, Probstfield JL, Riddle MC, Solomon SD, Tardif JC, Investigators E (2015) Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 373:2247–2257. https://doi.org/10.1056/NEJMoa1509225
Barsotti A, Giannoni A, Di NP, Emdin M (2009) Energy metabolism in the normal and in the diabetic heart. Curr Pharm Des 15:836–840
Mansor LS, Mehta K, Aksentijevic D, Carr CA, Lund T, Cole MA, Le Page L, Sousa Fialho Mda L, Shattock MJ, Aasum E, Clarke K, Tyler DJ, Heather LC (2016) Increased oxidative metabolism following hypoxia in the type 2 diabetic heart, despite normal hypoxia signalling and metabolic adaptation. J Physiol 594:307–320. https://doi.org/10.1113/JP271242
Fukushima A, Milner K, Gupta A, Lopaschuk GD (2015) Myocardial energy substrate metabolism in heart failure: from pathways to therapeutic targets. Curr Pharm Des 21:3654–3664
Gilbert RE, Krum H (2015) Heart failure in diabetes: effects of anti-hyperglycaemic drug therapy. Lancet 385:2107–2117
Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357:1121–1135
Baillie GS, Tejeda GS, Kelly MP (2019) Therapeutic targeting of 3’,5’-cyclic nucleotide phosphodiesterases: inhibition and beyond. Nat Rev Drug Discov 18:770–796. https://doi.org/10.1038/s41573-019-0033-4
Maurice DH, Ke H, Ahmad F, Wang Y, Chung J, Manganiello VC (2014) Advances in targeting cyclic nucleotide phosphodiesterases. Nat Rev Drug Discov 13:290–314. https://doi.org/10.1038/nrd4228
Frey MK, Lang I (2012) Tadalafil for the treatment of pulmonary arterial hypertension. Expert Opin Pharmacother 13:747–755. https://doi.org/10.1517/14656566.2012.662220
Galie N, Brundage BH, Ghofrani HA, Oudiz RJ, Simonneau G, Safdar Z, Shapiro S, White RJ, Chan M, Beardsworth A, Frumkin L, Barst RJ (2009) Tadalafil therapy for pulmonary arterial hypertension. Circulation 119:2894–2903
Hemnes AR, Champion HC (2006) Sildenafil, a PDE5 inhibitor, in the treatment of pulmonary hypertension. Expert Rev Cardiovasc Ther 4:293–300. https://doi.org/10.1586/14779072.4.3.293[doi]
Daugan A, Grondin P, Ruault C, Monnier Le, de Gouville AC, Coste H, Linget JM, Kirilovsky J, Hyafil F, Labaudiniere R (2003) The discovery of tadalafil: a novel and highly selective PDE5 inhibitor. 2: 2,3,6,7,12,12a-hexahydropyrazino[1’,2’:1,6]pyrido[3,4-b]indole-1,4-dione analogues. J Med Chem 46:4533–4542. https://doi.org/10.1021/jm0300577
Das A, Durrant D, Salloum FN, Xi L, Kukreja RC (2015) PDE5 inhibitors as therapeutics for heart disease, diabetes and cancer. Pharmacol Ther 147:12–21
Das A, Xi L, Kukreja RC (2005) Phosphodiesterase-5 inhibitor sildenafil preconditions adult cardiac myocytes against necrosis and apoptosis. Essential role of nitric oxide signaling. J Biol Chem 280:12944–12955
Salloum F, Yin C, Xi L, Kukreja RC (2003) Sildenafil induces delayed preconditioning through inducible nitric oxide synthase-dependent pathway in mouse heart. Circ Res 92:595–597
Salloum FN, Chau VQ, Hoke NN, Abbate A, Varma A, Ockaili RA, Toldo S, Kukreja RC (2009) Phosphodiesterase-5 inhibitor, tadalafil, protects against myocardial ischemia/reperfusion through protein-kinase g-dependent generation of hydrogen sulfide. Circulation 120:S31–S36
Das A, Salloum FN, Xi L, Rao YJ, Kukreja RC (2009) ERK phosphorylation mediates sildenafil-induced myocardial protection against ischemia-reperfusion injury in mice. Am J Physiol Heart Circ Physiol 296:H1236–H1243
Ockaili R, Salloum F, Hawkins J, Kukreja RC (2002) Sildenafil (Viagra) induces powerful cardioprotective effect via opening of mitochondrial K(ATP) channels in rabbits. Am J Physiol Heart Circ Physiol 283:H1263–H1269
Rentoukas E, Tsarouhas K, Kaplanis I, Korou E, Nikolaou M, Marathonitis G, Kokkinou S, Haliassos A, Mamalaki A, Kouretas D, Tsitsimpikou C (2012) Connection between telomerase activity in PBMC and markers of inflammation and endothelial dysfunction in patients with metabolic syndrome. PLoS ONE. https://doi.org/10.1371/journal.pone.0035739
Weiss R, Bremer AA, Lustig RH (2013) What is metabolic syndrome, and why are children getting it? Ann N Y Acad Sci 1281:123–140. https://doi.org/10.1111/nyas.12030
Tune JD, Goodwill AG, Sassoon DJ, Mather KJ (2017) Cardiovascular consequences of metabolic syndrome. Transl Res 183:57–70. https://doi.org/10.1016/j.trsl.2017.01.001
Ayala JE, Bracy DP, Julien BM, Rottman JN, Fueger PT, Wasserman DH (2007) Chronic treatment with sildenafil improves energy balance and insulin action in high fat-fed conscious mice. Diabetes 56:1025–1033. https://doi.org/10.2337/db06-0883
