Abstract
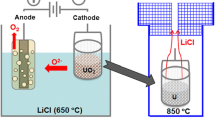
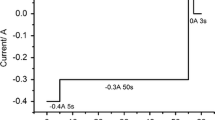
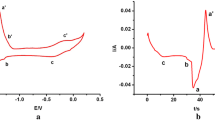
Molten salt mixtures containing LiCl–KCl and NaCl–MgCl2 have been infused with UCl3 via reaction of U metal and FeCl2. The process starts with base salt (LiCl–KCl or NaCl–MgCl2) drying/purification using hydrochlorination via bubbling anhydrous HCl. An auto-titrator running in pH-stat mode was used to determine the point at which there is no net reaction with the salt. U metal is contained in a porous stainless steel basket as it is submerged in the molten salt. The byproduct Fe metal forms dendrites on the basket walls, allowing for simple separation from the molten salt. Based on analysis of salt samples using inductively coupled plasma mass spectroscopy, UCl3 yield of 90% was attained in both NaCl-MgCl2 at 550 °C and in LiCl–KCl at 450 °C. Open circuit potential difference between a W rod working electrode and a Ag/AgCl reference electrode indicated a UCl4 to UCl3 activity ratio of 1 × 10−5. The iron dendrites were comprised of linked cubic structures with length scale of about 50 µm.











Similar content being viewed by others
References
Zhang H, Newton ML, Hamilton ED, Simpson MF (2021) High temperature UCl3 synthesis in molten salt mixtures via reaction of U metal with iron chlorides. J Radioanal Nucl Chem 327(1):1–8
Perhach C, Chamberlain J, Rood N, Hamilton ED, Simpson MF (2022) Chlorination of uranium metal in molten NaCl–CaCl2 via bubbling HCl. J Radioanal Nucl Chem 331:2303–2309
Herrmann SD, Zhao H, Bawane KK, He L, Tolman KR, Pu X (2022) Synthesis and characterization of uranium trichloride in alkali-metal chloride media. J Nucl Mater 565:153728
Kook DH, Cho DK, Lee MS, Lee JY, Choi HJ, Kim YS (2012) Pyroprocess waste disposal system design and dose calculation. Nucl Eng Technol 44:483–490
Holcomb DE, Flanagan GF, Patton BW, Gehin JC, Howard RL, and Harrison TJ (2011) Fast spectrum molten salt reactor options. ORNL/TM-2011/105
Li B, Dai S, Jiang D (2022) Adding MgCl2 to Molten NaCl−UCln (n=3, 4): insights from first-principles molecular dynamics. ChemPhysChem 23:11
Miller WE, Tomczuk Z (2004) Method for making a uranium chloride salt product. US Patent 6,800,262
Woo MS, Kang HS, Lee HS (2009) LiCl–KCl–UCl3 Salt production and transfer for the uranium electrorefining. In: Transactions of the Korean nuclear society spring meeting, Jeju
Bae S-E, Cho Y-H, Park YJ, Ahn HJ, Song K (2010) Oxidation state shift of uranium during U(III) synthesis with Cd(II) and Bi(III) in LiCl–KCl melt. Electrochem Solid-State Lett 13:F25–F27
Westphal BR, Price JC, Mariani RD (2011) Synthesis of uranium trichloride for the pyrometallurgical processing of used nuclear fuel. Idaho National Laboratory, No. INL/CON-10-20111
Lambert H, Kerry T, Sharrad CA (2018) Preparation of uranium (III) in a molten chloride salt: a redox mechanistic study. J Radioanal Nucl Chem 317:925–932
Lee CH, Kim T-J, Yoon D, Jang J, Kim G-Y, Lee S-J (2019) Efficient preparation of UCl3 by ZnCl2 mediated chlorination. J Radioanal Nucl Chem 322:331–336
Inman D, Hills GJ, Young L, Bockris JOM (1959) Electrode reactions in molten salts: the uranium + uranium trichloride system. Trans Faraday Soc 55:1904–1914
Outotec, HSC Chemistry 9.0, Pori (2019)
Ong TC, Sarvghad M, Lippiatt K, Griggs L, Ryan H, Will G, Steinberg TA (2020) Review of the solubility, monitoring, and purification of impurities in molten salts for energy storage in concentrated solar power plants. Renew Sustain Energy Rev 131:110006
Chamberlain J, Simpson MF (2022) Development of a scalable method to chlorinate UO2 to UCl3 using ZrCl4. J Radioanal Nucl Chem
Townsend HE (2001) Effects of alloying elements on the corrosion of steel in industrial atmospheres. Corrosion 57(6):497–501
Polovov IB, Volkovich VA, Charnock JM, Kralj B, Lewin RG, Kinoshita H, May I, Sharrad CA (2008) In situ spectroscopy and Spectroelectrochemistry of uranium in high-temperature alkali chloride molten salts. Inorg Chem 47(17):7474–7482
Vogel SC, Andersson DA, Monreal MJ, Jackson JM, Parker SS, Wang G, Yang P, Zhang J (2021) Crystal structure evolution of UCl3 from room temperature to melting. J Min Met Mater Soc 73(11):3555–3563
Acknowledgements
The authors acknowledge funding from the Los Alamos National Laboratory under subcontract 641280, through LANL Laboratory Directed Research and Development (LDRD) Project #20210113DR.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yankey, J., Chamberlain, J., Monreal, M. et al. UCl3 synthesis in molten LiCl–KCl and NaCl–MgCl2 via galvanically coupled uranium oxidation and FeCl2 reduction. J Radioanal Nucl Chem 332, 2317–2328 (2023). https://doi.org/10.1007/s10967-023-08866-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-08866-9