Abstract
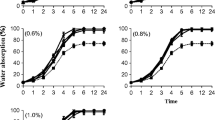
Experimental alkaline and blended seaweed liquid extracts (ASLE and BSLE, respectively) were produced from red seaweeds (Acanthophora spicifera, Gelidium robustum, and Gracilaria parvispora) and brown seaweeds (Macrocystis pyrifera, Sargassum horridum, and Ecklonia arborea). The effects as biostimulants on the germination and growth of the mung bean (Vigna radiata) were investigated. Variable concentrations (0.06, 0.12, 0.25, 0.5, 1, and 2%) of ASLE and BSLE were applied. There was a significant difference between the effects of the varying seaweed liquid extracts on the mung bean. The maximum increment in the germination percent (9%) over the control was obtained using the 0.5% BSLE from M. pyrifera and G. robustum (MPGR) administered of the original extract, as well as using the 2% BSLE from E. arborea and G. robustum. The best biostimulant effect was observed in the mean shoot length, with an increase of 39%, compared to the control using 0.25% ASLE from E. arborea. Additionally, a maximum total average length increase of 27% was obtained using 2% ASLE from E. arborea, and an increase of 25% was obtained with a 0.25% blend composed of E. arborea and G. parvispora. Compared to the control, the maximum dry weight average with an increase of 45% was obtained with a 0.05% BSLE produced from E. arborea and G. parvispora applied. Overall, the blended extracts from the brown E. arborea and the red G. parvispora showed the best biostimulant effect as shown in the increase of length and weight of the mung bean, compared to all other BSLE tested. This result suggests that the biostimulant activity was enhanced when two extracts were blended, compared with the effect of individual seaweed liquid extracts. The application of algal blends as formulations with biostimulant properties for seed coating/soaking is a promising option for a sustainable agriculture which aims to reduce the use of mineral fertilizers and pesticides. However, more research is necessary to elucidate the relationship between the biostimulant activity and the extract chemical composition.






Similar content being viewed by others
References
AOAC (Association of Official Analytical Chemists) (1990) Official methods of analysis, AOAC 15th edn. AOAC, Washington DC
AOAC (Association of Official Analytical Chemists) (2000) Official methods of analysis of the AOAC international. In: Horwitz W (ed), vol 1, 17th edn. Inc. Washington DC
AOSA (Association of Official Seed Analysts) (2005) In: Rules for testing seed. Capashew (ed), Las Cruces, 4
Arioli T, Mattner SW, Winberg PC (2015) Applications of seaweed extracts in Australian agriculture: past, present and future. J Appl Phycol 27:2007–2015
Avila E, Méndez-Trejo MC, Riosmena-Rodríguez R, López-Vivas JM, Senties A (2012) Epibiotic traits of the invasive red seaweed Acanthophora spicifera in La Paz Bay, South Baja California (Eastern Pacific). Mar Ecol 33:470–480
Babu S, Rengasamy R (2012) Effect of Kappaphycus alvarezii SLF treadment on seed germination, growth and development of seedling in some crop plants. J Acad Indus Res 1:186–195
Bharath B, Nirmalraj S, Mahendrakumar M, Perinbam K (2018) Biofertilizing efficiency of Sargassum polycystum extract on growth and biochemical composition of Vigna radiata and Vigna mungo. Asian Pac J Reprod 7:27–32
Briceño-Domínguez D, Hernández-Carmona G, Moyo M, Stirk W, van Staden J (2014) Plant growth promoting activity of seaweed liquid extracts produced from Macrocystis pyrifera under different pH and temperature conditions. J Appl Phycol 26:2203–2210
Casas-Valdez M, Lluch-Belda D, Ortega-García S, Hernández-Vázquez S, Serviere-Zaragoza E, Lora-Sánchez D (2005) Estimation of maximum sustainable yield of Gelidium robustum seaweed fishery in Mexico. J Mar Biol Assoc UK 85:775–778
Castellanos-Barriga LG, Santacruz-Ruvalcaba F, Hernández-Carmona G, Ramírez-Briones E, Hernández-Herrera RM (2017) Effect of seaweed extracts from Ulva lactuca on seedling growth of mung bean (Vigna radiata). J Appl Phycol 29:2479–2488