Doghri Y, Chetaneau F, Rhimi M, Kriaa A, Lalanne V, Thorin C, Maguin E, Mallem MY, Desfontis JC (2019) Sildenafil citrate long-term treatment effects on cardiovascular reactivity in a SHR experimental model of metabolic syndrome. PLoS ONE. https://doi.org/10.1371/journal.pone.0223914
Montorsi F, Briganti A, Salonia A, Rigatti P, Burnett AL (2006) Can phosphodiesterase type 5 inhibitors cure erectile dysfunction? Eur Urol 49:979–986. https://doi.org/10.1016/j.eururo.2005.12.055
Schwartz BG, Jackson G, Stecher VJ, Campoli-Richards DM, Kloner RA (2013) Phosphodiesterase type 5 inhibitors improve endothelial function and may benefit cardiovascular conditions. Am J Med 126:192–199. https://doi.org/10.1016/j.amjmed.2012.08.015
Stirban A, Laude D, Elghozi JL, Sander D, Agelink MW, Hilz MJ, Ziegler D (2009) Acute effects of sildenafil on flow mediated dilatation and cardiovascular autonomic nerve function in type 2 diabetic patients. Diabetes Metab Res Rev 25:136–143. https://doi.org/10.1002/dmrr.921
Gazzaruso C, Solerte SB, Pujia A, Coppola A, Vezzoli M, Salvucci F, Valenti C, Giustina A, Garzaniti A (2008) Erectile dysfunction as a predictor of cardiovascular events and death in diabetic patients with angiographically proven asymptomatic coronary artery disease: a potential protective role for statins and 5-phosphodiesterase inhibitors. J Am Coll Cardiol 51:2040–2044. https://doi.org/10.1016/j.jacc.2007.10.069
Aversa A, Greco E, Bruzziches R, Pili M, Rosano G, Spera G (2007) Relationship between chronic tadalafil administration and improvement of endothelial function in men with erectile dysfunction: a pilot study. Int J Impot Res 19:200–207. https://doi.org/10.1038/sj.ijir.3901513
Desouza C, Parulkar A, Lumpkin D, Akers D, Fonseca VA (2002) Acute and prolonged effects of sildenafil on brachial artery flow-mediated dilatation in type 2 diabetes. Diabetes Care 25:1336–1339. https://doi.org/10.2337/diacare.25.8.1336
Cai Z, Zhang J, Li H (2019) Two birds with one stone: regular use of PDE5 inhibitors for treating male patients with erectile dysfunction and cardiovascular diseases. Cardiovasc Drugs Ther 33:119–128. https://doi.org/10.1007/s10557-019-06851-7
Hutchings DC, Anderson SG, Caldwell JL, Trafford AW (2018) Phosphodiesterase-5 inhibitors and the heart: compound cardioprotection? Heart 104:1244–1250. https://doi.org/10.1136/heartjnl-2017-312865
Pofi R, Gianfrilli D, Badagliacca R, Di Dato C, Venneri MA, Giannetta E (2016) Everything you ever wanted to know about phosphodiesterase 5 inhibitors and the heart (but never dared ask): how do they work? J Endocrinol Invest 39:131–142. https://doi.org/10.1007/s40618-015-0339-y
Itoh T, Kouzu H, Miki T, Tanno M, Kuno A, Sato T, Sunaga D, Murase H, Miura T (2012) Cytoprotective regulation of the mitochondrial permeability transition pore is impaired in type 2 diabetic Goto-Kakizaki rat hearts. J Mol Cell Cardiol 53:870–879. https://doi.org/10.1016/j.yjmcc.2012.10.001
Van der Mieren G, Nevelsteen I, Vanderper A, Oosterlinck W, Flameng W, Herijgers P (2012) Angiotensin-converting enzyme inhibition and food restriction in diabetic mice do not correct the increased sensitivity for ischemia-reperfusion injury. Cardiovasc Diabetol 11:89. https://doi.org/10.1186/1475-2840-11-89
Varma A, Das A, Hoke NN, Durrant DE, Salloum FN, Kukreja RC (2012) Anti-inflammatory and cardioprotective effects of tadalafil in diabetic mice. PLoS ONE. https://doi.org/10.1371/journal.pone.0045243
Koka S, Xi L, Kukreja RC (2020) Chronic inhibition of phosphodiesterase 5 with tadalafil affords cardioprotection in a mouse model of metabolic syndrome: role of nitric oxide. Mol Cell Biochem 468:47–58. https://doi.org/10.1007/s11010-020-03710-0
Koka S, Aluri HS, Xi L, Lesnefsky EJ, Kukreja RC (2014) Chronic inhibition of phosphodiesterase 5 with tadalafil attenuates mitochondrial dysfunction in type 2 diabetic hearts: potential role of NO/SIRT1/PGC-1alpha signaling. Am J Physiol Heart Circ Physiol 306:H1558–H1568. https://doi.org/10.1152/ajpheart.00865.2013
Koka S, Das A, Salloum FN, Kukreja RC (2013) Phosphodiesterase-5 inhibitor tadalafil attenuates oxidative stress and protects against myocardial ischemia/reperfusion injury in type 2 diabetic mice. Free Radic Biol Med 60:80–88
Koka S, Xi L, Kukreja RC (2012) Chronic treatment with long acting phosphodiesterase-5 inhibitor tadalafil alters proteomic changes associated with cytoskeletal rearrangement and redox regulation in Type 2 diabetic hearts. Basic Res Cardiol 107:249. https://doi.org/10.1007/s00395-012-0249-5
