Abstract
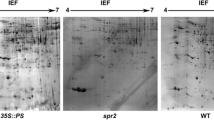
Angiosperms have developed self-incompatibility (SI) systems to reject self-pollen, thereby promoting outcrossing. The Brassicaceae belongs to typical sporophytic system, having a single S-locus controlled SI response, and was chosen as a model system to study SI-related intercellular signal transduction. In this regard, the downstream factor of EXO70A1 was unknown. Here, protein two-dimensional electrophoresis (2-DE) method and coupled with matrix-assisted laser desorption ionization/time of flight of flight mass spectrometry (MALDI-TOF -MS) and peptide mass fingerprinting (PMF) was used to further explore the mechanism of SI responses in Brassica oleracea L. var. capitata L. at protein level. To further confirm the time point of protein profile change, total proteins were collected from B. oleracea pistils at 0 min, 1 h, and 2 h after self-pollination. In total 902, 1088 and 1023 protein spots were separated in 0 min, 1 h and 2 h 2-DE maps, respectively. Our analyses of self-pollination profiles indicated that proteins mainly changed at 1 h post-pollination in B. oleracea. Moreover, 1077 protein spots were separated in cross-pollinated 1 h (CP) pistil 2-DE map. MALDI-TOF-MS and PMF successfully identified 34 differentially-expressed proteins (DEPs) in SP and CP 1 h 2-DE maps. Gene ontology and KEGG analysis revealed an array of proteins grouped in the following categories: stress and defense response (35%), protein metabolism (18%), carbohydrate and energy metabolism (12%), regulation of translation (9%), pollen tube development (12%), transport (9%) and cytoskeletal (6%). Sets of DEPs identified specifically in SP or only up-regulated expressed in CP pistils were chosen for funther investigating in floral organs and during the process of self- and cross-pollination. The function of these DEPs in terms of their potential involvement in SI in B. oleracea is discussed.






Similar content being viewed by others
Abbreviations
- SI:
-
Self-incompatibility
- 2-DE:
-
Two-dimension electrophoresis
- MALDI-TOF-MS:
-
Matrix assisted laser desorption/lionization time of flight mass spectrometry
- PMF:
-
Peptide mass fingerprinting
- SP:
-
Self-pollinated
- CP:
-
Cross-pollinated
- DEPs:
-
Differentially-expressed proteins
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- SCR:
-
S-locus cysteine rich protein
- SRK:
-
S-locus receptor kinase protein
- SLG:
-
S-locus glycoprotein protein
- ARC1:
-
Armadillo repeat containing 1
- EXO70A1:
-
Exocyst 70 A1
- MLPK:
-
M-locus protein kinase protein
- THL1/THL2:
-
Thioredoxin-h-like proteins
- TCA:
-
Trichloroacetic acid
- CHAPS:
-
3-cholamidopropyl dimethylammonio propanesulfonate
- DTT:
-
Dithiothreitol
- IEF:
-
Isoelectric focusing
- SDS-PAGEs:
-
Sodium dodecyl sulfate polyacrylamide gels
- SD:
-
Standard deviation
References
Stephenson AG, Doughty J, Dixon S, Elleman C, Hiscock S, Dickinson HG (1997) The male determinant of self-incompatibility in Brassica oleracea is located in the pollen coating. Plant J 12:1351–1359
Schopfer CR, Nasrallah ME, Nasrallah JB (1999) The male determinant of self-incompatibility in Brassica. Science 286:1697 1700
Kachroo A, Schopfer CR, Nasrallah ME, Nasrallah JB (2001) Allele-specific receptor ligand interactions in Brassica self- incompatibility. Science 293:1824–1826
Shiba H, Takayama S, Iwano M, Shimosato H, Funato M, Nakagawa T, Suzuki G, Watanabe M, Hinata K, Isogai A (2001) A pollen coat protein, SP11/SCR, determines the pollen S-specificity in the self-incompatibility of Brassica species. Plant Physiol 125:2095–2103
Joshua CS, Dixit M, Nasrallah ME, Nasrallah JB (1996) SRK, the stigma-specific S locus receptor kinase of Brassica, is targeted to plasma membrane in transgenic tobacco. Plant Cell 8:429–445
Takasaki T, Hatakeyama K, Suzuki G, Watanabe M, Isogai A, Hinata K (2000) The S receptor kinase determines self- incompatibility in Brassica stigma. Nature 403:913–916
