Abstract
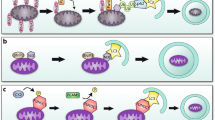
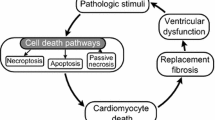
All multicellular organisms develop during evolution the highly regulated and interconnected pathways of cell death. This complex network contributes to the pathogenesis of various cardiovascular disorders including ischemia/reperfusion injury, myocardial infarction, heart failure, dysrhythmias and atherosclerosis. Chronic cardiac remodeling response and transition to overt HF have been associated with modestly increased apoptosis, although the actual burden of chronic cell loss attributable to apoptosis is not clear. Central mediators of cardiomyocyte survival and death are the mitochondrial organelles. Based on its morphological characteristics, cell death can be classified into three major types: apoptosis, necrosis and autophagy. Recently, a new pathway of regulated necrosis, necroptosis, has also been reported in the failing heart. The mitochondrial (intrinsic) and the death-receptor-mediated (extrinsic) converge at mitochondria inducing release of mitochondrial apoptogens to initiate the caspase cascade and eventually degradation of the doomed cardiomyocyte. Activation of death receptors can initiate not only extrinsic apoptotic pathway, but also necrosis. On the other hand, autophagy, which is characterized by the massive formation of lysosomal-derived vesicles, containing degenerating cytoplasmic contents, is primarily a survival response to nutrient deprivation, and a selective form of autophagy, mitophagy, is also a protective mechanism that allows to eliminate damaged mitochondria and thereby to attenuate mitochondria-mediated apoptosis and necrosis in the myocardium. Further insight into the molecular mechanisms underlying cell death will increase the efficiency and repertoire of therapeutic interventions available in cardiovascular disease.



Similar content being viewed by others
References
Wencker D, Chandra M, Nguyen K, Miao W, Garantziotis S et al (2003) A mechanistic role for cardiac myocyte apoptosis in heart failure. J Clin Investig 111:1497–1504
Hein S, Arnon E, Kostin S, Schonburg M, Elsasser A et al (2003) Progression from compensated hypertrophy to failure in the pressure-overloaded human heart: structural deterioration and compensatory mechanisms. Circulation 107:984–991
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Fuchs Y, Steller H (2011) Programmed cell death in animal development and disease. Cell 147:742–758
Micheau O, Tschopp J (2003) Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114:181–190
Peter ME, Krammer PH (2003) The CD95(APO-1/Fas) DISC and beyond. Cell Death Differ 10:26–35
Kischkel FC, Hellbardt S, Behrmann I, Germer M, Pawlita M et al (1995) Cytotoxicity-dependent APO-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. The EMBO journal 14:5579–5588
Pop C, Salvesen GS (2009) Human caspases: activation, specificity, and regulation. J Biol Chem 284:21777–21781
Bao Q, Shi Y (2007) Apoptosome: a platform for the activation of initiator caspases. Cell Death Differ 14:56–65
Boatright KM, Renatus M, Scott FL, Sperandio S, Shin H et al (2003) A unified model for apical caspase activation. Mol Cell 11:529–541
Li H, Zhu H, Xu CJ, Yuan J (1998) Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94:491–501
Bertrand MJ, Milutinovic S, Dickson KM, Ho WC, Boudreault A et al (2008) cIAP1 and cIAP2 facilitate cancer cell survival by functioning as E3 ligases that promote RIP1 ubiquitination. Mol Cell 30:689–700
Ea CK, Deng L, Xia ZP, Pineda G, Chen ZJ (2006) Activation of IKK by TNFalpha requires site-specific ubiquitination of RIP1 and polyubiquitin binding by NEMO. Mol Cell 22:245–257
Hitomi J, Christofferson DE, Ng A, Yao J, Degterev A et al (2008) Identification of a molecular signaling network that regulates a cellular necrotic cell death pathway. Cell 135:1311–1323
Wang L, Du F, Wang X (2008) TNF-alpha induces two distinct caspase-8 activation pathways. Cell 133:693–703
Lin Y, Devin A, Rodriguez Y, Liu ZG (1999) Cleavage of the death domain kinase RIP by caspase-8 prompts TNF-induced apoptosis. Genes Dev 13:2514–2526