West TM, Wang Q, Deng B, Zhang Y, Barbagallo F, Reddy GR, Chen D, Phan KS, Xu B, Isidori A, Xiang YK (2019) Phosphodiesterase 5 associates with beta2 adrenergic receptor to modulate cardiac function in type 2 diabetic hearts. J Am Heart Assoc. https://doi.org/10.1161/JAHA.119.012273
Giannetta E, Isidori AM, Galea N, Carbone I, Mandosi E, Vizza CD, Naro F, Morano S, Fedele F, Lenzi A (2012) Chronic Inhibition of cGMP phosphodiesterase 5A improves diabetic cardiomyopathy: a randomized, controlled clinical trial using magnetic resonance imaging with myocardial tagging. Circulation 125:2323–2333. https://doi.org/10.1161/CIRCULATIONAHA.111.063412
Pofi R, Giannetta E, Feola T, Galea N, Barbagallo F, Campolo F, Badagliacca R, Barbano B, Ciolina F, Defeudis G, Filardi T, Sesti F, Minnetti M, Vizza CD, Pasqualetti P, Caboni P, Carbone I, Francone M, Catalano C, Pozzilli P, Lenzi A, Venneri MA, Gianfrilli D, Isidori AM (2022) Sex-specific effects of daily tadalafil on diabetic heart kinetics in RECOGITO, a randomized, double-blind, placebo-controlled trial. Sci Transl Med 14(649):eabl8503. https://doi.org/10.1126/scitranslmed.abl8503
Plantone D, Koudriavtseva T (2018) Current and future use of chloroquine and hydroxychloroquine in infectious, immune, neoplastic, and neurological diseases: a mini-review. Clin Drug Investig 38:653–671. https://doi.org/10.1007/s40261-018-0656-y
Anderson RJ (1995) Hydroxychloroquine therapy in rheumatic diseases. Bull Rheum Dis 44:6–7
Morand EF, McCloud PI, Littlejohn GO (1992) Continuation of long term treatment with hydroxychloroquine in systemic lupus erythematosus and rheumatoid arthritis. Ann Rheum Dis 51:1318–1321
Weyerhauser P, Kantelhardt SR, Kim EL (2018) Re-purposing chloroquine for glioblastoma: potential merits and confounding variables. Front Oncol 8:335. https://doi.org/10.3389/fonc.2018.00335
Halaby MJ, Kastein BK, Yang DQ (2013) Chloroquine stimulates glucose uptake and glycogen synthase in muscle cells through activation of Akt. Biochem Biophys Res Commun 435:708–713
Blazar BR, Whitley CB, Kitabchi AE, Tsai MY, Santiago J, White N, Stentz FB, Brown DM (1984) In vivo chloroquine-induced inhibition of insulin degradation in a diabetic patient with severe insulin resistance. Diabetes 33:1133–1137
Hage MP, Al-Badri MR, Azar ST (2014) A favorable effect of hydroxychloroquine on glucose and lipid metabolism beyond its anti-inflammatory role. Ther Adv Endocrinol Metab 5:77–85
Powrie JK, Shojaee-Moradie F, Watts GF, Smith GD, Sonksen PH, Jones RH (1993) Effects of chloroquine on the dyslipidemia of non-insulin-dependent diabetes mellitus. Metabolism 42:415–419
Pareek A, Yeole PG, Tenpe CR, Chandurkar N, Payghan R (2009) Effect of atorvastatin and hydroxychloroquine combination on blood glucose in alloxan-induced diabetic rats. Indian J Pharmacol 41:125–128. https://doi.org/10.4103/0253-7613.55213
Solomon DH, Garg R, Lu B, Todd DJ, Mercer E, Norton T, Massarotti E (2014) Effect of hydroxychloroquine on insulin sensitivity and lipid parameters in rheumatoid arthritis patients without diabetes mellitus: a randomized, blinded crossover trial. Arthritis Care Res (Hoboken) 66:1246–1251
Bili A, Sartorius JA, Kirchner HL, Morris SJ, Ledwich LJ, Antohe JL, Dancea S, Newman ED, Wasko MC (2011) Hydroxychloroquine use and decreased risk of diabetes in rheumatoid arthritis patients. J Clin Rheumatol 17:115–120. https://doi.org/10.1097/RHU.0b013e318214b6b5
Quatraro A, Consoli G, Magno M, Caretta F, Nardozza A, Ceriello A, Giugliano D (1990) Hydroxychloroquine in decompensated, treatment-refractory noninsulin-dependent diabetes mellitus. A new job for an old drug? Ann Intern Med 112:678–681. https://doi.org/10.7326/0003-4819-112-9-678
Gerstein HC, Thorpe KE, Taylor DW, Haynes RB (2002) The effectiveness of hydroxychloroquine in patients with type 2 diabetes mellitus who are refractory to sulfonylureas: a randomized trial. Diabetes Res Clin Pract 55:209–219
Wasko MC, McClure CK, Kelsey SF, Huber K, Orchard T, Toledo FG (2015) Antidiabetogenic effects of hydroxychloroquine on insulin sensitivity and beta cell function: a randomised trial. Diabetologia 58:2336–2343
Bourke L, McCormick J, Taylor V, Pericleous C, Blanchet B, Costedoat-Chalumeau N, Stuckey D, Lythgoe MF, Stephanou A, Ioannou Y (2015) Hydroxychloroquine protects against cardiac ischaemia/reperfusion injury in vivo via enhancement of ERK1/2 phosphorylation. PLoS ONE. https://doi.org/10.1371/journal.pone.0143771