Takayama S, Shimosato H, Shiba H, Shiba H, Funato FS, Chen M, Watanabe M, Iwano M, Isogai A (2001) Direct ligand receptor complex interaction controls Brassica self-incompatibility. Nature 413:534–538
Nasrallah JB, Kao TH, Goldberg ML, Nasrallah ME (1985) A cDNA clone encoding an S-locus-specific glycoprotein from Brassica oleracea. Nature 318:263–267
Stone SL, Anderson EM, Mullen RT, Goring DR (2003) ARC1 is an E3 ubiquitin ligase and promotes the ubiquitination of proteins during the rejection of self-incompatible Brassica pollen. Plant Cell 15:885–898
Samuel MA, Chong YT, Haasen KE, Aldea-Brydges MG, Stone SL, Goring DR (2009) Cellular pathways regulating responses to compatible and self-incompatible pollen in Brassica and Arabidopsis stigmas intersect at Exo70A1, a putative component of the exocyst complex. Plant Cell 21:2655–2671
Mazzurco M, Sulaman W, Elina H, Cock JM, Goring DR (2001) Further analysis of the interactions between the Brassica S receptor kinaseand three interacting proteins (ARC1, THL1 and THL2) in the yeast. Plant Mol Biol 45:365–376
Kakita M, Shimosato H, Murase K, Isogai A, Takayama S (2007) Direct interaction between S- locus receptor kinase and M locus protein kinase involved in Brassica self-incompatibility signaling. Plant Biotechnol 24:185–190
Tantikanjana T, Nasrallah ME, Nasrallah JB (2010) Complex networks of self-incompatibility signaling in the Brassicaceae. Plant Biol 13:520–526
Rumen I, Isabelle FL, Thierry G (2010) When no means no: guide to Brassicaceae self-incompatibility. Trends Plant Sci 15:387–394
Niu Y, Wang ZM, Gao QG, Song M, Wang XJ, Zhu LQ (2009) Expression of ARCl in vitro and test of interaction between ARC1 and SRK from Brassica oleracea L. in signal transduction pathway of self-incompatibility. Acta Agronomica Sinica 35(7):1202–1208
Yang K, Zhang HC, Converse R, Zhu LQ, Yang YJ, Xue LY, Luo B, Chang DL, Gao QG, Wang XJ (2012) Interaction between two self-incompatible signal elements, EXO70A1 and ARC1. Acta Agronomica Sinica 37 (12):2136–2144
Safavian D, Goring DR (2013) Secretory activity is rapidly induced in stigmatic papillae by compatible pollen, but inhibited for self-incompatible pollen in the Brassicaceae. PLoS ONE 8(12):1–13
Goring DR, Indriolo E, Samuel MA (2014) The ARC1 E3 ligase promotes a strong and stable self-incompatibility response in Arabidopsis species: response to the Nasrallah and Nasrallah commentary. Plant Cell 26:3842–3846
Samuel MA, Tang WQ, Jamshed M, Northey J, Patel D, Smith D, Siu KWM, Muench DG, Wang ZY, Goring DR (2011) Proteomic analysis of Brassica stigmatic proteins following the self-incompatibility reaction reveals a role for microtubule dynamics during pollen responses. Mol Cell Proteom 10:1–13
Dickinson H (1995) Dry stigma, water and self-incompatibility in Brassica. Sex Plant Repord 8:1–10
Wang L, Wang C, Ge TT, Wang JJ, Liu TK, Hou XL, Li Y (2014) Expression analysis of self-incompatibility associated genes in non-heading Chinese cabbage. Genetics Mol Res 13(3):5025–5035
Wang L, Peng H, Ge T, Liu T, Hou X, Li Y (2014) Identification of differentially accumulating pistil proteins associated with self-incompatibility of non-heading Chinese cabbage. Plant Biol 16:49–57
Chen S, Zeng J, Gao QG, Zhu LQ, Liu YD, Ren XS, Wang XJ (2013) Differential proteomic analysis of Brassica oleracea stigma and pollen proteins during the early stage of compatible pollination. Acta Horticulturae Sinica 40 (1):69–78
Haffani YZ, Gaude T, Cock JM, Goring DR (2004) Antisense suppression of thioredoxin h mRNA in Brassica napus cv.Westar pistils causes a low level constitutive pollen rejection response. Plant Mol Biol 55:619–630
Sheoran IS, Sproule KA, Olson DJH, Ross ARS, Sawhney V (2006) Proteome profile and functional classification of proteins in Arabidopsis thaliana (Landsberg erecta) mature pollen. Sex Plant Repord 19:185–196
Zou JJ, Song LF, Zhang WZ, Wang Y, Ruan SL, Wu WH (2009) Comparative proteomic analysis of Arabidopsis mature pollen and germinated pollen. J Integr Plant Biol 51(5):438–455
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-∆∆Ct method. Methods 25:402–408
