Abstract
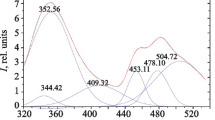
The binding of lanthanide(III) cations with p-isopropylcalix[n]arenes (n = 4, 6, 8) after partial deprotonation has been investigated using UV-Vis absorption spectrophotometry. The ease and degree of deprotonation of the calixarenes closely parallels the behaviour of their t-butyl analogues in the same solvents. The conditional stability constants of the praseodymium, europium, ytterbium and yttrium complexes were determined in DMF and in acetonitrile. The effect of water on the stability of lanthanide complexes in DMF was also treated. The results show that the cation binding selectivity, stoichiometry and stability of the complexes depend on various factors such as the degree of condensation (n) and the consequent number of O-donor atoms as well as the nature of the cation solvation. The stability of the Y(III), Pr(III), Eu(III) and Yb(III) complexes in DMF increased in all cases with increase of the ring size. DFT calculations were used to estimate the influence of the solvent on both the acidity and the conformations of the calixarene anions.

















Similar content being viewed by others
References
Gutsche, C.D.: Ch. 1. In: Vicens, J., Boehmer, V. (eds.) Calixarenes: A Versatile Class of Macrocyclic Compounds. Kluwer Academic Publishers, Dordrecht (1991)
Gutsche, C.D.: Calixarenes: An Introduction. RSC Publishing, Cambridge (2008)
Gutsche, C. D.: Calixarenes, Monographs in Supramolecular Chemistry No 1. In: Stoddart, J. F (ed.) Royal Society of Chemistry, Cambridge (1989). (revised as Calixarenes Revisited, 1998)
Böhmer, V.: Calixarenes, macrocycles with (almost) unlimited possibilities. Angew. Chem. Int. Ed. Engl. 34(7), 713–745 (1995)
Asfari, Z., Boehmer, V., Harrowfield, J., Vicens, J.: Calixarenes 2001. Kluwer, Dordrecht, The Netherlands (2001)
Zinke, A., Ziegler, E.: Zur Kenntnis des Härtungsprozesses von Phenol-Formaldehyd-Harzen, X. Mitteilung. Berichte der deutschen chemischen Gesellschaft (A and B Series) 77(3–4), 264–272 (1944)
Zinke, A., Ziegler, E.: Untersuchungen über Perylen und seine Derivate, LIII. Mitteil.: Synthese des 23, 1011‐Dibenz‐perylens. Berichte der deutschen chemischen Gesellschaft (A and B Series) 74(1), 115–118 (1941)
Cornforth, J.W., Hart, P.A., Nicholls, G.A., Rees, R.J.W., Stock, J.A.: Antituberculous effects of certain surface-active polyoxyethylene ethers. Br. J. Pharmacol. Chemother 10(1), 73–86 (1955)
Cornforth, J.W., Morgan, E.D., Potts, K.T., Rees, R.J.W.: Preparation of antituberculous polyoxyethylene ethers of homogeneous structure. Tetrahedron 29(11), 1659–1667 (1973)
Georghiou, P.E., Li, Z.: Calix [4] naphthalenes: cyclic tetramers of 1-naphthol and formaldehyde. Tetrahedron Lett. 34(18), 2887–2890 (1993)
Georghiou, P.E., Li, Z.: Conformational properties of the calix [4] naphthalenes. J. Incl. Phenomen. Mol. Recognit. Chem. 19(1–4), 55–66 (1994)
Poh, B.L., Ng, Y.Y.: Synthesis and conformations of a water-soluble cyclic tetramer obtained from reacting 5-sulfonatotropolone with formaldehyde. Tetrahedron 53(25), 8635–8642 (1997)
Yamato, T., Saruwatari, Y., Doamekpor, L.K., Hasegawa, K.I., Koike, M.: Preparation and conformational studies of ethylene-bridged calixarene-analogous macrocyclic metacyclophanes. Chem. Berichte. 126(11), 2501–2504 (1993)
Sorrell, T.N., Yuan, H.: Synthesis of covalently-linked exo-calix [4] arenes. J. Org. Chem. 62(6), 1899–1902 (1997)
