Abstract
Purpose
The fertility of women decreases with age because of factors such as an increased incidence of aneuploidies and—possibly—decreased mitochondrial activity in oocytes. However, the relationship between maternal aging and mitochondrial function of their embryos remains unknown. Here, we assessed the relationship between maternal age and mitochondrial functions in their oocytes and embryos
Methods
The relationships between maternal age and oxygen consumption rates (OCRs), mitochondrial DNA (mtDNA) copy numbers, or blastocyst development was investigated using 81 embryos donated from 63 infertility couples. The developmental rates from morulae to blastocysts were retrospectively analyzed using data of 105 patients.
Results
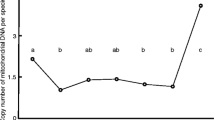
The OCRs of morulae decreased with maternal age (r2 = 0.48, P < 0.05) although there were no relationships between maternal age and mtDNA copy number in any stages. The more oxygen consumed at the morula stage, the shorter time was required for embryo development to the mid-stage blastocyst (r2 = 0.236, P < 0.05). According to the clinical data analysis, the developmental rate from morulae to blastocysts decreased with maternal age (P < 0.05, < 37 years, 81.1%, vs. ≥ 37 years, 64.1%).
Conclusions
The data of the present study revealed that mitochondrial function at the morula stage of human embryos decreased with their maternal age and a decrease of mitochondrial function led to slow-paced development and impaired developmental rate from morulae to blastocysts.



Similar content being viewed by others
References
Cimadomo D, Fabozzi G, Vaiarelli A, Ubaldi N, Ubaldi FM, Rienzi L. Impact of maternal age on oocyte and embryo competence. Front Endocrinol. 2018;29:327.
Nagaoka SI, Hassold TJ, Hunt PA. Human aneuploidy: mechanisms and new insights into an age-old problem. Nat Rev Genet. 2012;13:493–504.
Sakakibara Y, Hashimoto S, Nakaoka Y, Kouznetsova A, Höög C, Kitajima TS. Bivalent separation into univalents precedes age-related meiosis I errors in oocytes. Nat Commun. 2015;6:7550.
Shigenaga MK, Hagen TM, Ames BN. Oxidative damage and mitochondrial decay in aging. Proc Natl Acad Sci U S A. 1994;91:10771–8.
Bartmann AK, Romão GS, Ramos Eda S, Ferriani RA. Why do older women have poor implantation rates? A possible role of the mitochondria. J Assist Reprod Genet. 2004;21:79–83.
May-Panloup P, Chrétien MF, Jacques C, Vasseur C, Malthièry Y, Reynier P. Low oocyte mitochondrial DNA content in ovarian insufficiency. Hum Reprod. 2005;20:593–7.
Bentov Y, Yavorska T, Esfandiari N, Jurisicova A, Casper RF. The contribution of mitochondrial function to reproductive aging. J Assist Reprod Genet. 2011;28:773–83.
Muller-Hocker J, Schafer S, Weis S, Munscher C, Strowitzki T. Morphological-cytochemical and molecular genetic analyses of mitochondria in isolated human oocytes in the reproductive age. Mol Hum Reprod. 1996;2:951–8.
Van Blerkom J. Mitochondrial function in the human oocyte and embryo and their role in developmental competence. Mitochondrion. 2011;1:797–813.
Chan CC, Liu VW, Lau EY, Yeung WS, Ng EH, Ho PC. Mitochondrial DNA content and 4977 bp deletion in unfertilized oocytes. Mol Hum Reprod. 2005;11:843–6.
Murakoshi Y, Sueoka K, Takahashi K, Sato S, Sakurai T, Tajima H, et al. Embryo developmental capability and pregnancy outcome are related to the mitochondrial DNA copy number and ooplasmic volume. J Assist Reprod Genet. 2013;30:1367–75.
Thompson JG, Partridge RJ, Houghton FD, Cox CI, Leese HJ. Oxygen uptake and carbohydrate metabolism by in vitro derived bovine embryos. J Reprod Fertil. 1996;106:299–306.
Trimarchi JR, Liu L, Porterfield DM, Smith PJ, Keefe DL. Oxidative phosphorylation-dependent and -independent oxygen consumption by individual preimplantation mouse embryos. Biol Reprod. 2000;62:1866–74.
Ottosen LD, Hindkjaer J, Lindenberg S, Ingerslev HJ. Murine pre-embryo oxygen consumption and developmental competence. J Assist Reprod Genet. 2007;24:359–65.
Hashimoto S, Morimoto N, Yamanaka M, Matsumoto H, Yamochi T, Goto H, et al. Quantitative and qualitative changes of mitochondria in human preimplantation embryos. J Assist Reprod Genet. 2017;34:573–80.
Magnusson C, Hillensjo T, Hamberger L, Nilsson L. Oxygen consumption by human oocytes and blastocysts grown in vitro. Hum Reprod. 1986;1:183–4.