Isogai M, Yoshida T, Shimura T, Yoshikawa N (2015) Pollen tubes introduce Raspberry bushy dwarf virus into embryo sacs during fertilization processes. Virology 484:341–345
Li MN, Xu WY, Yang WQ, Kong ZS, Xue YB (2007) Genome-wide gene expression profiling reveals conserved and novel molecular functions of the stigma in rice. Plant Physiol 144:1797–1812
Holmes-Davis R, Tanaka CK, Vensel WH, Hurkman WJ, McCormick S (2005) Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 5:4864–4884
Englbrecht CC, Schoof H, Bohm S (2004) Conservation, diversification and expansion of C2H2 zinc finger proteins in the Arabidopsis thaliana genome. BMC Genom 5(39):1–17
Jung BG, Lee KO, Lee SS, Chi YH, Jang HH, Kang SS, Lee K, Lim D, Yoon SC, Yun DJ, Inoue Y, Cho MJ (2002) A Chinese cabbage cDNA with high sequence identity to phospholipid hydroperoxide glutathione peroxidases encodes a novel isoform of thioredoxin-dependent peroxidase. J Biol Chem 277(15):12572–12578
Li Y, Hassan YI, Moriyama H, Zempleni J (2012) Holocarboxylase synthetase interacts physically with euchromatic histone lysine N-methyltransferase, linking histone biotinylation with methylation events. J Nutr Biochem 24:1446:1452
Chrispeels MJ, Raikhel NV (1991) Lectins, lectin genes, and their role in plant defense. Plant Cell 3:1–9
Sun H, Kim MK, Pulla RK (2010) Isolation and expression analysis of a novel major latex-like protein (MLP151) gene from Panax ginseng. Mol Biol Rep 37:2215–2222
Sun L, Ren H, Liu R, Li B, Wu T, Sun F, Liu H, Wang X, Dong H (2010) An h-Type thioredoxin functions in tobacco defense responses to two species of viruses and an abiotic oxidative stress. Mol Plant Microbe Interactions 23(11):1470–1485
Serrels B, Sandilands E, Frame MC (2011) Signaling of the direction-sensing FAK/RACK1/PDE4D5 complex to the small GTPase Rap1. Small Gtpases 2(1):54–61
Deswal R, Sopory SK (1998) Biochenical and immunochemical characterization of Brassica juncea glyoxalase I. Phytochemistry 49:2245–2253
Verdoucq L, Moriniere J, Bevan DR, Esen A, Vasella A, Henrissat B, Czjzek M (2004) Structural determinants of substrate specificity in Family 1-glucosidases. J Biol Chem 279(30):31796–31803
Jammes F, Song C, Shin D, Munemasa S, Takeda K, Gu D, Cho D, Lee S, Giordo R, Sritubtim S, Leonhardt N, Ellis BE, Mureta Y, Kwak JM (2009) MPK9 and MPK12 are preferentially expressed in guard cells and positively regulated ROS-mediated ABA signaling. PNAS 106:20520–20525
Shimizu S, Narita M, Tsujimoto Y (1999) Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 399:483–487
Egebjerg J, Douthwaite SR, Liljas A, Garrett RA (1990) Characterization of the binding sites of protein L11 and the L10. (L12)4 pentameric complex in the GTPase domain of 23 S ribosomal RNA from Escherichia coli. J Mol Biol 213(2):275–288
Leitner-Dagan Y, Ovadis M, Shklarman E, Elad Y, David DR, Vainstein A (2006) Expression and functional analyses of the plastid lipid-associated protein CHRC suggest its role in chromoplastogenesis and stress. Plant Physiol 142:233–244
Schubert M, Petersson UA, Haas BJ, Funk C, Schröder WP, Kieselbach T (2002) Proteome Map of the Chloroplast Lumen of Arabidopsis thaliana. J Biol Chem 277(10):8354–8365
Peltier JB, Emanuelsson O, Kalume DE, Ytterberg J, Friso G, Rudella A, Liberles DA, Soderberg L, Roepstorff P, Heijne GV, Wijk KJ (2002) Central functions of the lumenal and peripheral thylakoid proteome of Arabidopsis determined by experimentation and genome-wide prediction. Plant Cell 14:211–236
Reichheld JP, Meyer E, Khafif M, Bonnard G, Meyer Y (2005) AtNTRB is the major mitochondrial thioredoxin reductase in Arabidopsis thaliana. Febs Letters 579:337–342
Vieratra RD (2003) The uniquitin/26 S proteasome pathway, the complex last chapter in the life of many plant proteins. Trends Plant Sci 8(3):1360–1385
Olsiewski PJ, Kaczorowski GJ, Walsh C (1979) Purification and properties of D-Amino Acid dehydrogenase, an inducible membrane-bound iron-sulfur flavoenzymeform from Escherichia coli. B. J Biol Chem 255(10):4487–4494.