Yang F, Yin C, Xi L, Kukreja RC (2015) Hydroxychloroquine pretreatment reduces myocardial ischemia-reperfusion injury: role of cardiac extracellular-signal-regulated kinase 5 and autophagy. Circulation 132:A10246–A10246. https://doi.org/10.1161/circ.132.suppl_3.10246
Long L, Yang X, Southwood M, Lu J, Marciniak SJ, Dunmore BJ, Morrell NW (2013) Chloroquine prevents progression of experimental pulmonary hypertension via inhibition of autophagy and lysosomal bone morphogenetic protein type II receptor degradation. Circ Res 112:1159–1170
Wang R, Xi L, Kukreja RC (2017) PDE5 inhibitor tadalafil and hydroxychloroquine cotreatment provides synergistic protection against type 2 diabetes and myocardial infarction in mice. J Pharmacol Exp Ther 361:29–38
Hill KD, Eckhauser AW, Marney A, Brown NJ (2009) Phosphodiesterase 5 inhibition improves beta-cell function in metabolic syndrome. Diabetes Care 32:857–859
Russell SJ, Kahn CR (2007) Endocrine regulation of ageing. Nat Rev Mol Cell Biol 8:681–691. https://doi.org/10.1038/nrm2234
Guler HP, Zapf J, Froesch ER (1987) Short-term metabolic effects of recombinant human insulin-like growth factor I in healthy adults. N Engl J Med 317:137–140. https://doi.org/10.1056/NEJM198707163170303
Buerke M, Murohara T, Skurk C, Nuss C, Tomaselli K, Lefer AM (1995) Cardioprotective effect of insulin-like growth factor I in myocardial ischemia followed by reperfusion. Proc Natl Acad Sci U S A 92:8031–8035. https://doi.org/10.1073/pnas.92.17.8031
Jonassen AK, Sack MN, Mjos OD, Yellon DM (2001) Myocardial protection by insulin at reperfusion requires early administration and is mediated via Akt and p70s6 kinase cell-survival signaling. Circ Res 89:1191–1198. https://doi.org/10.1161/hh2401.101385
Oates A, Nubani R, Smiley J, Kistler L, Hughey S, Theiss P, Perez-Tamayo RA, Eiferman D, Lonchyna V, Higgins RS (2009) Myocardial protection of insulin and potassium in a porcine ischemia-reperfusion model. Surgery 146:23–30
Sack MN, Yellon DM (2003) Insulin therapy as an adjunct to reperfusion after acute coronary ischemia: a proposed direct myocardial cell survival effect independent of metabolic modulation. J Am Coll Cardiol 41:1404–1407
Ji L, Fu F, Zhang L, Liu W, Cai X, Zhang L, Zheng Q, Zhang H, Gao F (2010) Insulin attenuates myocardial ischemia/reperfusion injury via reducing oxidative/nitrative stress. Am J Physiol Endocrinol Metab 298:E871–E880
Ribeiro M, Rosenstock TR, Oliveira AM, Oliveira CR, Rego AC (2014) Insulin and IGF-1 improve mitochondrial function in a PI-3K/Akt-dependent manner and reduce mitochondrial generation of reactive oxygen species in Huntington’s disease knock-in striatal cells. Free Radic Biol Med 74:129–144
Abbas A, Grant PJ, Kearney MT (2008) Role of IGF-1 in glucose regulation and cardiovascular disease. Expert Rev Cardiovasc Ther 6:1135–1149. https://doi.org/10.1586/14779072.6.8.1135
Hamilton EB, Scott JT (1962) Hydroxychloroquine sulfate (“plaguenil”) in treatment of rheumatoid arthritis. Arthritis Rheum 5:502–512. https://doi.org/10.1002/art.1780050507
Martinez GP, Zabaleta ME, Di Giulio C, Charris JE, Mijares MR (2020) The role of chloroquine and hydroxychloroquine in immune regulation and diseases. Curr Pharm Des 26:4467–4485. https://doi.org/10.2174/1381612826666200707132920
Durcan L, Petri M (2016) Immunomodulators in SLE: clinical evidence and immunologic actions. J Autoimmun 74:73–84. https://doi.org/10.1016/j.jaut.2016.06.010
Tang TT, Lv LL, Pan MM, Wen Y, Wang B, Li ZL, Wu M, Wang FM, Crowley SD, Liu BC (2018) Hydroxychloroquine attenuates renal ischemia/reperfusion injury by inhibiting cathepsin mediated NLRP3 inflammasome activation. Cell Death Dis 9:351. https://doi.org/10.1038/s41419-018-0378-3
Feve B, Bastard JP (2009) The role of interleukins in insulin resistance and type 2 diabetes mellitus. Nat Rev Endocrinol 5:305–311. https://doi.org/10.1038/nrendo.2009.62
Hotamisligil GS (2006) Inflammation and metabolic disorders. Nature 444:860–867. https://doi.org/10.1038/nature05485
Arend WP, Palmer G, Gabay C (2008) IL-1, IL-18, and IL-33 families of cytokines. Immunol Rev 223:20–38. https://doi.org/10.1111/j.1600-065X.2008.00624.x
Cassel SL, Sutterwala FS (2010) Sterile inflammatory responses mediated by the NLRP3 inflammasome. Eur J Immunol 40:607–611. https://doi.org/10.1002/eji.200940207
