Abstract
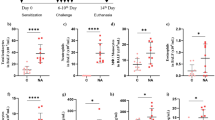
The response of lungs with emphysema to an acute lung injury (ALI) remains unclear. This study compared the lung response to intratracheal instillation of lipopolysaccharide (LPS) in rats with and without emphysema. Twenty-four Wistar rats were randomized to four groups: control group (C-G), ALI group (ALI-G), emphysema group (E-G), emphysema and ALI group (E-ALI-G). Euthanasia and the following analysis were performed 24 h after ALI induction: lung histology, bronchoalveolar lavage (BAL), mRNA expression of inflammatory mediators, and blood gas measures. The histological analysis showed that animals of ALI-G (0.55 ± 0.15) and E-ALI-G (0.69 ± 0.08) had a higher ALI score compared to C-G (0.12 ± 0.04) and E-G (0.16 ± 0.04) (p < 0.05). The analysis of each component of the score demonstrated that ALI-G and E-ALI-G had greater alveolar and interstitial neutrophil infiltration, as well as greater amount of alveolar proteinaceous debris. Comparing the two groups that received LPS, there was a trend of higher ALI in the E-ALI-G, specially due to a higher neutrophil infiltration in the alveolar spaces and a higher septal thickening. Total cell count (E-G = 3.09 ± 0.83; ALI-G = 4.45 ± 1.9; E-ALI-G = 5.9 ± 2.1; C-G = 0.73 ± 0.37 × 105) and neutrophil count (E-G = 0.69 ± 0.35; ALI-G = 2.53 ± 1.09; E-ALI-G = 3.86 ± 1.4; C-G = 0.09 ± 0.07 × 105) in the BAL were higher in the groups E-G, ALI-G, and E-ALI-G when compared to C-G (p < 0.05). The IL-6, TNF-α, and CXCL2 mRNA expressions were higher in the animals that received LPS (ALI-G and E-ALI-G) compared to the C-G and E-G (p < 0.05). No statistically significant difference was observed in the BAL cellularity and in the expression of inflammatory mediators between the ALI-G and the E-ALI-G. The severity of ALI in response to intratracheal instillation of LPS did not show difference in rats with and without intratracheal-induced emphysema.




Similar content being viewed by others
References
Macnee, W. 2005. Pathogenesis of chronic obstructive pulmonary disease. Proceedings of the American Thoracic Society 2 (4): 258–266.
Finkelstein, R., R.S. Fraser, H. Ghezzo, and M.G. Cosio. 1995. Alveolar inflammation and its relation to emphysema in smokers. American Journal of Respiratory and Critical Care Medicine 152 (5 Pt 1): 1666–1672.
Retamales, I., W. Elliott, B. Meshi, H.O. Coxson, P.D. Pare, F.C. Sciurba, R.M. Rogers, S. Hayashi, and J.C. Hogg. 2001. Amplification of inflammation in emphysema and its association with latent adenoviral infection. American Journal of Respiratory and Critical Care Medicine 164 (3): 469–473.
Baines, K.J., J.L. Simpson, and P.G. Gibson. 2011. Innate immune responses are increased in chronic obstructive pulmonary disease. PLoS One 6: e18426. https://doi.org/10.1371/journal.pone.0018426.
Hodge, S., G. Hodge, R. Scicchitano, P.N. Reynolds, and M. Holmes. 2003. Alveolar macrophages from subjects with chronic obstructive pulmonary disease are deficient in their ability to phagocytose apoptotic airway epithelial cells. Immunology and Cell Biology 81 (4): 289–296.
Berenson, C.S., C.T. Wrona, L.J. Grove, J. Maloney, M.A. Garlipp, P.K. Wallace, C.C. Stewart, and S. Sethi. 2006. Impaired alveolar macrophage response to Haemophilus antigens in chronic obstructive lung disease. American Journal of Respiratory and Critical Care Medicine 174 (1): 31–40.
Pang, B., W. Hong, S.L. West-Barnette, N.D. Kock, and W.E. Swords. 2008. Diminished ICAM-1 expression and impaired pulmonary clearance of nontypeable Haemophilus influenzae in a mouse model of chronic obstructive pulmonary disease/emphysema. Infection and Immunity 76 (11): 4959–4967.
Taylor, A.E., T.K. Finney-Hayward, J.K. Quint, C.M. Thomas, S.J. Tudhope, J.A. Wedzicha, P.J. Barnes, and L.E. Donnelly. 2010. Defective macrophage phagocytosis of bacteria in COPD. The European Respiratory Journal 35 (5): 1039–1047.
Kaur, M., and D. Singh. 2013. Neutrophil chemotaxis caused by chronic obstructive pulmonary disease alveolar macrophages: the role of CXCL8 and the receptors CXCR1/CXCR2. Journal of Pharmacology and Experimental Therapeutics 347 (1): 173–180.
Owen, C.A., M.A. Campbell, P.L. Sannes, S.S. Boukedes, and E.J. Campbell. 1995. Cell surface-bound elastase and cathepsin G on human neutrophils: a novel, non-oxidative mechanism by which neutrophils focus and preserve catalytic activity of serine proteinases. The Journal of Cell Biology 131 (3): 775–789.
Fonseca, L.M., M.M. Reboredo, L.M. Lucinda, T.F. Fazza, M.A. Rabelo, A.S. Fonseca, F. de Paoli, and B.V. Pinheiro. 2016. Emphysema induced by elastase enhances acute inflammatory pulmonary response to intraperitoneal LPS in rats. International Journal of Experimental Pathology 97 (6): 430–437.
