Abstract
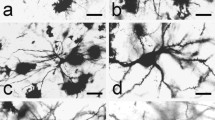
The claustrum is a subcortical nucleus found in the telencephalon of all placental mammals. It is a symmetrical, thin and irregular sheet of grey matter which lies between the inner surface of the insular cortex and the outer surface of the putamen. The claustrum has extensive connections with the visual, auditory, somatosensory and motor regions of the cortex, as well as with subcortical and allocortical regions. The aim of this study was to provide a detailed description of the morphology of different types of Golgi-impregnated and gold-toned neurons and fibers in the dorsal claustrum of the cat employing the combined Golgi-electron microscope Fairén method. We were able to distinguish two major types of neurons: those with dendritic spines (spiny) and those without dendritic spines (aspiny). In both groups we observed large (21–40 µm in diameter), medium-sized (16–21 µm in diameter) and small cells (10–16 µm in diameter), describing their ultrastructural organization and characteristic features, including the presence of terminal boutons. These ultrastructural findings allow us to conclude that large and medium-sized spiny claustral neurons are indeed efferent neurons, projecting to the cortex, while the small spiny and the different types of aspiny neurons are most likely inhibitory local circuit interneurons. The findings in the present study will hopefully contribute to a better understanding of the role of the claustrum.




Similar content being viewed by others
References
Agster KL, Tomás Pereira I, Saddoris MP, Burwell RD (2016) Subcortical connections of the perirhinal, postrhinal, and entorhinal cortices of the rat. II. efferents. Hippocampus 26(9):1213–1230. https://doi.org/10.1002/hipo.22600
Amaral DG, Cowan WM (1980) Subcortical afferents to the hippocampal formation in the monkey. J Comp Neurol 189(4):573–591. https://doi.org/10.1002/cne.901890402
Arikuni T, Kubota K (1985) Claustral and amygdaloid afferents to the head of the caudate nucleus in macaque monkeys. Neurosci Res 2(4):239–254. https://doi.org/10.1016/0168-0102(85)90003-3
Atlan G, Terem A, Peretz-Rivlin N, Groysman M, Citri A (2017) Mapping synaptic cortico-claustral connectivity in the mouse. J Comp Neurol 525(6):1381–1402. https://doi.org/10.1002/cne.23997
Baizer JS, Sherwood CC, Noonan M, Hof PR (2014) Comparative organization of the claustrum: what does structure tell us about function? Front Syst Neurosci 8:117. https://doi.org/10.3389/fnsys.2014.00117
Baugh LA, Lawrence JM, Marotta JJ (2011) Novel claustrum activation observed during a visuomotor adaptation task using a viewing window paradigm. Behav Brain Res 223(2):395–402. https://doi.org/10.1016/j.bbr.2011.05.009
Braak H, Braak E (1982) Neuronal types in the claustrum of man. Anat Embryol (Berlin) 163(4):447–460. https://doi.org/10.1007/BF00305558
Brand S (1981) A serial section Golgi analysis of the primate claustrum. Anat Embryol (Berlin) 162(4):475–488. https://doi.org/10.1007/BF00301872
Brown SP, Mathur BN, Olsen SR, Luppi PH, Bickford ME, Citri A (2017) New breakthroughs in understanding the role of functional interactions between the neocortex and the claustrum. J Neurosci 37(45):10877–10881. https://doi.org/10.1523/JNEUROSCI.1837-17.2017
Buchanan KJ, Johnson JI (2011) Diversity of spatial relationships of the claustrum and insula in branches of the mammalian radiation. Ann N Y Acad Sci 1225(S1):E30–E63. https://doi.org/10.1111/j.1749-6632.2011.06022.x
Chau A, Salazar AM, Krueger F, Cristofori I, Grafman J (2015) The effect of claustrum lesions on human consciousness and recovery of function. Conscious Cogn 36:256–264. https://doi.org/10.1016/j.concog.2015.06.017