Yin Y, Yan Y, Jiang X, Mai J, Chen NC, Wang H, Yang XF (2009) Inflammasomes are differentially expressed in cardiovascular and other tissues. Int J Immunopathol Pharmacol 22:311–322. https://doi.org/10.1177/039463200902200208
Hornung V, Latz E (2010) Critical functions of priming and lysosomal damage for NLRP3 activation. Eur J Immunol 40:620–623. https://doi.org/10.1002/eji.200940185[doi]
Mariathasan S, Newton K, Monack DM, Vucic D, French DM, Lee WP, Roose-Girma M, Erickson S, Dixit VM (2004) Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430:213–218. https://doi.org/10.1038/nature02664[doi];nature02664[pii]
Miao EA, Leaf IA, Treuting PM, Mao DP, Dors M, Sarkar A, Warren SE, Wewers MD, Aderem A (2010) Caspase-1-induced pyroptosis is an innate immune effector mechanism against intracellular bacteria. Nat Immunol 11:1136–1142
Tschopp J, Schroder K (2010) NLRP3 inflammasome activation: The convergence of multiple signalling pathways on ROS production? Nat Rev Immunol 10:210–215
Frantz S, Ducharme A, Sawyer D, Rohde LE, Kobzik L, Fukazawa R, Tracey D, Allen H, Lee RT, Kelly RA (2003) Targeted deletion of caspase-1 reduces early mortality and left ventricular dilatation following myocardial infarction. J Mol Cell Cardiol 35:685–694
Merkle S, Frantz S, Schon MP, Bauersachs J, Buitrago M, Frost RJ, Schmitteckert EM, Lohse MJ, Engelhardt S (2007) A role for caspase-1 in heart failure. Circ Res 100:645–653
Stienstra R, Joosten LA, Koenen T, van TB, van Diepen JA, van den Berg SA, Rensen PC, Voshol PJ, Fantuzzi G, Hijmans A, Kersten S, Muller M, van den Berg WB, van RN, Wabitsch M, Kullberg BJ, van der Meer JW, Kanneganti T, Tack CJ, Netea MG (2010) The inflammasome-mediated caspase-1 activation controls adipocyte differentiation and insulin sensitivity. Cell Metab 12:593–605
Stienstra R, van Diepen JA, Tack CJ, Zaki MH, van de Veerdonk FL, Perera D, Neale GA, Hooiveld GJ, Hijmans A, Vroegrijk I, van den Berg S, Romijn J, Rensen PC, Joosten LA, Netea MG, Kanneganti TD (2011) Inflammasome is a central player in the induction of obesity and insulin resistance. Proc Natl Acad Sci U S A 108:15324–15329
Vandanmagsar B, Youm YH, Ravussin A, Galgani JE, Stadler K, Mynatt RL, Ravussin E, Stephens JM, Dixit VD (2011) The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat Med 17:179–188
Wen H, Gris D, Lei Y, Jha S, Zhang L, Huang MT, Brickey WJ, Ting JP (2011) Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat Immunol 12:408–415
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J (2010) Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol 11:136–140
Samidurai A, Das A (2020) Cardiovascular complications associated with COVID-19 and potential therapeutic strategies. Int J Mol Sci. https://doi.org/10.3390/ijms21186790
Zaki N, Alashwal H, Ibrahim S (2020) Association of hypertension, diabetes, stroke, cancer, kidney disease, and high-cholesterol with COVID-19 disease severity and fatality: a systematic review. Diabetes Metab Syndr 14:1133–1142. https://doi.org/10.1016/j.dsx.2020.07.005
Wang S, Ma P, Zhang S, Song S, Wang Z, Ma Y, Xu J, Wu F, Duan L, Yin Z, Luo H, Xiong N, Xu M, Zeng T, Jin Y (2020) Fasting blood glucose at admission is an independent predictor for 28-day mortality in patients with COVID-19 without previous diagnosis of diabetes: a multi-centre retrospective study. Diabetologia 63:2102–2111. https://doi.org/10.1007/s00125-020-05209-1
Holman N, Knighton P, Kar P, O’Keefe J, Curley M, Weaver A, Barron E, Bakhai C, Khunti K, Wareham NJ, Sattar N, Young B, Valabhji J (2020) Risk factors for COVID-19-related mortality in people with type 1 and type 2 diabetes in England: a population-based cohort study. Lancet Diabetes Endocrinol 8:823–833. https://doi.org/10.1016/S2213-8587(20)30271-0
Giordo R, Paliogiannis P, Mangoni AA, Pintus G (2021) SARS-CoV-2 and endothelial cell interaction in COVID-19: molecular perspectives. Vasc Biol 3:R15–R23. https://doi.org/10.1530/VB-20-0017
Serebrovska ZO, Chong EY, Serebrovska TV, Tumanovska LV, Xi L (2020) Hypoxia, HIF-1alpha, and COVID-19: from pathogenic factors to potential therapeutic targets. Acta Pharmacol Sin 41:1539–1546. https://doi.org/10.1038/s41401-020-00554-8
Mario L, Roberto M, Marta L, Teresa CM, Laura M (2020) Hypothesis of COVID-19 therapy with sildenafil. Int J Prev Med 11:76. https://doi.org/10.4103/ijpvm.IJPVM_258_20
Nunes AK, Raposo C, Rocha SW, Barbosa KP, Luna RL, da Cruz-Hofling MA, Peixoto CA (2015) Involvement of AMPK, IKbetaalpha-NFkappaB and eNOS in the sildenafil anti-inflammatory mechanism in a demyelination model. Brain Res 1627:119–133. https://doi.org/10.1016/j.brainres.2015.09.008