Enright, G.D., Udachin, K.A., Ripmeester, J.A.: Electrostatic and short-range interactions compete in directing the structure of p-tert-butylcalix [4] arene inclusion compounds of fluorinated benzenes. Chem. Commun. 12, 1360–1361 (2004)
Udachin, K.A., Enright, G.D., Brown, P.O., Ripmeester, J.A.: Pseudopolymorphism in the p-tert-butylcalix [4] arene—n-butylamine system: directing the structural motifs. Chem. Commun. 18, 2162–2163 (2002)
Brouwer, E., Udachin, K., Enright, G., Ratcliffe, C., Ripmeester, J.: A chlorophobic pocket in the p-tert-butylcalix [4] arene cavity: a test site for molecular recognition investigated by 13 C CP MAS NMR and X-ray crystallography. Chem. Commun. 5, 587–588 (1998)
Meot-Ner, M.: The ionic hydrogen bond. Chem. Rev. 105(1), 213–284 (2005)
Vicens, J., Harrowfield, J. (eds.): Calixarenes in the Nanoworld. Springer, Dordrecht (2007)
Mandolini, L., Ungaro, R.: Calixarenes in Action. Imperial College Press, London (2000)
Redshaw, C.: Coordination chemistry of the larger calixarenes. Coord. Chem. Rev. 244(1), 45–70 (2003)
Homden, D.M., Redshaw, C.: The use of calixarenes in metal-based catalysis. Chem. Rev. 108(12), 5086–5130 (2008)
Lumetta, G.J., Rogers, R.D, Gopalan, A.S.: Calixarenes for separations, American Chemical Society Symposium Series (2000)
Li, Y., Zhao, K.-Q., Redshaw, C., Ortega, B. A. M., Nuñez, A. Y and Hanna, T. A.: Coordination chemistry and applications of phenolic calixarene–metal complexes, Patai’s Chemistry of Functional Groups (2014).
Araki, K., Iwamoto, K., Shinkai, S., Matsuda, T.: “ pKa” of calixarenes and analogs in nonaqueous solvents. Bull. Chem. Soc. Jpn. 63(12), 3480–3485 (1990)
Cunningham, I.D., Woolfall, M.: Deprotonation of calixarenes in acetonitrile. J. Org. Chem. 70(23), 9248–9256 (2005)
Miñambres, G.G., Jaques, T.S., Veglia, A.V., Lazzarotto, M., Nachtigall, F.F.: Counter-ion and solvent effects on the acidity of calix [4] arene and para-tert-butylcalix [6] arene. J. Incl. Phenomen. Macrocycl. Chem. 81(3–4), 311–319 (2014)
Charbonnière, L.J., Balsiger, C., Schenk, K.J., Bünzli, J.C.G.: Complexes of p-tert-butylcalix [5] arene with lanthanides: synthesis, structure and photophysical properties†. J. Chem. Soc. Dalton Trans. 3, 505–510 (1998)
Arnaud-Neu, F., Schwing-Weill, M.J., Dozol, J.F.: Calixarenes for nuclear waste treatment. In: Asfari, Z., Böhmer, V., Harrowfield, J.M., Vicens, J. (eds.) Calixarenes 2001, pp. 642–662. Kluwer Academic Publishers, Dordrecht (2001)
Izatt, R.M., Lamb, J.D., Hawkins, R.T., Brown, P.R., Izatt, S.R., Christensen, J.J.: Selective M+–H+ coupled transport of cations through a liquid membrane by macrocyclic calixarene ligands. J. Am. Chem. Soc. 105(7), 1782–1785 (1983)
Atwood, J.L., Coleman, A.W., Zhang, H., Bott, S.G.: Organic clays. Synthesis and structure of Na5 [calix [4] arene sulfonate] 12 H2O, K5 [calix [4] arene sulfonate] 8 H2O, Rb5 [calix [4] arene sulfonate] 5 H2O, and Cs5 [calix [4] arene sulfonate] 4 H2O. J. Incl. Phenom. Mol. Recognit. Chem. 7(2), 203–211 (1989)
Coleman, A.W., Bott, S.G., Morley, S.D., Means, C.M., Robinson, K.D., Zhang, H., Atwood, J.L.: Novel layer structure of sodium calix [4] arenesulfonate complexes—a class of organic clay mimics? Angew Chem. Int. Ed. Engl. 27(10), 1361–1362 (1988)
Harrowfield, J. M., Koutsantonis, G. A. In: Vicens, J., Harrowfield, J., Baklouti, L (Eds) Calixarenes in the Nanoworld, Ch 10, pp. 197–212. Springer, Dordrecht (2007).
