Abstract
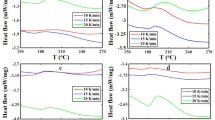
In this study, the effect of nano-silica (nano-SiO2) and boron carbide (B4C) on the curing behavior of the resole resin was evaluated using differential scanning calorimetry (DSC). For the curing behavior study, DSC analysis, carried out at different heating rates, was employed to evaluate the effect of fillers on heat generated during curing and evaporation of volatile components during the curing process. A kinetic model was suggested, and its parameters were determined. The results indicated that the existence of fillers in the resin decreased the evaporation heat of volatile components; meanwhile, the incorporation of the fumed silica increased the curing heat of the resin. Also the plot of reaction rate versus conversion showed that the curing kinetic of filled resin followed the autocatalytic mechanism because of the catalytic effects of water and hydroxyl groups on the surface of nano-silica particles. Model-plotting indicated the excellent agreement between experimental and model data.












Similar content being viewed by others
References
Barthel H, Dreyer M, Gottschalk Gaudig T, Litvinov V, Nikitina E. Fumed silica-rheological additive for adhesives, resins, and paints. Macromol. Symp. 2002;187:752–66.
Ratna D. Handbook of thermoset resins. Shawbury: ISmithers Shawbury; 2009.
Wang J, Jiang H, Jiang N. Study on the pyrolysis of phenol-formaldehyde (PF) resin and modified PF resin. Thermochim Acta. 2009;496:136–42.
Wang J, Jiang N, Jiang H. Effect of the evolution of phenol–formaldehyde resin on the high-temperature bonding. Int J Adhes Adhes. 2009;29:718–23.
Haddadi SA, Mahdavian-ahadi M, Abbasi F. Effect of nanosilica and boron carbide on adhesion strength of high temperature adhesive based on phenolic resin for graphite bonding. Ind Eng Chem Res. 2014;53:11747–54.
Packham DE. Handbook of adhesion. In: Packham DE, editor. Chichester, Wiley; 2005.
Forsdyke KL, Starr TF. Thermoset resins Marcket Report. Smithers Rapra Press; 2002.
Periadurai T, Vijayakumar CT, Balasubramanian M. Thermal decomposition and flame retardant behaviour of SiO2-phenolic nanocomposite. J Anal Appl Pyrolysis. 2010;89:244–9.
Bindu RL, Nair CP, Ninan KN. Phenolic resins with phenyl maleimide functions: thermal characteristics and laminate composite properties. J Appl Polym Sci. 2001;80:1664–74.
Matsumoto A, Hasegawa K, Fukuda A. Studies on modified phenolic resin. Iv: properties of phenolic resin modified with 4-hydroxyphenyl-maleimide/n-butylacrylate copolymers. Polym Int. 1993;30:65–72.
Alonso MV, Oliet M, Pérez JM, Rodríguez F, Echeverría J. Determination of curing kinetic parameters of lignin–phenol–formaldehyde resol resins by several dynamic differential scanning calorimetry methods. Thermochim Acta. 2004;419:161–7.
Zhang C, Binienda WK, Zeng L, Ye X, Chen S. Kinetic study of the novolac resin curing process using model fitting and model-free methods. Thermochim Acta. 2011;523:63–9.
Domínguez JC, Alonso MV, Oliet M, Rojo E, Rodríguez F. Chemorheological analysis of a gelled resol resin curing under non-isothermal conditions by shear strain. Eur Polym J. 2010;46:1237–43.
Domínguez JC, Alonso MV, Oliet M, Rojo E, Rodríguez F. Kinetic study of a phenolic-novolac resin curing process by rheological and DSC analysis. Thermochim Acta. 2010;498:39–44.
Domínguez JC, Alonso MV, Oliet M, Rodríguez F. Chemorheological study of the curing kinetics of a phenolic resol resin gelled. Eur Polym J. 2010;46:50–7.