Chbani A, Mawlawi H, Zaouk L (2013) Evaluation of brown seaweed (Padina pavonica) as bioestimulant of plant growth and development. Afr J Agric Res 3:1155–1165
Colla G, Nardi S, Cardarelli M, Ertani A, Lucini L, Canaguier R, Rouphael Y (2015) Protein hydrolysates as biostimulants in horticulture. Sci Hortic 196:28–38
Craigie JS (2011) Seaweed extract stimuli in plant science and agriculture. J Appl Phycol 23:371–393
Dembitsky VM, Smoum R, Al-Quntar A, Ali HA, Pergament I, Srebnik M (2002) Natural occurrence of boron-containing compounds in plants, algae and microorganisms. Plant Sci 163:931–942
du Jardin P (2015) Review: plant biostimulants: definition, concept, main categories and regulation. Sci Hortic 196:3–14
Ertani A, Francioso O, Tinti A, Schiavon M, Pizzeghello D, Nardi S (2018) Evaluation of seaweed extracts from Laminaria and Ascophyllum nodosum spp. as biostimulants in Zea mays L. using a combination of chemical, biochemical and morphological approaches. Front Plant Sci 9:428
Godlewska K, Michalak I, Tuhy L, Chojnacka K (2016) Plant growth biostimulants based on different methods of seaweed extraction with water. Biomed Res Int 2016:1–11
Hernández-Carmona G, Casas-Valdez MM, Fajardo-León C, Sánchez Rodríguez I, Rodríguez-Montesinos EY (1990) Evaluación de Sargassum spp. en la Bahía de La Paz, B.C.S. México. Inv Mar CICIMAR 5:11–18
Hernández-Carmona G, Rodríguez-Montesinos EY, Casas-Valdez MM, Vilchis MA, Sánchez-Rodríguez I (1991) Evaluation of the beds of Macrocystis pyrifera (Phaeophyta, Laminariales) in the Baja California Peninsula, Mexico. III. Summer 1986 and seasonal variation. Cienc Mar 17:121–145
Hernández-Herrera RM, Santacruz-Ruvalcaba F, Ruíz-López MA, Norrie J, Hernández-Carmona G (2014) Effect of liquid seaweed extracts on growth of tomato seedlings (Solanum lycopersicum L.). J Appl Phycol 26:619–628
Hernández-Herrera RM, Santacruz-Ruvalcaba F, Zañudo-Hernández J, Hernández-Carmona G (2016) Activity of seaweed extracts and polysaccharide-enriched extracts from Ulva lactuca and Padina gymnospora as growth promoters of tomato and mung bean plants. J Appl Phycol 28:2549–2560
Hernández-Herrera RM, Santacruz-Ruvalcaba F, Briceño-Domínguez D, Di-Filippo-Herrera DA, Hernández-Carmona G (2018) Seaweed as potential plant growth stimulants for agriculture in Mexico. Hidrobiológica 28:129–140
Hong DD, Hien HM, Son PN (2007) Seaweeds from Vietnam used for functional food, medicine and biofertilizer. J Appl Phycol 19:817–826
Khan W, Rayirath UP, Subramanian S, Jithesh MN, Rayorath P, Hodges DM, Critchley AT, Craigie JS, Norrie J, Prithiviraj B (2009) Seaweed extracts as biostimulants of plant growth and development. J Plant Growth Regul 28:386–399
Krueger-Hadfield SA, Hernández-Carmona G, Terada R, López-Vivas JM, Riosmena-Rodríguez R (2016) New record of the non-native seaweed Gracilaria parvispora in Baja California—a note on Vergara-Rodante et al. (2016). Cryptogam Algol 37:257–263
Layek J, Das A, Idapuganti RG, Sarka D, Ghosh A, Zodape ST, Lal R, Yadav GS, Panwar AS, Ngachan S, Meena RS (2018) Seaweed extract as organic bio-stimulant improves productivity and quality of rice in eastern Himalayas. J Appl Phycol 30:547–558
Lourenco SO, Barbarino E, De-Paula JC (2002) Amino acid composition, protein content and calculation of nitrogen-to-protein conversion factors for 19 tropical seaweeds. Phycol Res 50:233–241
Mattner SW, Wite D, Riches DA, Porter IJ, Arioli T (2013) The effect of kelp extract on seedling establishment of broccoli on contrasting soil types in southern Victoria, Australia. Biol Agric Hortic 29:258–270
Mattner SW, Milinkovic M, Arioli T (2018) Increased growth response of strawberry roots to a commercial extract from Durvillaea potatorum and Ascophyllum nodosum. J Appl Phycol 30:2943–2951
McHugh DJ (2002) Prospects for seaweed production in developing countries. FAO Fisheries Circular No. 968 FIIU/C968
