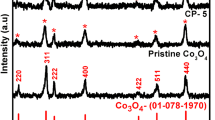
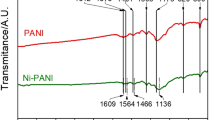
Abstract
As outstanding electrocatalytic materials, both metal oxide and conducting polymers have been synergically explored to fabricate electrochemical sensors. A novel electrochemical sensing interface was obtained by electrodeposition of the composite film based on polyaniline (PANI), carbon nanotubes (CNTs) and nickel oxide modified on lab-made screen-printed carbon electrode (SPE) for methanol electrooxidation in alkaline medium. The prepared composite electrode was characterized using various methods, including the Fourier-transform infrared spectroscopy (FT-IR), X-ray Diffraction pattern (XRD), Energy-dispersive X-ray spectroscopy (EDX) and Scanning electron microscope (SEM). The methanol electrochemical oxidation of NiO–NiOOH/PANI-CNTs/SPE in an alkaline medium was studied by using cyclic voltammetry (CV), chronoamperometry (CA) and electrochemical impedance spectroscopy (EIS). The obtained excellent sensing parameters including a sensor sensitivity of 8.056 × 10–5 mA/(mM cm2) with a limit detection LOD of 5 mM (S/N = 3) in wide range of methanol from 30 to 880 mM (R2 = 0.997) demonstrated that NiO–NiOOH/PANI-CNTs/SPE truly displayed highly electrochemical and catalytic activities for methanol electrooxidation. Furthermore, our proposed SPE sensors exhibited the good stability, repeatability and reproducibility that are crucial for on-site real sample detection and validation.









Similar content being viewed by others
References
L. O. Peres, R. W. C. Li, E. Y. Yamauchi, R. Lippi, and J. Gruber (2012). Food Chem. 130, 1105. https://doi.org/10.1016/j.foodchem.2011.08.014.
D. S. Islek and S. Ramadanoglu (2017). Med. Sci. Int. Med. J. 6, 372. https://doi.org/10.5455/medscience.2017.06.8588.
F. Bindler, E. Voges, and P. Laugel (1988). Food Addit. Contam. 5, 343. https://doi.org/10.1080/02652038809373713.
J. A. Kruse (2012). Crit. Care Clin. 28 (4), 661. https://doi.org/10.1016/j.ccc.2012.07.002.
H. Ghorbani, A. Nezami, B. Sheikholeslami, A. Hedjazi, and M. Ahmadimanesh (2018). J Occup. Med. Toxicol. 13, 1. https://doi.org/10.1186/s12995-017-0184-3(201.
H. Pontes, P. Guedes de Pinho, S. Casal, H. Carmo, A. Santos, T. Magalhães, F. Remião, F. Carvalho, and M. L. Bastos (2009). J. Chromatogr. Sci. 47, 272. https://doi.org/10.1093/chromsci/47.4.272.
A. I. Haj-Yehia and L. Z. Benet (1996). J. Chromatogr. A 724, 107. https://doi.org/10.1016/0021-9673(95)00986-8.
S. H. Chen, H. L. Wu, C. H. Yen, S. M. Wu, S. J. Lin, and H. S. Kou (1998). J. Chromatogr. A 799, 93. https://doi.org/10.1016/S0021-9673(97)01055-8.
W.-K. Kuo, H.-P. Weng, J.-J. Hsu, and H. H. Yu (2016). Mater. Chem. Phys. 173, 285. https://doi.org/10.1016/j.matchemphys.2016.02.014.
B. Tao, J. Zhang, S. Hui, X. Chen, and L. Wan (2010). Electrochim. Acta 55, 5019. https://doi.org/10.1016/j.electacta.2010.04.013.
M. M. Rahman, M. A. Hussein, K. A. Alamry, F. M. Al Shehry, and A. M. Asiri (2016). Talanta 150, 71. https://doi.org/10.1016/j.talanta.2015.12.012.
S. K. Verma, P. Kar, D. J. Yang, and A. Choudhury (2015). Sens. Actuators B 219, 199. https://doi.org/10.1016/j.snb.2015.04.117.
Q. Qiu, N. Jiang, L. Ge, X. Li, and X. Chen (2020). J. Mater. Sci. 55, 15681. https://doi.org/10.1007/s10853-020-05146-9.
M. Hasanzadeh and R. E. Sabzi (2015). Curr. Chem. Lett. 4, 45. https://doi.org/10.5267/j.ccl.2015.3.003.
D.-S. Park, M.-S. Won, R. N. Goyal, and Y.-B. Shim (2012). Sens. Actuators B 174, 45. https://doi.org/10.1016/j.snb.2012.08.017.
M. S. Lee, J. Sohn, J. Shim, and W. M. Lee (2007). Sens. Actuators B 124, 323. https://doi.org/10.1016/j.snb.2006.12.038.
J. Geng, X. Li, G. Sun, and B. Yi (2010). Sen. Actuators B 147, 612. https://doi.org/10.1016/j.snb.2010.03.047.
Y. Li, D. Deng, X. Xing, N. Chen, X. Liu, X. Xiao, and Y. Wang (2016). Sens. Actuators B 237, 133. https://doi.org/10.1016/j.snb.2016.06.088.
L. Chandra, R. Dwivedi, and V. N. Mishra (2017). Mater. Res. Express. 4, 105030. https://doi.org/10.1088/2053-1591/aa8dc0.
F.-T. Liu, S. F. Gao, S. K. Pei, S. C. Tseng, and C. H. J. Liu (2009). J. Taiwan Inst. Chem. Eng. 40 (5), 528. https://doi.org/10.1016/j.jtice.2009.03.008.
R. A. Hameed and K. El-Khatib (2010). Int. J. Hydrog. Energy 35 (6), 2517. https://doi.org/10.1016/j.ijhydene.2009.12.145.