Esmizadeh E, Naderi G, Yousefi AA, Milone C. Investigation of curing kinetics of epoxy resin/novel nanoclay–carbon nanotube hybrids by non-isothermal differential scanning calorimetry. J Therm Anal Calorim. 2016;. doi:10.1007/s10973-016-5594-4.
Kandelbauer A, Wuzella G, Mahendran A, Taudes I, Widsten P. Model-free kinetic analysis of melamine–formaldehyde resin cure. Chem Eng J. 2009;152:556–65.
Rivero G, Pettarin V, Vázquez A, Manfredi LB. Curing kinetics of a furan resin and its nanocomposites. Thermochim Acta. 2011;516:79–87.
Fan M, Liu J, Li X, Cheng J, Zhang J. Curing behaviors and properties of an extrinsic toughened epoxy/anhydride system and an intrinsic toughened epoxy/anhydride system. Thermochim Acta. 2013;554:39–47.
Jubsilp C, Punson K, Takeichi T, Rimdusit S. Curing kinetics of benzoxazine-epoxy copolymer investigated by non-isothermal differential scanning calorimetry. Polym Degrad Stab. 2010;95:918–24.
Okabe T, Takehara T, Inose K, Hirano N, Nishikawa M, Uehara T. Curing reaction of epoxy resin composed of mixed base resin and curing agent: experiments and molecular simulation. Polymer. 2013;54:4660–8.
Harsch M, Karger-Kocsis J, Holst M. Influence of fillers and additives on the cure kinetics of an epoxy/anhydride resin. Eur Polym J. 2007;43:1168–78.
Lin Y, Song M, Stone CA, Shaw SJ. A comprehensive study on the curing kinetics and network formation of cyanate ester resin/clay nanocomposites. Thermochim Acta. 2013;552:77–86.
Guo J, Lin K, Xu Z. Curing kinetic analysis of phenolic resin filled with nonmetallic materials reclaimed from waste printed circuit boards. Thermochim Acta. 2013;556:13–7.
Ittner Mazali CA, Felisberti MI. Vinyl ester resin modified with silicone-based additives: III. Curing kinetics. Eur Polym J. 2009;45:2222–33.
Jost N. On the curing of a vinylester–urethane hybrid resin. Polymer. 2002;43:1383–9.
Macia-Agullo TG, Fernandez-Garcia JC, Torro-palau A, Orgiles Barcelo AC, Martin-Martinez JM. Hydrophobic or hydrophilic fumed silica as filler of polyurethane adhesives. J Adhes. 1995;50:265–77.
Jauregui-Beloqui B, Fernandez-Garcia JC, Orgiles-Barcelo AC, Mahiques-Bujanda MM, Martin-Martinez JM. Rheological properties of thermoplastic polyurethane adhesive solutions containing fumed silicas of different surface areas. Int J Adhes Adhes. 1999;19:321–8.
Hill RM. Silicone surfactants. CRC Press: Surfactant Science; 1999.
Sourour S, Kamal MR. Differential scanning calorimetry of epoxy cure: isothermal cure kinetics. Thermochim Acta. 1976;14:41–59.
Li C, Liu M-H, Liu Z-Y, Qing M-L, Wang G. DSC and curing kinetics of epoxy resin using cyclohexanediol diglycidyl ether as active diluents. J Therm Anal Calorim. 2014;116:411–6.
Fan M, Li X, Zhang J, Cheng J. Curing kinetics and shape-memory behavior of an intrinsically toughened epoxy resin system. J Therm Anal Calorim. 2015;119:537–46.
Janković B. The kinetic analysis of isothermal curing reaction of an unsaturated polyester resin: estimation of the density distribution function of the apparent activation energy. Chem Eng J. 2010;162:331–40.
Zhang C, Liu X, Cheng J, Zhang J. Study on curing kinetics of diglycidyl 1,2-cyclohexane dicarboxylate epoxy/episulfide resin system with hexahydro-4-methylphthalic anhydride as a curing agent. J Therm Anal Calorim. 2015;120:1893–903.