Shirvaliloo M (2021) Targeting the SARS-CoV-2 3CL(pro) and NO/cGMP/PDE5 pathway in COVID-19: a commentary on PDE5 inhibitors. Future Cardiol 17:765–768. https://doi.org/10.2217/fca-2020-0201
Chu CM, Cheng VC, Hung IF, Wong MM, Chan KH, Chan KS, Kao RY, Poon LL, Wong CL, Guan Y, Peiris JS, Yuen KY, Group HUSS (2004) Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax 59:252–256. https://doi.org/10.1136/thorax.2003.012658
Jin Z, Du X, Xu Y, Deng Y, Liu M, Zhao Y, Zhang B, Li X, Zhang L, Peng C, Duan Y, Yu J, Wang L, Yang K, Liu F, Jiang R, Yang X, You T, Liu X, Yang X, Bai F, Liu H, Liu X, Guddat LW, Xu W, Xiao G, Qin C, Shi Z, Jiang H, Rao Z, Yang H (2020) Structure of M(pro) from SARS-CoV-2 and discovery of its inhibitors. Nature 582:289–293. https://doi.org/10.1038/s41586-020-2223-y
Qiao Z, Zhang H, Ji HF, Chen Q (2020) Computational view toward the inhibition of SARS-CoV-2 spike glycoprotein and the 3CL protease. Computation (Basel). https://doi.org/10.3390/computation8020053
Li H, Yang L, Liu FF, Ma XN, He PL, Tang W, Tong XK, Zuo JP (2020) Overview of therapeutic drug research for COVID-19 in China. Acta Pharmacol Sin 41:1133–1140. https://doi.org/10.1038/s41401-020-0438-y
Fantini J, Di Scala C, Chahinian H, Yahi N (2020) Structural and molecular modelling studies reveal a new mechanism of action of chloroquine and hydroxychloroquine against SARS-CoV-2 infection. Int J Antimicrob Agents. https://doi.org/10.1016/j.ijantimicag.2020.105960
Yuen CK, Wong WM, Mak LF, Wang X, Chu H, Yuen KY, Kok KH (2021) Suppression of SARS-CoV-2 infection in ex-vivo human lung tissues by targeting class III phosphoinositide 3-kinase. J Med Virol 93:2076–2083. https://doi.org/10.1002/jmv.26583
Bonam SR, Muller S, Bayry J, Klionsky DJ (2020) Autophagy as an emerging target for COVID-19: lessons from an old friend, chloroquine. Autophagy 16:2260–2266. https://doi.org/10.1080/15548627.2020.1779467
Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, Li Y, Hu Z, Zhong W, Wang M (2020) Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov 6:16. https://doi.org/10.1038/s41421-020-0156-0
Barratt-Due A, Olsen IC, Nezvalova-Henriksen K, Kasine T, Lund-Johansen F, Hoel H, Holten AR, Tveita A, Mathiessen A, Haugli M, Eiken R, Kildal AB, Berg A, Johannessen A, Heggelund L, Dahl TB, Skara KH, Mielnik P, Le LAK, Thoresen L, Ernst G, Hoff DAL, Skudal H, Kittang BR, Olsen RB, Tholin B, Ystrom CM, Skei NV, Tran T, Dudman S, Andersen JT, Hannula R, Dalgard O, Finbraten AK, Tonby K, Blomberg B, Aballi S, Fladeby C, Steffensen A, Muller F, Dyrhol-Riise AM, Troseid M, Aukrust P, trial NO-S (2021) Evaluation of the effects of remdesivir and hydroxychloroquine on viral clearance in COVID-19: a randomized trial. Ann Intern Med 174:1261–1269. https://doi.org/10.7326/M21-0653
Szente Fonseca SN, de Queiroz SA, Wolkoff AG, Moreira MS, Pinto BC, Valente Takeda CF, Reboucas E, Vasconcellos Abdon AP, Nascimento ALA, Risch HA (2020) Risk of hospitalization for Covid-19 outpatients treated with various drug regimens in Brazil: comparative analysis. Travel Med Infect Dis. https://doi.org/10.1016/j.tmaid.2020.101906
Fantini J, Chahinian H, Yahi N (2020) Synergistic antiviral effect of hydroxychloroquine and azithromycin in combination against SARS-CoV-2: what molecular dynamics studies of virus-host interactions reveal. Int J Antimicrob Agents. https://doi.org/10.1016/j.ijantimicag.2020.106020
Doyno C, Sobieraj DM, Baker WL (2021) Toxicity of chloroquine and hydroxychloroquine following therapeutic use or overdose. Clin Toxicol (Phila) 59:12–23. https://doi.org/10.1080/15563650.2020.1817479
Joyce E, Fabre A, Mahon N (2013) Hydroxychloroquine cardiotoxicity presenting as a rapidly evolving biventricular cardiomyopathy: key diagnostic features and literature review. Eur Heart J Acute Cardiovasc Care 2:77–83. https://doi.org/10.1177/2048872612471215
Romani S, Gerard A, Fresse A, Viard D, Van-Obberghen E, Micallef J, Rocher F, Drici MD, French Pharmacovigilance N (2021) Insights on the evidence of cardiotoxicity of hydroxychloroquine prior and during COVID-19 epidemic. Clin Transl Sci 14:163–169. https://doi.org/10.1111/cts.12883
Mirvis DM (1985) Spatial variation of QT intervals in normal persons and patients with acute myocardial infarction. J Am Coll Cardiol 5:625–631. https://doi.org/10.1016/s0735-1097(85)80387-9