Holler N, Zaru R, Micheau O, Thome M, Attinger A et al (2000) Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat Immunol 1:489–495
Vercammen D, Beyaert R, Denecker G, Goossens V, Van Loo G et al (1998) Inhibition of caspases increases the sensitivity of L929 cells to necrosis mediated by tumor necrosis factor. J Exp Med 187:1477–1485
Konstantinidis K, Whelan RS, Kitsis RN (2012) Mechanisms of cell death in heart disease. Arterioscler Thromb Vasc Biol 32:1552–1562
Sun L, Wang H, Wang Z, He S, Chen S et al (2012) Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell 148:213–227
Wilkins JM, McConnell C, Tipton PA, Hannink M (2014) A conserved motif mediates both multimer formation and allosteric activation of phosphoglycerate mutase 5. J Biol Chem 289:25137–25148
Perrone GG, Tan SX, Dawes IW (2008) Reactive oxygen species and yeast apoptosis. Biochim Biophys Acta 1783:1354–1368
Vandenabeele P, Melino G (2012) The flick of a switch: which death program to choose? Cell Death Differ 19:1093–1095
Yang Y, Fang S, Jensen JP, Weissman AM, Ashwell JD (2000) Ubiquitin protein ligase activity of IAPs and their degradation in proteasomes in response to apoptotic stimuli. Science 288:874–877
Suzuki Y, Nakabayashi Y, Takahashi R (2001) Ubiquitin-protein ligase activity of X-linked inhibitor of apoptosis protein promotes proteasomal degradation of caspase-3 and enhances its anti-apoptotic effect in Fas-induced cell death. Proc Natl Acad Sci USA 98:8662–8667
Ngo JK, Pomatto LC, Davies KJ (2013) Upregulation of the mitochondrial Lon Protease allows adaptation to acute oxidative stress but dysregulation is associated with chronic stress, disease, and aging. Redox Biol 1:258–264
Tatsuta T, Langer T (2009) AAA proteases in mitochondria: diverse functions of membrane-bound proteolytic machines. Res Microbiol 160:711–717
Guerra S, Leri A, Wang X, Finato N, Di Loreto C et al (1999) Myocyte death in the failing human heart is gender dependent. Circ Res 85:856–866
Saraste A, Pulkki K, Kallajoki M, Heikkila P, Laine P et al (1999) Cardiomyocyte apoptosis and progression of heart failure to transplantation. Eur J Clin Invest 29:380–386
Hayakawa Y, Chandra M, Miao W, Shirani J, Brown JH et al (2003) Inhibition of cardiac myocyte apoptosis improves cardiac function and abolishes mortality in the peripartum cardiomyopathy of Galpha(q) transgenic mice. Circulation 108:3036–3041
Diwan A, Krenz M, Syed FM, Wansapura J, Ren X et al (2007) Inhibition of ischemic cardiomyocyte apoptosis through targeted ablation of Bnip3 restrains postinfarction remodeling in mice. J Clin Investig 117:2825–2833
Honda O, Kuroda M, Joja I, Asaumi J, Takeda Y et al (2000) Assessment of secondary necrosis of Jurkat cells using a new microscopic system and double staining method with annexin V and propidium iodide. Int J Oncol 16:283–288
Malhi H, Gores GJ, Lemasters JJ (2006) Apoptosis and necrosis in the liver: a tale of two deaths? Hepatology 43:S31–S44
Kim JS, He L, Lemasters JJ (2003) Mitochondrial permeability transition: a common pathway to necrosis and apoptosis. Biochemical and biophysical research communications 304:463–470
Lemasters JJ, Nieminen AL, Qian T, Trost LC, Elmore SP et al (1998) The mitochondrial permeability transition in cell death: a common mechanism in necrosis, apoptosis and autophagy. Biochim Biophys Acta 1366:177–196
Zamzami N, Hirsch T, Dallaporta B, Petit PX, Kroemer G (1997) Mitochondrial implication in accidental and programmed cell death: apoptosis and necrosis. J Bioenerg Biomembr 29:185–193
Nakayama H, Chen X, Baines CP, Klevitsky R, Zhang X et al (2007) Ca2 + - and mitochondrial-dependent cardiomyocyte necrosis as a primary mediator of heart failure. J Clin Investig 117:2431–2444
Ucar A, Gupta SK, Fiedler J, Erikci E, Kardasinski M et al (2012) The miRNA-212/132 family regulates both cardiac hypertrophy and cardiomyocyte autophagy. Nature communications 3:1078
Kostin S, Pool L, Elsasser A, Hein S, Drexler HC et al (2003) Myocytes die by multiple mechanisms in failing human hearts. Circ Res 92:715–724
Nishida K, Otsu K (2008) Cell death in heart failure. Circ J 72(Suppl A):A17–A21
Nakai A, Yamaguchi O, Takeda T, Higuchi Y, Hikoso S et al (2007) The role of autophagy in cardiomyocytes in the basal state and in response to hemodynamic stress. Nat Med 13:619–624