Furphy, B.M., Harrowfield, J.M., Ogden, M.I., Skelton, B.W., White, A.H., Wilner, F.R.: Lanthanide ion complexes of the calixarenes. Part 4. Double inclusion by p–t-butylcalix [4] arene (H 4 L). Crystal structures of [Eu 2 (HL) 2 (dmf) 4]. 7dmf (dmf= dimethylformamide) and H 4 L. dmso (dmso= dimethyl sulphoxide). J. Chem. Soc. Dalton Trans. 11, 2217–2221 (1989)
Furphy, B.M., Harrowfield, J.M., Kepert, D.L., Skelton, B.W., White, A.H., Wilner, F.R.: Bimetallic lanthanide complexes of the calixarenes: europium(III) and tert-butylcalix [8] arene. Inorg. Chem. 26(25), 4231–4236 (1987)
Fleming, S., Gutsche, C.D., Harrowfield, J.M., Ogden, M.I., Skelton, B.W., Stewart, D.F., White, A.H.: Calixarenes as aryloxides: oligonuclear europium (III) derivatives. Dalton Trans. 17, 3319–3327 (2003)
Engelhardt, L.M., Furphy, B.M., Harrowfield, J.M., Kepert, D.L., White, A.H., Wilner, F.R.: Coordination chemistry of the calixarenes. II. Europium (III) and pt-butyl calix [6] arene. Aust. J. Chem. 41(9), 1465–1476 (1988)
Furphy, B. M: Honours Thesis, University of Western Australai, Perth (1987)
Dalla Cort, A., Mandolini, L.: Calixarenes as hosts for quats. In: Mandolini, L., Ungaro, R. (eds.) Calixarenes in Action. Imperial College Press, London (2000)
Harrowfield, J. M., Richmond,W. R. and Sobolev, A. N. Springer Netherlands, Calixarenes 50th Anniversary: Commemorative Issue. 257-276 (1994)
Harrowfield, J.M., Richmond, W.R., Sobolev, A.N., White, A.H.: Alkylammonium cation interactions with calixarene anions. Part 2. Structural characterisation of a salt of 2: 3 cation: calixarene stoichiometry. J. Chem. Soc. Perkin Trans. 2(1), 5–9 (1994)
Gutsche, C.D., Iqbal, M., Alam, I.: Calixarenes. 20. The interaction of calixarenes and amines. J. Am. Chem. Soc. 109(14), 4314–4320 (1987)
Thuéry, P., Asfari, Z., Nierlich, M., Vicens, J.: Two triethylammonium ion complexes of monoanionic calix [4] arene. Acta Crystallog. Sect. C 58(4), 223–225 (2002)
Abidi, R., Baker, M.V., Harrowfield, J.M., Ho, D.S.C., Richmond, W.R., Skelton, B.W., White, A.H., Wipff, G.: Complexation of the p–t-butyl-calix [4] arene anion with alkali metal cations in polar, non-aqueous solvents: Experimental and theoretical studies. Inorg. Chim Acta 246(1), 275–286 (1996)
Harrowfield, J.M., Ogden, M.I., Richmond, W.R., Skelton, B.W., White, A.H.: Cation–calixarene interactions: tetraalkylammonium cation binding by calixarene anions. J. Chem. Soc. Perkin Trans. 2(11), 2183–2190 (1993)
Cox, B.G.: Acids and Bases: Solvent Effects on Acid-Base Strength, p. 136. Oxford University Press, Oxford (2013)
Kaljurand, I., Kütt, A., Sooväli, L., Rodima, T., Mäemets, V., Leito, I., Koppel, I.A.: Extension of the self-consistent spectrophotometric basicity scale in acetonitrile to a full span of 28 p K a units: unification of different basicity scales. J. Org. Chem. 70(3), 1019–1028 (2005)
Mayer, U., Gutmann, V., Gerger, W.: The acceptor number—a quantitative empirical parameter for the electrophilic properties of solvents. Chem. Mon 106(6), 1235–1257 (1975)
Helm, L., Merbach, A.E.: Inorganic and bioinorganic solvent exchange mechanisms. Chem. Rev. 105(6), 1923–1960 (2005)
Bunzli, J.C.G., Froidevaux, P., Harrowfield, J.M.: Complexes of lanthanoid salts with macrocyclic ligands. 41. Photophysical properties of lanthanide dinuclear complexes with p-tert-butylcalix [8] arene. Inorg. Chem. 32(15), 3306–3311 (1993)
