Abstract
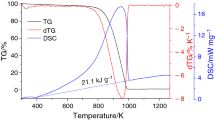
The improved Hummers method uses graphite powder as the raw material to produce graphene oxide, whose preparation releases a large amount of heat. This heat release increases the risks associated with the process as it contributes to combustion and explosion accidents. Based on reaction calorimetry, differential scanning calorimetry, scanning electron microscopy, and energy-dispersive spectrometry, the mechanisms underlying such heat release and related hazards are discussed. According to the conditions influencing the heat release, an orthogonal experimental design was applied to quantify the amount of heat released and the oxidation degree during the preparation process. The optimum working conditions were determined in terms of the stirring speed (250 rpm), feeding time (60 min), feeding temperature (0 °C), and temperature of the intermediate-temperature stage (20 °C). These conditions effectively reduce the risks of the overall manufacturing process.








Similar content being viewed by others
References
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev AS, et al. Correction to improved synthesis of graphene oxide. ACS Nano. 2018;12(2):325–56.
Xiang Q, Yu J, Jaroniec M. Graphene-based semiconductor photocatalysts. Chem Soc Rev. 2012;41(2):782–96.
Cooper DR, D’Anjou B, Ghattamaneni N, Harack B, Hilke M, Horth A, et al. Experimental review of graphene. ISRN Condens Matter Phys. 2012;2012(1):31–8.
Dikin DA, Stankovich S, Zimney EJ, Piner RD, Dommett GH, Evmenenko G, et al. Preparation and characterization of graphene oxide paper. Nature. 2007;448(7152):457–60.
Zhang X, Weeks BL. Improved thermal stability and reduced sublimation rate of pentaerythritol tetranitrate through doping graphene oxide. J Therm Anal Calorim. 2015;122(3):1061–7.
Liu X, Wu W, Qi Y, Qu H, Xu J. Synthesis of a hybrid zinc hydroxystannate/reduction graphene oxide as a flame retardant and smoke suppressant of epoxy resin. J Therm Anal Calorim. 2016;126(2):1–7.
Brodie BC. On the Atomic Weight of Graphite. Philos Trans R Soc Lond. 2009;149(1):249–59.
Hummers WS, Offeman RE. Preparation of graphitic oxide. J Am Chem Soc. 1958;80(6):1339.
Casabianca LB, Shaibat MA, Cai WW, Park S, Piner R, Ruoff RS, et al. NMR-based structural modeling of graphite oxide using multidimensional 13C solid-state NMR and ab initio chemical shift calculations. J Am Chem Soc. 2010;132(16):5672–6.
Figueiredo JL, Pereira MFR, Freitas MMA, Órfão JJM. Modification of the surface chemistry of activated carbons. Carbon. 1999;37(9):1379–89.
Yen YC, Jain A, Altan T. A finite element analysis of orthogonal machining using different tool edge geometries. J Mater Process Technol. 2004;146(1):72–81.
Eigler S, Dotzer C, Hof F, Bauer W, Hirsch A. Sulfur species in graphene oxide. Chemistry. 2013;19(29):9490–6.
Krishnamoorthy K, Mohan R, Kim SJ. Graphene oxide as a photocatalytic material. Appl Phys Lett. 2011;98(24):032107.
Zhang ZB, Wu JJ, Su Y, Zhou J, Gao Y, Yu HY, et al. Layer-by-layer assembly of graphene oxide on polypropylene macroporous membranes via click chemistry to improve antibacterial and antifouling performance. Appl Surf Sci. 2015;332(1):300–7.
Stobinski L, Lesiak B, Malolepszy A, Mazurkiewicz M, Mierzwa B, Zemek J, et al. Graphene oxide and reduced graphene oxide studied by the XRD, TEM and electron spectroscopy methods. J Electron Spectrosc Relat Phenom. 2014;195(15):145–54.
Ju H, Li H, Qin W, Yun Z, Jiao Q. Synthesis of TiO2 nanowire/reduced graphene oxide nanocomposites and their photocatalytic performances. Chem Eng J. 2015;263:144–50.
Chen J, Li Y, Huang L, Li C, Shi G. High-yield preparation of graphene oxide from small graphite flakes via an improved Hummers method with a simple purification process. Carbon. 2015;81(1):826–34.
Wu T, Wang X, Qiu H, Gao J, Wang W, Liu Y. Graphene oxide reduced and modified by soft nanoparticles and its catalysis of the Knoevenagel condensation. J Mater Chem. 2012;22(11):4772–9.
Allahbakhsh A, Haghighi AH, Sheydaei M. Poly(ethylene trisulfide)/graphene oxide nanocomposites. J Therm Anal Calorim. 2017;128(1):427–42.
Zhou Y, Feng P, Li P, Hu H, Shuai C. An optimization scheme of single-spacer nozzle of aluminum roll casting using coupled fluid-thermal finite element analysis. Berlin: Springer; 2011.
Zeng SY, Kang LS, Ding LX. An orthogonal multi-objective evolutionary algorithm for multi-objective optimization problems with constraints. Evol Comput. 2004;12(1):77–98.
Gao S, Zeng S, Xiao B, Zhang L, Shi Y, Tian X et al., editors. An orthogonal multi-objective evolutionary algorithm with lower-dimensional crossover. In: Eleventh conference on congress on evolutionary computation; 2009.
Jiang S, Cai Z, editors. Faster convergence and higher hypervolume for multi-objective evolutionary algorithms by orthogonal and uniform design. In: International symposium on intelligence computation and applications; 2010.
Torréns-Martín D, Fernández-Carrasco L, Blanco-Varela MT. Conduction calorimetric studies of ternary binders based on Portland cement, calcium aluminate cement and calcium sulphate. J Therm Anal Calorim. 2013;114(2):799–807.
Acknowledgements
Thanks to Qingdao Qingke Insafe Science and Technology Co., Ltd. for funding. Funding was provided by The National Key Research and Development Program of China (Grant No. 2017YFC0804801-2).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, Y., Xie, C., Su, L. et al. Study of the risks of the graphene oxide preparation process by reaction calorimetry. J Therm Anal Calorim 139, 101–112 (2020). https://doi.org/10.1007/s10973-019-08364-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-08364-9