Cuervo AM, Dice JF (2000) Age-related decline in chaperone-mediated autophagy. J Biol Chem 275:31505–31513
Cuervo AM (2004) Autophagy: many paths to the same end. Mol Cell Biochem 263:55–72
Levine B, Klionsky DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6:463–477
Ohsumi Y, Mizushima N (2004) Two ubiquitin-like conjugation systems essential for autophagy. Semin Cell Dev Biol 15:231–236
Lemasters JJ (2005) Selective mitochondrial autophagy, or mitophagy, as a targeted defense against oxidative stress, mitochondrial dysfunction, and aging. Rejuvenation research 8:3–5
Zhu H, Tannous P, Johnstone JL, Kong Y, Shelton JM et al (2007) Cardiac autophagy is a maladaptive response to hemodynamic stress. J Clin Investig 117:1782–1793
Takemura G, Miyata S, Kawase Y, Okada H, Maruyama R et al (2006) Autophagic degeneration and death of cardiomyocytes in heart failure. Autophagy 2:212–214
Terman A, Brunk UT (1998) On the degradability and exocytosis of ceroid/lipofuscin in cultured rat cardiac myocytes. Mech Ageing Dev 100:145–156
Grune T, Merker K, Jung T, Sitte N, Davies KJ (2005) Protein oxidation and degradation during postmitotic senescence. Free Radic Biol Med 39:1208–1215
Rooyackers OE, Adey DB, Ades PA, Nair KS (1996) Effect of age on in vivo rates of mitochondrial protein synthesis in human skeletal muscle. Proc Natl Acad Sci USA 93:15364–15369
Brunk UT, Terman A (2002) The mitochondrial-lysosomal axis theory of aging: accumulation of damaged mitochondria as a result of imperfect autophagocytosis. European journal of biochemistry/FEBS 269:1996–2002
Terman A, Brunk UT (2005) Autophagy in cardiac myocyte homeostasis, aging, and pathology. Cardiovasc Res 68:355–365
Kurz T, Eaton JW, Brunk UT (2011) The role of lysosomes in iron metabolism and recycling. The international journal of biochemistry & cell biology 43:1686–1697
Brunk UT, Neuzil J, Eaton JW (2001) Lysosomal involvement in apoptosis. Redox report: communications in free radical research 6:91–97
Terman A, Gustafsson B, Brunk UT (2006) The lysosomal-mitochondrial axis theory of postmitotic aging and cell death. Chem Biol Interact 163:29–37
Yan L, Sadoshima J, Vatner DE, Vatner SF (2006) Autophagy: a novel protective mechanism in chronic ischemia. Cell Cycle 5:1175–1177
Kunapuli S, Rosanio S, Schwarz ER (2006) “How do cardiomyocytes die?” apoptosis and autophagic cell death in cardiac myocytes. J Cardiac Fail 12:381–391
Iglewski M, Hill JA, Lavandero S, Rothermel BA (2010) Mitochondrial fission and autophagy in the normal and diseased heart. Curr Hypertens Rep 12:418–425
Taneike M, Yamaguchi O, Nakai A, Hikoso S, Takeda T et al (2010) Inhibition of autophagy in the heart induces age-related cardiomyopathy. Autophagy 6:600–606
Moe GW, Naik G, Konig A, Lu X, Feng Q (2002) Early and persistent activation of myocardial apoptosis, bax and caspases: insights into mechanisms of progression of heart failure. Pathophysiology 8:183–192
Hamacher-Brady A, Brady NR, Gottlieb RA, Gustafsson AB (2006) Autophagy as a protective response to Bnip3-mediated apoptotic signaling in the heart. Autophagy 2:307–309
Matsui Y, Takagi H, Qu X, Abdellatif M, Sakoda H et al (2007) Distinct roles of autophagy in the heart during ischemia and reperfusion: roles of AMP-activated protein kinase and Beclin 1 in mediating autophagy. Circ Res 100:914–922
Gottlieb RA, Gustafsson AB (2011) Mitochondrial turnover in the heart. Biochim Biophys Acta 1813:1295–1301
Kuzmicic J, Del Campo A, Lopez-Crisosto C, Morales PE, Pennanen C et al (2011) Mitochondrial dynamics: a potential new therapeutic target for heart failure. Rev Esp Cardiol 64:916–923
Liu Y, Levine B (2015) Autosis and autophagic cell death: the dark side of autophagy. Cell Death Differ 22:367–376
Ng AC (2010) Integrative systems biology and networks in autophagy. Seminars in immunopathology 32:355–361
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moe, G.W., Marín-García, J. Role of cell death in the progression of heart failure. Heart Fail Rev 21, 157–167 (2016). https://doi.org/10.1007/s10741-016-9532-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-016-9532-0