Bun SS, Taghji P, Courjon J, Squara F, Scarlatti D, Theodore G, Baudouy D, Sartre B, Labbaoui M, Dellamonica J, Doyen D, Marquette CH, Levraut J, Esnault V, Bun SS, Ferrari E (2020) QT interval prolongation under hydroxychloroquine/ azithromycin association for inpatients with SARS-CoV-2 lower respiratory tract infection. Clin Pharmacol Ther. https://doi.org/10.1002/cpt.1968
Hooks M, Bart B, Vardeny O, Westanmo A, Adabag S (2020) Effects of hydroxychloroquine treatment on QT interval. Heart Rhythm. https://doi.org/10.1016/j.hrthm.2020.06.029
Sarayani A, Cicali B, Henriksen CH, Brown JD (2020) Safety signals for QT prolongation or Torsades de Pointes associated with azithromycin with or without chloroquine or hydroxychloroquine. Res Social Adm Pharm. https://doi.org/10.1016/j.sapharm.2020.04.016
Uzelac I, Iravanian S, Ashikaga H, Bhatia NK, Herndon C, Kaboudian A, Gumbart JC, Cherry EM, Fenton FH (2020) Fatal arrhythmias: another reason why doctors remain cautious about chloroquine/hydroxychloroquine for treating COVID-19. Heart Rhythm. https://doi.org/10.1016/j.hrthm.2020.05.030
Veglio M, Bruno G, Borra M, Macchia G, Bargero G, D’Errico N, Pagano GF, Cavallo-Perin P (2002) Prevalence of increased QT interval duration and dispersion in type 2 diabetic patients and its relationship with coronary heart disease: a population-based cohort. J Intern Med 251:317–324. https://doi.org/10.1046/j.1365-2796.2002.00955.x
Arshad S, Kilgore P, Chaudhry ZS, Jacobsen G, Wang DD, Huitsing K, Brar I, Alangaden GJ, Ramesh MS, McKinnon JE, O’Neill W, Zervos M, Henry Ford C-TF (2020) Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19. Int J Infect Dis. https://doi.org/10.1016/j.ijid.2020.06.099
Lagier JC, Million M, Gautret P, Colson P, Cortaredona S, Giraud-Gatineau A, Honore S, Gaubert JY, Fournier PE, Tissot-Dupont H, Chabriere E, Stein A, Deharo JC, Fenollar F, Rolain JM, Obadia Y, Jacquier A, La Scola B, Brouqui P, Drancourt M, Parola P, Raoult D, force IC-T, (2020) Outcomes of 3,737 COVID-19 patients treated with hydroxychloroquine/azithromycin and other regimens in Marseille, France: a retrospective analysis. Travel Med Infect Dis. https://doi.org/10.1016/j.tmaid.2020.101791
Yu B, Li C, Chen P, Zhou N, Wang L, Li J, Jiang H, Wang DW (2020) Low dose of hydroxychloroquine reduces fatality of critically ill patients with COVID-19. Sci China Life Sci. https://doi.org/10.1007/s11427-020-1732-2
Covid R, Treatments C, COVID-19 RISK and Treatments (CORIST) Collaboration (2020) Use of hydroxychloroquine in hospitalised COVID-19 patients is associated with reduced mortality: findings from the observational multicentre Italian CORIST study. Eur J Intern Med 82:38–47. https://doi.org/10.1016/j.ejim.2020.08.019
Horby PW, Emberson JR (2020) Hydroxychloroquine for COVID-19: balancing contrasting claims. Eur J Intern Med 82:25–26. https://doi.org/10.1016/j.ejim.2020.11.018
Zhang XL, Li ZM, Ye JT, Lu J, Ye LL, Zhang CX, Liu PQ, Duan DD (2020) Pharmacological and cardiovascular perspectives on the treatment of COVID-19 with chloroquine derivatives. Acta Pharmacol Sin 41:1377–1386. https://doi.org/10.1038/s41401-020-00519-x
Nagy O, Hajnal A, Parratt JR, Vegh A (2004) Sildenafil (Viagra) reduces arrhythmia severity during ischaemia 24 h after oral administration in dogs. Br J Pharmacol 141:549–551. https://doi.org/10.1038/sj.bjp.0705658
Hutchings DC, Pearman CM, Madders GWP, Woods LS, Eisner DA, Dibb KM, Trafford AW (2021) PDE5 inhibition suppresses ventricular arrhythmias by reducing SR Ca(2+) Content. Circ Res 129:650–665. https://doi.org/10.1161/CIRCRESAHA.121.318473
Dutta D, Sharma M, Sharma R (2020) Short-term hydroxychloroquine in COVID-19 infection in people with or without metabolic syndrome - clearing safety issues and good clinical practice. Eur Endocrinol 16:109–112. https://doi.org/10.17925/EE.2020.16.2.109
Ulander L, Tolppanen H, Hartman O, Rissanen TT, Paakkanen R, Kuusisto J, Anttonen O, Nieminen T, Yrjola J, Ryysy R, Drews T, Utriainen S, Karjalainen P, Anttila I, Nurmi K, Silventoinen K, Koskinen M, Kovanen PT, Lehtonen J, Eklund KK, Sinisalo J (2021) Hydroxychloroquine reduces interleukin-6 levels after myocardial infarction: the randomized, double-blind, placebo-controlled OXI pilot trial. Int J Cardiol 337:21–27. https://doi.org/10.1016/j.ijcard.2021.04.062