Vyazovkin S, Wight CA. Model-free and model-fitting approaches to kinetic analysis of isothermal and nonisothermal data. Thermochim Acta. 1999;340–341:53–68.
Pérez JM, Rodríguez F, Alonso MV, Oliet M, Domínguez JC. Curing kinetics of lignin-novolac phenolic resins using non-isothermal methods. J Therm Anal Calorim. 2009;97:979–85.
Wang HM, Zhang YC, Zhu LR, Zhang BL, Zhang YY. Curing behavior and kinetics of epoxy resins cured with liquid crystalline curing agent. J Therm Anal Calorim. 2012;107:1205–11.
Wellen RMR, Canedo EL. On the Kissinger equation and the estimate of activation energies for non-isothermal cold crystallization of PET. Polym Test. 2014;40:33–8.
Budrugeac P, Cucos A. Application of Kissinger, isoconversional and multivariate non-linear regression methods for evaluation of the mechanism and kinetic parameters of phase transitions of type I collagen. Thermochim Acta. 2013;565:241–52.
Morancho JM, Fernandez-Francos X, Acebo C, Ramis X, Salla JM, Serra À. Thermal curing of an epoxy-anhydride system modified with hyperbranched poly(ethylene imine)s with different terminal groups. J. Therm. Anal. Calorim. 2016. doi:10.1007/s10973-016-5376-z.
Bai Y, Yang P, Zhang S, Li Y, Gu Y. Curing kinetics of phenolphthalein-aniline-based benzoxazine investigated by non-isothermal differential scanning calorimetry. J Therm Anal Calorim. 2015;120:1755–64.
Zhang J, Dong H, Tong L, Meng L, Chen Y, Yue G. Investigation of curing kinetics of sodium carboxymethyl cellulose/epoxy resin system by differential scanning calorimetry. Thermochim Acta. 2012;549:63–8.
Natarajan M, Murugavel SC. Cure kinetics of bio-based epoxy resin developed from epoxidized cardanol–formaldehyde and diglycidyl ether of bisphenol—a networks. J Therm Anal Calorim. 2016;125:387–96.
Šesták J, Málek J. Diagnostic limits of phenomenological models of heterogeneous reactions and thermal analysis kinetics. Solid State Ion. 1993;63:245–54.
Zhou D, Du S, Yu L, Liu Z. Nonisothermal curing of a solid resole phenolic resin. J Appl Polym Sci. 2011;121:1938–45.
Rogers ME, Long TE. Synthetic methods in step-growth polymers. NewYork: Wiley; 2003.
Nagieb ZA. Demethylation of thiolignin by reaction with potassium dichromate—a kinetic study. Wood Sci Technol. 1985;19:233–42.
Simon P. Fourty years of the Sestak–Berggren equation. Thermochim Acta. 2011;520:156–7.
Kissounko DA, Deitzel JM, Doherty SP, Shah A, Gillespie JW. Understanding the role of clay silicate nanoparticles with organic modifiers in thermal curing of cyanate ester resin. Eur Polym J. 2008;44:2807–19.
Wan J, Bu ZY, Xu CJ, Li BG, Fan H. Learning about novel amine-adduct curing agents for epoxy resins: butyl-glycidylether-modified poly(propyleneimine) dendrimers. Thermochim Acta. 2011;519:72–82.
Alonso MV, Oliet M, García J, Rodríguez F, Echeverría J. Transformation of dynamic DSC results into isothermal data for the curing kinetics study of the resol resins. J Therm Anal Calorim. 2006;86:797–802.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haddadi, S.A., Kardar, P., Abbasi, F. et al. Effects of nano-silica and boron carbide on the curing kinetics of resole resin. J Therm Anal Calorim 128, 1217–1226 (2017). https://doi.org/10.1007/s10973-016-5951-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5951-3