Studart-Guimaraes C, Gibon Y, Frankel N, Wood CC, Zanor MI, Fernie AR, Carrari F (2005) Identification and characterization of the α and β subunits of succinyl CoA ligase of tomato. Plant Mol Biol 59:781–791
Landschutze V, Willmitzer L, Muller-Rober B (1995) Inhibition of flower formation by antisense repression of mitochondrial citrate synthase in transgenic potato plants leads to a specific disintegration of the ovary tissues of flowers. EMBO J 14(4):660–666
Taylor LP, Hepler PK (1997) Pollen germination and tube growth. Annu Rev Plant Physiol Plant Mol Biol 48:461–491
Feng HZ, Chen QG, Feng J, Zhang J, Yang XH, Zuo JR (2007) Functional characterization of the Arabidopsis eukaryotic translation initiation factor 5 A-2 that plays a crucial role in plant growth and development by regulating cell division cell growth, and cell death. Plant Physiol 144:1531–1545
Fang ZZ, Zhang YL, Lai CC, Lai ZX (2014) Developmental regulation of Ran-3 untranslated region duringsomatic embryogenesis in Dimocarpus longan Lour. Sci Horti 176:297–302
Clark GB, Lee D, Dauwalder M, Roux SJ (2005) Immunolocalization and histochemical evidence for the association of two different Arabidopsis annexins with secretion during early seedling growth and development. Planta 220:621–631
Hayes, MJ, Shao D, Bailly M, Moss SE (2006) Regulation of actin dynamics by annexin2. Embo J 25:1816–1826
Sun ZJ, Xu JQ, Song M, Wei JY, Tang QL, Wang ZM, Wang XJ (2012) Molecular cloning and expression analysis of annexin gene from cabbage. Acta Horti Sin 39 (8):1567–1574
Bar M, Sharfman M, Schuster S, Avni A (2009) The Coiled-Coil domain of EHD2 mediates inhibition of LeEix2 endocytosis and signaling. PLoS ONE 4:1–13
Boavida LC, Shuai B, Yu HJ, Pagnussat GC, Sundaresan V, McCormick S (2009) A collection of Ds insertional mutants associated with defects in male gametophyte development and function in Arabidopsis thaliana. Genetics 181:1369–1385
Jaillais Y, Santambrogio M, Rozier F, Fobis-Loisy I, Miege C (2007) The retromer protein VPS29 links cell polarity and organ initiation in plants. Cell 130:1057–1070
Harrison MS, Hung CS, Liu TT, Christiano R, Walther TC, Bure CG (2014) A mechanism for retromer endosomal coat complex assembly with cargo. PNAS 111:267–272
Baldwin A, Wardle A, Patel R, Dudley P, Park SK, Twell D, Inoue K, Jarvis P (2005) A molecular-genetic study of the Arabidopsis Toc75 gene family. Plant Physiol 138:715–733
Masters TA, Kendrick-Jones J, Bus F (2016) Calcium gets myosin VI ready for work. PNAS 113(9):2325–2327
Jost W, Baur A, Nick P, Reski R, Gorr G (2004) A large plant beta-tubulin family with minimal C-terminal variation but differences in expression. Gene 340:151–160
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, there is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the position presented in, or the review of, the manuscript entitled. And this article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Jing Zeng, Qiguo Gao authors have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zeng, J., Gao, Q., Shi, S. et al. Dissecting Pistil Responses to Incompatible and Compatible Pollen in Self-Incompatibility Brassica oleracea Using Comparative Proteomics. Protein J 36, 123–137 (2017). https://doi.org/10.1007/s10930-017-9697-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-017-9697-y