Clascá F, Avendaño C, Román-Guindo A, Llamas A, Reinoso-Suárez F (1992) Innervation from the claustrum of the frontal association and motor areas: axonal transport studies in the cat. J Comp Neurol 326(3):402–422. https://doi.org/10.1002/cne.903260307
Cozzi B, Roncon G, Granato A, Giurisato M, Castagna M, Peruffo A, Panin M, Ballarin C, Montelli S, Pirone A (2014) The claustrum of the bottlenose dolphin Tursiops truncatus (Montagu 1821). Front Syst Neurosci 8:42. https://doi.org/10.3389/fnsys.2014.00042
Crick FC, Koch C (2005) What is the function of the claustrum? Philos Trans R Soc Lond B 360(1458):1271–1279. https://doi.org/10.1098/rstb.2005.1661
Day-Brown JD, Slusarczyk AS, Zhou N, Quiggins R, Petry HM, Bickford ME (2017) Synaptic organization of striate cortex projections in the tree shrew: a comparison of the claustrum and dorsal thalamus. J Comp Neurol 525(6):1403–1420. https://doi.org/10.1002/cne.23998
Deutch AY, Mathur BN (2015) Editorial: the claustrum: charting a way forward for the brain’s most mysterious nucleus. Front Syst Neurosci 15(9):103. https://doi.org/10.3389/fnsys.2015.00103
Dinopoulos A, Papadopoulos GC, Michaloudi H, Parnavelas JG, Uylings HB, Karamanlidis AN (1992) Claustrum in the hedgehog (Erinaceus europaeus) brain: cytoarchitecture and connections with cortical and subcortical structures. J Comp Neurol 316(2):187–205. https://doi.org/10.1002/cne.903160205
Druga R (1966) The claustrum of the cat (Felis domestica). Folia Morphol (Praha) 14:7–16
Edelstein LR, Denaro FJ (2004) The claustrum: a historical review of its anatomy, physiology, cytochemistry and functional significance. Cell Mol Biol 50:675–702. https://doi.org/10.1170/T558
Fairén A, Peters A, Saldanha J (1977) A new procedure for examining Golgi impregnated neurons by light and electron microscopy. J Neurocytol 6(3):311–337. https://doi.org/10.1007/BF01175194
Goll Y, Atlan G, Citri A (2015) Attention: the claustrum. Trends Neurosci 38(8):486–495. https://doi.org/10.1016/j.tins.2015.05.006
Hinova-Palova DV, Dimova R, Ivanov DP (1987) Identification of small neurons (dwarf cells) in the claustrum of the cat. Light and electron microscopic observations. Verh Anat Ges Leipzig 82(Anat Anz Suppl 164):83
Hinova-Palova DV, Paloff A, Christova T, Ovtscharoff W (1997) Topographical distribution of NADPH-diaphorase-positive neurons in the cat’s claustrum. Eur J Morphol 35(2):105–116
Hinova-Palova DV, Edelstein LR, Paloff AM, Hristov S, Papantchev VG, Ovtscharoff WA (2007) Parvalbumin in the cat claustrum: ultrastructure, distribution and functional implications. Acta Histochem 109(1):61–77. https://doi.org/10.1016/j.acthis.2006.09.006
Hinova-Palova D, Edelstein L, Paloff A, Hristov S, Papantchev V, Ovtscharoff W (2008) Neuronal nitric oxide synthase immunopositive neurons in cat claustrum—a light and electron microscopic study. J Mol Histol 39(4):447–457. https://doi.org/10.1007/s10735-008-9184-z
Hinova-Palova D, Edelstein L, Papantchev V, Landzhov B, Malinova L, Todorova-Papantcheva D, Minkov M, Paloff A, Ovtscharoff W (2012) Light and electron-microscopic study of leucine enkephalin immunoreactivity in the cat claustrum. J Mol Histol 43(6):641–649. https://doi.org/10.1007/s10735-012-9448-5
Hinova-Palova DV, Edelstein L, Landzhov B, Minkov M, Malinova L, Hristov S, Denaro FJ, Alexandrov A, Kiriakova T, Brainova I, Paloff A, Ovtscharoff W (2014a) Topographical distribution and morphology of NADPH-diaphorase-stained neurons in the human claustrum. Front Syst Neurosci 8:96. https://doi.org/10.3389/fnsys.2014.00096
Hinova-Palova DV, Edelstein L, Landzhov BV, Braak E, Malinova LG, Minkov M, Paloff A, Ovtscharoff W (2014b) Parvalbumin-immunoreactive neurons in the human claustrum. Brain Struct Funct 219(5):1813–1830. https://doi.org/10.1007/s00429-013-0603-x