Yamanaka M, Hashimoto S, Amo A, Ito-Sasaki T, Abe H, Morimoto Y. Developmental assessment of human vitrified-warmed blastocysts based on oxygen consumption. Hum Reprod. 2011;26:3366–71.
Maezawa T, Yamanaka M, Hashimoto S, Amo A, Ohgaki A, Nakaoka Y, et al. Possible selection of viable human blastocysts after vitrification by monitoring morphological changes. J Assist Reprod Genet. 2014;31:1099–104.
Hashimoto S, Kato N, Saeki K, Morimoto Y. Selection of high potential embryos by culture in poly-(dimethylsiloxane) microwells and time-lapse imaging. Fertil Steril. 2012;97:332–7.
Biggers JD, Racowsky C. The development of fertilized human ova to the blastocyst stage in KSOMAA medium: is a two-step protocol necessary? Reprod BioMed Online. 2002;5:133–40.
Gardner DK, Lane M. Embryo culture systems. In: Trounson AO, Gardner DK, editors. Handbook of in vitro fertilization. 2nd ed. Boca Raton: CRC Press; 1999. p. 205–64.
Hashimoto S, Nakano T, Yamagata K, Inoue M, Morimoto Y, Nakaoka Y. Multinucleation per se is not always sufficient as a marker of abnormality to decide against transferring human embryos. Fertil Steril. 2016;106:133–9.
Kuwayama M, Vajta G, Leda S, Kato O. Comparison of open and closed methods for vitrification of human embryos and the elimination of potential contamination. Reprod BioMed Online. 2005;11:608–14.
Shiku H, Shiraishi T, Aoyagi S, Utsumi Y, Matsudaira M, Abe H, et al. Respiration activity of single bovine embryos entrapped in a cone-shaped microwell monitored by scanning electrochemical microscopy. Anal Chim Acta. 2004;522:51–8.
Sathananthan AH, Trounson AO. Mitochondrial morphology during preimplantational human embryogenesis. Hum Reprod. 2000;15(Suppl 2):148–59.
Watson AJ. The cell biology of blastocyst development. Mol Reprod Dev. 1992;33:492–504.
Diez-Juan A, Rubio C, Marin C, Martinez S, Al-Asmar N, Riboldi M, et al. Mitochondrial DNA content as a viability score in human euploid embryos: less is better. Fertil Steril. 2015;104:534–41.
Fragouli E, Spath K, Alfarawati S, Kaper F, Craig A, Michel CE, et al. Altered levels of mitochondrial DNA are associated with female age, aneuploidy, and provide an independent measure of embryonic implantation potential. PLoS Genet. 2015;11:e1005241.
Tan Y, Yin X, Zhang S, Jiang H, Tan K, Li J, et al. Clinical outcome of preimplantation genetic diagnosis and screening using next generation sequencing. Gigascience. 2014;3:30.
Leese HJ. Quiet please, do not disturb: a hypothesis of embryo metabolism and viability. Bioessays. 2002;24:845–9.
Victor AR, Brake AJ, Tyndall JC, Griffin DK, Zouves CG, Barnes FL, et al. Accurate quantitation of mitochondrial DNA reveals uniform levels in human blastocysts irrespective of ploidy, age, or implantation potential. Fertil Steril. 2017;107:34–42.
Acknowledgments
The authors thank Drs. M. Inoue, G. Udayanga, A. Takeshita, and T. Yamochi for their helpful comments, and James Cummins, PhD, from EDANZ Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Authors’ roles
N.M. and S.H. designed the experiment, interpreted the results, and wrote the manuscript with help from all authors. M.Y. measured OCRs. T.N., M.S., and Y.N. were involved in the analysis of clinical data. H.I. designed the qPCR procedures. A.F., H.S., and Y.M. supervised the project.
Funding
Part of this work was supported by a grant from the Japan Agency for Medical Research and Development (17gk0110014h0002 and 18gk0110014h0003 to S.H. and Y.M.), and a grant from the Japan Society for the Promotion of Science (KAKENHI 17K08144 and 20K09674 to S.H.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they no conflict of interest exists.
Ethical approval
The study was performed in accordance with Declaration of Helsinki protocols 7th version. This study was approved by the local ethics Institutional Review Board of IVF Namba Clinic, the institutional review board of Hyogo College of Medicine, and the Japan Society of Obstetrics and Gynecology (Registry Nos 135 and 138).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Fig. 1
Experimental design (PPTX 50 kb).
Rights and permissions
About this article
Cite this article
Morimoto, N., Hashimoto, S., Yamanaka, M. et al. Mitochondrial oxygen consumption rate of human embryos declines with maternal age. J Assist Reprod Genet 37, 1815–1821 (2020). https://doi.org/10.1007/s10815-020-01869-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-020-01869-5