Mandosi E, Giannetta E, Filardi T, Lococo M, Bertolini C, Fallarino M, Gianfrilli D, Venneri MA, Lenti L, Lenzi A, Morano S (2015) Endothelial dysfunction markers as a therapeutic target for Sildenafil treatment and effects on metabolic control in type 2 diabetes. Expert Opin Ther Targets 19:1617–1622. https://doi.org/10.1517/14728222.2015.1066337
Venneri MA, Barbagallo F, Fiore D, De Gaetano R, Giannetta E, Sbardella E, Pozza C, Campolo F, Naro F, Lenzi A, Isidori AM (2019) PDE5 inhibition stimulates Tie2-expressing monocytes and angiopoietin-1 restoring angiogenic homeostasis in diabetes. J Clin Endocrinol Metab 104:2623–2636. https://doi.org/10.1210/jc.2018-02525
Pofi R, Giannetta E, Galea N, Francone M, Campolo F, Barbagallo F, Gianfrilli D, Venneri MA, Filardi T, Cristini C, Antonini G, Badagliacca R, Frati G, Lenzi A, Carbone I, Isidori AM (2020) Diabetic cardiomiopathy progression is triggered by miR122-5p and involves extracellular matrix: a 5-year prospective study. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2020.10.009
Jansson PA, Murdolo G, Sjogren L, Nystrom B, Sjostrand M, Strindberg L, Lonnroth P (2010) Tadalafil increases muscle capillary recruitment and forearm glucose uptake in women with type 2 diabetes. Diabetologia 53:2205–2208. https://doi.org/10.1007/s00125-010-1819-4
Scheele W, Diamond S, Gale J, Clerin V, Tamimi N, Le V, Walley R, Grover-Paez F, Perros-Huguet C, Rolph T, El Nahas M (2016) Phosphodiesterase type 5 inhibition reduces albuminuria in subjects with overt diabetic nephropathy. J Am Soc Nephrol 27:3459–3468. https://doi.org/10.1681/ASN.2015050473
Santi D, Granata AR, Guidi A, Pignatti E, Trenti T, Roli L, Bozic R, Zaza S, Pacchioni C, Romano S, Nofer JR, Rochira V, Carani C, Simoni M (2016) Six months of daily treatment with vardenafil improves parameters of endothelial inflammation and of hypogonadism in male patients with type 2 diabetes and erectile dysfunction: a randomized, double-blind, prospective trial. Eur J Endocrinol 174:513–522. https://doi.org/10.1530/EJE-15-1100
Powrie JK, Smith GD, Shojaee-Moradie F, Sonksen PH, Jones RH (1991) Mode of action of chloroquine in patients with non-insulin-dependent diabetes mellitus. Am J Physiol 260:E897–E904
Hartman O, Kovanen PT, Lehtonen J, Eklund KK, Sinisalo J (2017) Hydroxychloroquine for the prevention of recurrent cardiovascular events in myocardial infarction patients: rationale and design of the OXI trial. Eur Heart J Cardiovasc Pharmacother 3:92–97. https://doi.org/10.1093/ehjcvp/pvw035
Chakravarti HN, Nag A (2021) Efficacy and safety of hydroxychloroquine as add-on therapy in uncontrolled type 2 diabetes patients who were using two oral antidiabetic drugs. J Endocrinol Invest 44:481–492. https://doi.org/10.1007/s40618-020-01330-5
Funding
This study was partially supported by grants from the National Institutes of Health (R01DK120866; R01CA221813 to RCK; R01HL134366 to RCK & AD; R56HL143809 to SK). LX was a recipient of Pauley Pilot Research Grant of Virginia Commonwealth University. SK was also supported by American Heart Association grant (19AIREA34380223).
Author information
Authors and Affiliations
Contributions
All authors contributed significantly to this work in literature review, data analysis, and manuscript writing and graph preparation. RCK and LX made final assemble of the manuscript, which was approved by all co-authors.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare no known competing financial interests or personal relationships that could have influenced the publication of this review article.
Ethical approval
Not applicable. The present review article does not involve intervention on humans and/or animals directly and it only analyzes and summarizes the information gathered from the published articles.
Consent to participate
Not applicable. The present review article does not involve intervention on humans and/or animals directly and it only analyzes and summarizes the information gathered from the published articles.
Consent for publication
Not applicable. The present review article does not involve intervention on humans and/or animals directly and it only analyzes and summarizes the information gathered from the published articles.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kukreja, R.C., Wang, R., Koka, S. et al. Treating diabetes with combination of phosphodiesterase 5 inhibitors and hydroxychloroquine—a possible prevention strategy for COVID-19?. Mol Cell Biochem 478, 679–696 (2023). https://doi.org/10.1007/s11010-022-04520-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04520-2