Bünzli, J.C.G., Ihringer, F.: Photophysical properties of lanthanide dinuclear complexes with p-nitro-calix [8] arene. Inorg. Chim Acta 246(1), 195–205 (1996)
Sillen, L.G., Warnqvist, B.: High-speed computers as a supplement to graphical methods. 6. A strategy for 2-level letagrop adjustment of common and group parameters. Some features that avoid divergence. Arkiv. Kemi. 31, 377–390 (1968)
Bunzli, J.C.G., Choppin, G.R.: Lanthanide Probes in Life, Chemical and Sciences. Elsevier Scientific Publ.B.V, Amsterdam (1989)
Bilyk, A., Dunlop, J.W., Fuller, R.O., Hall, A.K., Harrowfield, J.M., Hosseini, M., Koutsantonis, G.A., Murray, I.W., Skelton, B.W., Sobolev, A.N., Stamps, R.L., White, A.H.: Systematic structural coordination chemistry of p-tert-butyltetrathiacalix [4] arene: Further complexes of lanthanide metal ions. Eur. J. Inorg. Chem. 2010(14), 2127–2152 (2010)
Kim, K.H., Park, S.J., Choe, J.I.: DFT Conformational Study of Calix [5] arene and Calix [4] arene: hydrogen bond. Bull. Korean Chem. Soc. 29(10), 1893–1897 (2008)
Ugliengo, P., Viterbo, D., Chiari, Z.G.: Kristallogr. MOLDRAW: molecular graphics on a personal. Computer 9, 207 (1993)
Gutsche, C.D., Iqbal, M., Stewart, D.: Calixarenes. 19. Syntheses procedures for p-tert-butylcalix [4] arene. J. Org. Chem. 51(5), 742–745 (1986)
Arnaud-Neu, F., Schwing-Weill, M.J., Ziat, K., Cremin, S., Harris, S.J., McKervey, M.A.: Selective alkali and alkaline earth cation complexation by calixarene amides. N. J. Chem. 15(1), 33–37 (1991)
Ingri, N., Kakolowicz, W., Sillén, L.G., Warnqvist, B.: High-speed computers as a supplement to graphical methods—V: Haltafall, a general program for calculating the composition of equilibrium mixtures. Talanta 14(11), 1261–1286 (1967)
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian 09, Revision A.02, Gaussian, Inc., Wallingford CT, 2009.
Becke, A.D.: Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98(7), 5648–5652 (1993)
Lee, C., Yang, W., Parr, R.G.: Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37(2), 785–789 (1988)
Perdew, J.P., Burke, K., Ernzerhof, M.: Generalized gradient approximation made simple. Phys. Rev. Lett. 77(18), 3865–3868 (1996)
Durand, P., Barthelat, J.C.: A theoretical method to determine atomic pseudopotentials for electronic structure calculations of molecules and solids. Theor Chim Acta 38(4), 283–302 (1975)
Bouteiller, Y., Mijoule, C., Nizam, M., Barthelat, J.C., Daudey, J.P., Pelissier, M., Silvi, B.: Extended gaussian-type valence basis sets for calculations involving non-empirical core pseudopotentials: I. PS-31 G basis for Li to Ca and Ga to Kr atoms. Mol. Phys. 65(2), 295–312 (1988)
Acknowledgments
We are grateful to the Tunisian Ministry of High Education and Scientific Research and Technology for financial support as well as a fellowship from the Algerian Ministry of Higher Education and Scientific Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamzi, I., Essalah, K., Arnaud-Neu, F. et al. Lanthanide ion complexes of deprotonated p-isopropylcalix[n]arenes in dipolar aprotic solvents. J Incl Phenom Macrocycl Chem 85, 69–82 (2016). https://doi.org/10.1007/s10847-016-0606-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-016-0606-3