J. P. J. de Oliveira, M. B. S. de Emeterio, A. C. de Sá, L. L. Paim, and M. de Valle (2019). Proceedings 42, 5. https://doi.org/10.3390/ecsa-6-06544.
M. R. Anderson, B. R. Mattes, H. Reiss, and R. B. Kaner (1991). Science 252, 1412. https://doi.org/10.1126/science.252.5011.1412.
O. Lupan, V. Cretu, M. Deng, D. Gedamu, I. Paulowicz, S. Kaps, Y. K. Mishra, O. Polonskyi, C. Zamponi, L. Kienle, V. Trofim, I. Tiginyanu, and R. Adelung (2014). J. Phys. Chem. C 118, 15068. https://doi.org/10.1021/jp5038415.
P. V. Hrkac, S. Kaps, V. Cretu, O. Lupan, T. Braniste, V. Duppel, and Y. K. Mishra (2015). Adv. Electron. Mater. 1, 1500081. https://doi.org/10.1002/aelm.201500081.
Q. Li, N. Li, J. An, and H. Pang (2020). Controllable synthesis of mesoporous NiO/Ni nanorod as an excellent catalyst for urea electrooxidation. Inorg Chem Front. https://doi.org/10.1039/d0qi00316f.
N. Behera, J. Duan, W. Jin, and S. Kitagawa (2021). The chemistry and applications of flexible porous coordination polymers. EnergyChem 3 (6), 100067. https://doi.org/10.1016/j.enchem.2021.100067.
Q. Li, S. Zheng, M. Du, and H. Pang (2021). Ultrathin nanosheet metal–organic framework@NiO/Ni nanorod composites. Chem. Eng. J. 417 (129201), 1385–8947. https://doi.org/10.1016/j.cej.2021.129201.
W. Li, X. Guo, P. Geng, M. Du, Q. Jing, X. Chen, G. Zhang, H. Li, Q. Xu, P. Braunstein, and H. Pang (2021). Rational design and general synthesis of multimetallic metal-organic framework nano-octahedra for enhanced Li–S battery. Adv. Mater. 33, 2105163. https://doi.org/10.1002/adma.202105163.
S. Zheng, Q. Li, H. Xue, H. Pang, and Q. Xu (2020). A highly alkaline-stable metal oxide@metal–organic framework composite for high-performance electrochemical energy storage. Natl. Sci. Rev. 7 (2), 305–314. https://doi.org/10.1093/nsr/nwz137.
A. B. Abou Hammad, M. E. Abd El-Aziz, M. S. Hasanin, and S. Kamel (2019). A novel electromagnetic biodegradable nanocomposite based on cellulose, polyaniline, and cobalt ferrite nanoparticles. Carbohydr. Polym. 216, 54–62. https://doi.org/10.1016/j.carbpol.2019.03.038.
A. M. Youssef, M. S. Hasanin, M. E. Abd El-Aziz, and G. M. Turky (2021). Conducting chitosan/hydroxylethyl cellulose/polyaniline bionanocomposites hydrogel based on graphene oxide doped with Ag-NPs. Int. J. Biol. Macromol. 167, 1435–1444. https://doi.org/10.1016/j.ijbiomac.2020.11.097.
M. Hasanin, E. A. Mwafy, and A. M. Youssef (2021). Electrical properties of conducting tertiary composite based on biopolymers and polyaniline. J. Bio Tribo Corros. 7, 133. https://doi.org/10.1007/s40735-021-00573-w.
M. S. Hasanin and S. A. Al Kiey (2020). Environmentally benign corrosion inhibitors based on cellulose niacin nano-composite for corrosion of copper in sodium chloride solutions. Int. J. Biol. Macromol. 161, 345–354. https://doi.org/10.1016/j.ijbiomac.2020.06.040.
N. T. K. Ngan, N. T. Thom, N. N. X. An, L. V. Hai, P. T. Nam, H. L. T. Nguyen, P. V. Viet, N. T. T. Trang, N. T. Hoang, and D. L. Tran (2021). Design of NiOOH/PANI-Gr and NiOOH/PANI-CNTs interfaces for sensitive and selective methanol electrochemical sensors. J. Electrochem. Soc. 168, 107509. https://doi.org/10.1149/1945-7111/ac2d44.
N. X. Nguyen, T. K. Nguyen, T. N. Pham, T. T. Nguyen, T. T. Nguyen, N. T. Hoang, and T. Dai Lam (2021). Efficient nickel or copper oxides decorated graphene–polyaniline interface for application in selective methanol sensing. RSC Adv. 11, 28573. https://doi.org/10.1039/d1ra04164a.
L. D. Tran, D. T. Nguyen, B. H. Nguyen, Q. P. Do, and H. Le Nguyen (2011). Development of interdigitated arrays coated with functional polyaniline/MWCNT for electrochemical biodetection: Application for human papilloma virus. Talanta 85, 1560. https://doi.org/10.1016/j.talanta.2011.06.048.
Y. Qiao, C. M. Li, S.-J. Bao, and Q.-L. Bao (2007). Carbon nanotube/polyaniline composite as anode material for microbial fuel cells. J. Power Sources 170, 79. https://doi.org/10.1016/j.jpowsour.2007.03.048.
Acknowledgements
This work was funded by the Vietnam Academy of Science and Technology (VAST) under the grant number NCXS 01.01/22-24.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nguyen, T.T., Pham, N.T., Nguyen, D.T. et al. PANI-CNTs Microstructure with Interconnected NiO–NiOOH Particles as Selective Sensing Interface for Methanol Electrochemical Sensor. J Clust Sci 34, 1259–1267 (2023). https://doi.org/10.1007/s10876-022-02297-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-022-02297-4