Méndez-Trejo MC, Riosmena-Rodríguez R, Ávila E, López-Vivas JM, Sentíes A (2014) Evaluación de la invasión de Acanthophora spicifera (Rhodophyta) sobre la epifauna en Bahía de La Paz, B.C.S. In: Low Pfeng AM, Quijón PA y Peters Recargo EM (eds) Especies invasoras acuáticas: casos de estudio en ecosistemas de México, 1st edn. Mexico, pp 431–456
Michalak I, Tunhy T, Chojnacka K (2015) Seaweed extract by microwave assisted extraction as plant growth biostimulant. Open Chem 13:1183–1195
Michalak I, Chojnacka K, Saeid A (2017) Plant growth biostimulants, dietary feed supplements and cosmetics formulated with supercritical CO2 algal extracts. Molecules 22:66
Nabti E, Jha B, Hartmann A (2017) Impact of seaweeds on agricultural crop production as biofertilizer. Int J Environ Sci Technol 14:1119–1134
Prakash P, Mitra A, Nag R, Sunkar S (2018) Effect of seaweed liquid fertilizer and humic acid formulation on the growth and nutritional quality of Abelmoschus esculentus. Asian J Crop Sci 10:48–52
Rayorath P, Khan W, Palanisamy R, MacKinnon SL, Stefanova R, Hankins SD, Critchley AT, Prithiviraj B (2008) Extracts of the brown seaweed Ascophyllum nodosum induce gibberellic acid (GA3)-independent amylase activity in barley. J Plant Growth Regul 27:370–379
Rothman MD, Mattio L, Wernberg T, Anderson RJ, Uwai S, Mohring MB, Bolton JJ (2015) A molecular investigation of the genus Eklonia (Phaeophyceae, Laminariales) with special focus on the southern hemisphere. J Phycol 51:236–246
Sharma HS, Fleming C, Selby C, Rao JR, Martin T (2014) Plant biostimulants: a review on the processing of macroalgae and use of extracts for crop management to reduce abiotic and biotic stresses. J Appl Phycol 26:465–490
Stirk WA, Van Staden J (1997) Comparison of cytokinin- and auxin-like activity in some commercially used seaweed extracts. J Appl Phycol 8:503–508
Sunarpi JA, Kurnianingsih R, Julisaniah NI, Tullah AN (2010) Effect of seaweed extracts on growth and yield of rice plants. Bioscience 2:73–77
Vergara-Rodarte MA, Hernández-Carmona G, Riosmena-Rodríguez R (2016) Seasonal variation in the biomass, size and reproduction of the agarophytes Gracilariopsis sp. (Gracilareaceae, Rhodophyta) from a temperate lagoon in the Pacific coast of Baja California Sur, México. Crytogam Algol 37:1–12
Vinoth S, Gurusaravanan P, Jayabalan N (2012a) Effect of seaweed extracts and plant growth regulators on high-frequency in vitro mass propagation of Lycopersicon esculentum L. (tomato) through double cotyledonary nodal explant. J Appl Phycol 24:1329–1337
Vinoth S, Gurusaravanan P, Jayabalan N (2012b) Erratum to: effect of seaweed extracts and plant growth regulators on high-frequency in vitro mass propagation of Lycopersicon esculentum L (tomato) through double cotyledonary nodal explant. J Appl Phycol 24:1339–1340
Vinoth S, Gurusaravanan P, Jayabalan N (2014) Optimization of somatic embryogenesis protocol in Lycopersicon esculentum L. using plant growth regulators and seaweed extracts. J Appl Phycol 26:1527–1537
Acknowledgments
The authors thank the journal reviewers for their valuable comments, suggestions, and guidance.
Funding
The authors wish to thank the Consejo Nacional de Ciencia y Tecnología (CONACyT) for the financial support and to the Instituto Politécnico Nacional–Centro Interdisciplinario de Ciencias Marinas (IPN-CICIMAR) for the funds and resources granted to carry out this research. We thank to the Mexican company Algas Pacific for helping to carrying out the investigation. Gustavo Hernández-Carmona and Mauricio Muñoz Ochoa express their thanks to the “Comisión de Operaciones y Fomento de Actividades Académicas del IPN (COFFA) and to the program “Estimulo al Desempeño de los Investigadores del IPN (EDI),” and the SNI for the fellowship granted.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Filippo-Herrera, D.A., Muñoz-Ochoa, M., Hernández-Herrera, R.M. et al. Biostimulant activity of individual and blended seaweed extracts on the germination and growth of the mung bean. J Appl Phycol 31, 2025–2037 (2019). https://doi.org/10.1007/s10811-018-1680-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-018-1680-2