Abstract
Purpose
In search for a new marker of preimplantation embryo viability the present study investigated oxygen consumption of individual cleavage stage murine embryos, and evaluated the predictive value regarding subsequent development to expanded blastocysts.
Methods
In all, 248 embryos were investigated from 2 cell stage until blastocyst stage with individual measurement of oxygen consumption and recording of developmental stage. Cleavage stage embryos and morula were divided in groups according to their oxygen consumption, and odds ratios (OR) for subsequent development to expanded blastocyst were calculated.
Results
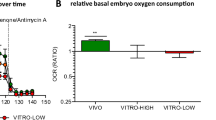
Cleavage stage (2–8 cell) individual oxygen consumption was 0.16–0.20 nl O2 h−1, with a significant increase to 0.21–0.23 nl O2 h−1 at the morula stage followed by a more than twofold increase for the expanded blastocyst 0.47 nl O2 h−1. A significantly higher chance of reaching the expanded blastocyst stage was found in 4-cell embryos with high oxygen consumption, than embryos with low consumption (OR 2.25, 95% CI 1.04–4.90). Among 2-cell embryos the chance of low and high consumers was not significantly different. The method used in the present study somewhat compromised embryo development (51% blastocyst rate) compared to controls (80% blastocystrate) which could make our results less robust.
Conclusion
Preliminary data from the present study suggest that oxygen consumption in cleavage stage embryos may be an indicator, but a not a strong predictor, of subsequent development to expanded blastocysts.

Similar content being viewed by others
References
Andersen AN, Gianaroli L, Felberbaum R, et al. Assisted reproductive technology in Europe, 2002. Results generated from European registers by ESHRE. Hum Reprod 2006;21:1680–97.
Ziebe S, Petersen K, Lindenberg S, et al. Embryo morphology or cleavage stage: how to select the best embryos for transfer after in-vitro fertilization. Hum Reprod 1997;12:1545–9.
Gardner DK, Lane M. Culture and selection of viable blastocysts: a feasible proposition for human IVF? Hum Reprod Updat 1997;3:367–82.
Blake D, Proctor M, Johnson N, Olive D. Cleavage stage versus blastocyst stage embryo transfer in assisted conception 2005 Cochrane Database of Systematic Reviews, Issue 4. Art. No.: CD002118. doi:10.1002/14651858.CD002118.pub2.
Leese HJ. What does an embryo need? Human Fertil 2003;6:180–5.
Janny L, Menezo YJ. Evidence for a strong effect on human preimplantation embryo development and blastocystformation. Mol Reprod Dev 1994;38(1):36–42.
Miller JE, Smith TT. The effect of intracytoplasmic sperm injection and semen parameters on blastocyst development in vitro. Hum Reprod 2001;16:918–24.
Brison DR, Leese HJ. Energy metabolism in late preimplantation rat embryos. J Reprod Fertil 1991;93(1):245–51.
Houghton FD, Hawkhead JA, Humpherson PG, et al. Non-invasive amino acid turnover predicts human embryo developmental capacity. Hum Reprod 2002;17:999–1005.
Gardner DK, Leese HJ. Assessment of embryo viability prior to transfer by the noninvasive measurement of glucose uptake. J Exp Zool 1987;242:103–5.
Overstrom EW. Manipulation of early embryonic development. Anim Reprod Sci 1992;28:277–85.
Houghton FD, Thompson JG, Kennedy CJ, et al. Oxygen consumption and energy metabolism of the early mouse embryo. Mol Reprod Dev 1996;44:476–85.
Kane MT, Buckley NJ. The effects of inhibitors of energy metabolism on the growth of one-cell rabbit ova to blastocysts in vitro. J Reprod Fertil 1977;49(2):261–6.
Thompson JG, Partridge RJ, Houghton FD, Cox CI, Leese HJ. Oxygen uptake and carbohydrate metabolism by in vitro derived bovine embryos. J Reprod Fertil 1996;106(2):299–306.
Liu L, Trimarchi JR, Keefe DL. Thiol oxidation-induced embryonic cell death in mice is prevented by the antioxidant dithiothreitol. Biol Reprod 1999;61:1162–9.
Crompton M. The mitochondrial permeability transition pore and its role in cell death. Biochem J 1999;341:233–49.
Gross A, McDonnell JM, Korsmeyer SJ. BCL-2 family members and the mitochondria in apoptosis. Genes Dev 1999;13:1899–911.
Trimarchi JR, Lui L, Porterfield DM, et al. Oxidative phosphorylation dependent and independent oxygen consumption by individual preimplantation mouse embryos. Biol Reprod 2000;62:1866–74.
Lopes AS, Larsen LH, Rasming N, et al. Respiration rate of individual bovine in vitro produced embryos measured with a novel, non invasive and highly sensitive microsensor system. Reproduction 2005;130:669–79.
Mills RM, Brinster RL. Oxygen consumption of preimplantation mouse embryos. Exp Cell Res 1967;47:337–44.
Barnett DK, Bavister BD. What is the relationship between the metabolism of preimplantation embryos and their developmental competence? Mol Reprod Dev 1996;43(1):105–33.
Acknowledgements
This work was supported by the Danish Medical Research Council, The Beckett Foundation, The Foundation of 17/12–1981, The Toyota Foundation, the A.P. Møller Foundation for the advancement of Medical Science, Karen Elise Jensens Foundation, Clinical Institute Aarhus University and Organon Denmark. Thanks to Morten Raaschou Image House Medical A/S for providing the Fertimorph equipment used in this study. Thanks to Puk Sandager for statistical advice.
Ethics
The use of experimental animals is approved and controlled by the Faculty Veterinary and follows the regulations of the National Committee of Experimental Animals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ottosen, L.D.M., Hindkjær, J., Lindenberg, S. et al. Murine pre-embryo oxygen consumption and developmental competence. J Assist Reprod Genet 24, 359–365 (2007). https://doi.org/10.1007/s10815-007-9138-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-007-9138-5