Tokairin, Y., Y. Shibata, M. Sata, S. Abe, N. Takabatake, A. Igarashi, T. Ishikawa, S. Inoue, and I. Kubota. 2008. Enhanced immediate inflammatory response to Streptococcus pneumoniae in the lungs of mice with pulmonary emphysema. Respirology 13 (3): 324–332.
Inoue, S., H. Nakamura, K. Otake, H. Saito, K. Terashita, J. Sato, H. Takeda, and H. Tomoike. 2003. Impaired pulmonary inflammatory responses are a prominent feature of Streptococcal pneumonia in mice with experimental emphysema. American Journal of Respiratory and Critical Care Medicine 167 (5): 764–770.
Black, P.N., P.S.T. Ching, B. Beaumont, S. Ranasinghe, G. Taylor, and M.J. Merrilees. 2008. Changes in elastic fibres in the small airways and alveoli in COPD. The European Respiratory Journal 31 (3): 998–1004.
Matute-Bello, G., G. Downey, B.B. Moore, S.D. Groshong, M.A. Matthay, A.S. Slutsky, W.M. Kuebler, and Acute Lung Injury in Animals Study Group. 2011. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. American Journal of Respiratory and Critical Care Medicine 44 (5): 725–738.
Dunnill, M.S. 1962. Quantitative methods in the study of pulmonary pathology. Thorax 17 (4): 320–328.
Yokoyama, E., Z. Nambu, I. Uchiyama, and H. Kyono. 1987. An emphysema model in rats treated intratracheally with elastase. Environmental Research Journal 42 (2): 340–352.
Kırkıl, G., M.H. Muz, F. Deveci, T. Turgut, F. İlhan, and I. Özercan. 2007. Effect of bosentan on the production of proinflammatory cytokines in a rat model of emphysema. Experimental & Molecular Medicine 39 (5): 614–620.
Van de Lest, C.H.A., E.M.M. Versteeg, J.H. Veerkamp, and T.H. Van Kuppevelt. 1995. Digestion of proteoglycans in porcine pancreatic elastase-induced emphysema in rats. The European Respiratory Journal 8 (2): 238–245.
Vecchiola, A., J.F. de la Llera, R. Ramírez, P. Olmos, C.I. Herrera, and G. Borzone. 2011. Differences in acute lung response to elastase instillation in two rodent species may determine differences in severity of emphysema development. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 301 (1): R148–R158.
Landgraf, M.A., R.C. Silva, M. Corrêa-Costa, M.I. Hiyane, M.H. Carvalho, R.G. Landgraf, and N.O. Câmara. 2014. Leptin downregulaes LPS-induced lung injury: role of corticosterone and insulin. Cellular Physiology and Biochemistry 33 (3): 835–846.
Silva, P.L., L. Moraes, R.S. Santos, C. Samary, M.B. Ramos, C.L. Santos, M.M. Morales, V.L. Capelozzi, C.S. Garcia, M.G. de Abreu, P. Pelosi, J.J. Marini, and P.R. Rocco. 2013. Recruitment maneuvers modulate epithelial and endothelial cell response according to acute lung injury etiology. Critical Care Medicine 41 (10): e256–e265.
Kabir, K., J.P. Gelinas, M. Chen, D. Chen, D. Zhang, X. Luo, J.H. Yang, D. Carter, and R. Rabinovici. 2002. Characterization of a murine model of endotoxin-induced acute lung injury. Shock 17 (4): 300–303.
Santana, M.C., C.S. Garcia, D.G. Xisto, L.K. Nagato, R.M. Lassance, L.F. Prota, F.M. Ornellas, V.L. Capelozzi, M.M. Morales, W.A. Zin, P. Pelosi, and P.R. Rocco. 2009. Prone position prevents regional alveolar hyperinflation and mechanical stress and strain in mild experimental acute lung injury. Respiratory Physiology and Neurobiology 167 (2): 181–188.
Wiener-Kronish, J.P., K.H. Albertine, and M.A. Matthay. 1991. Differential responses of the endothelial and epithelial barriers of the lung in sheep to Escherichia coli endotoxin. Journal of Clinical Investigation 8 (3): 864–875.
Shaykhiev, R., and R.G. Crystal. 2013. Innate immunity and chronic obstructive pulmonary disease: a mini-review. Gerontology 59 (6): 481–489.
Sajjan, U., S. Ganesan, A.T. Comstock, J. Shim, Q. Wang, D.R. Nagarkar, Y. Zhao, A.M. Goldsmith, J. Sonstein, M.J. Linn, J.L. Curtis, and M.B. Hershenson. 2009. Elastase- and LPS-exposed mice display altered responses to rhinovirus infection. American Journal of Physiology, Lung Cellular and Molecular Physiology 297 (5): L931–L944.
Wang, D., Y. Wang, and Y.N. Liu. 2010. Experimental pulmonary infection and colonization of Haemophilus influenzae in emphysematous hamsters. Pulmonary Pharmacology and Therapeutics 23 (4): 292–299.
Funding
This study was funded by a research grant from Rede Mineira TOXIFAR, Fundação de Amparo a Pesquisa do Estado de Minas Gerais (FAPEMIG), and Center of Reproductive Biology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This study was approved by the Ethics Committee in Animal Experiments of Federal University of Juiz de Fora and all procedures were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Rabelo, M.A.E., Lucinda, L.M.F., Reboredo, M.M. et al. Acute Lung Injury in Response to Intratracheal Instillation of Lipopolysaccharide in an Animal Model of Emphysema Induced by Elastase. Inflammation 41, 174–182 (2018). https://doi.org/10.1007/s10753-017-0675-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0675-5