Hinova-Palova DV, Landzhov B, Dzhambazova E, Minkov M, Edelstein L, Malinova L, Paloff A, Ovtscharoff W (2014c) Neuropeptide Y immunoreactivity in the cat claustrum: a light- and electron-microscopic investigation. J Chem Neuroanat 61–62:107–119. https://doi.org/10.1016/j.jchemneu.2014.08.007
Johnson JI, Fenske BA, Jaswa AS, Morris JA (2014) Exploitation of puddles for breakthroughs in claustrum research. Front Syst Neurosci 8:78. https://doi.org/10.3389/fnsys.2014.00078
Kavounoudias A, Roll JP, Anton JL, Nazarian B, Roth M, Roll R (2008) Proprio-tactile integration for kinesthetic perception: an fMRI study. Neuropsychologia 46(2):567–575. https://doi.org/10.1016/j.neuropsychologia.2007.10.002
Kim J, Matney CJ, Roth RH, Brown SP (2016) Synaptic organization of the neuronal circuits of the claustrum. J Neurosci 36(3):773–784. https://doi.org/10.1523/JNEUROSCI.364315.2016
Kowiański P, Dziewiatkowski J, Kowiańska J, Moryś J (1999) Comparative anatomy of the claustrum in selected species: a morphometric analysis. Brain Behav Evol 53(1):44–54. https://doi.org/10.1159/000006581
Landzhov B, Hinova-Palova D, Edelstein L, Dzhambazova E, Brainova I, Georgiev GP, Ivanova V, Paloff A, Ovtscharoff W (2017) Comparative investigation of neuronal nitric oxide synthase immunoreactivity in rat and human claustrum. J Chem Neuroanat 86:1–14. https://doi.org/10.1016/j.jchemneu.2017.07.003
LeVay S, Sherk H (1981) The visual claustrum of the cat. I. Structure and connections. J Neurosci 1(9):956–980. https://doi.org/10.1523/JNEUROSCI.01-09-00956.1981
Mamos L, Narkiewicz O, Moryś J (1986) Neurons of the claustrum in the cat: a Golgi study. Acta Neurobiol Exp (Wars) 46(4):171–178
Markowitsch HJ, Irle E, Bang-Olsen R, Flindt-Egebak P (1984) Claustral efferents to the cat’s limbic cortex studied with retrograde and anterograde tracing techniques. Neuroscience 12(2):409–425. https://doi.org/10.1016/0306-4522(84)90062-9
Mathur BN (2014) The claustrum in review. Fron Syst Neurosci 8:48. https://doi.org/10.3389/fnsys.2014.00048
Mathur BN, Reser D, Smith JB (2017) Conference Proceedings: 3rd Annual Society for Claustrum Research Meeting. Claustrum 2:1. https://doi.org/10.1080/20023294.2017.1349859
Moryś J, Berdel B, Maciejewska B, Sadowski M, Sidorowicz M, Kowiañska J, Narkiewicz O (1996) Division of the human claustrum according to its architectonics and morphometric parameters. Folia Morphol (Warsz) 55(2):69–82
Narkiewicz O (1964) Degeneration in the claustrum after regional neocortical ablations in the cat. J Comp Neurol 123:335–336. https://doi.org/10.1002/cne.901230304
Norita M, Hirata Y (1976) Some electron microscope findings of the claustrum of the cat. Arch Histol Japonicum 39(1):33–49
Olson CR, Graybiel AM (1980) Sensory maps in the claustrum of the cat. Nature 288(5790):479–481. https://doi.org/10.1038/288479a0
Pearson JC, Haines DE (1980) Somatosensory thalamus of a prosimian primate (Galago senegalensis). II. An HRP and Golgi study of the ventral posterolateral nucleus (VPL). J Comp Neurol 190(3):559–580. https://doi.org/10.1002/cne.901900310
Peters A, Fairén A (1978) Smooth and sparsely-spined stellate cells in the visual cortex of the rat: a study using a combined Golgi-electron microscopic technique. J Comp Neurol 181(1):129–171. https://doi.org/10.1002/cne.901810108
Pirone A, Cozzi B, Edelstein L, Peruffo A, Lenzi C, Quilici F, Antonini R, Castagna M (2012) Topography of Gng2- and NetrinG2-expression suggests an insular origin of the human claustrum. PLoS ONE 7(9):e44745. https://doi.org/10.1371/journal.pone.0044745
Pirone A, Castagna M, Granato A, Peruffo A, Quilici F, Cavicchioli L, Piano I, Lenzi C, Cozzi B (2014) Expression of calcium-binding proteins and selected neuropeptides in the human, chimpanzee, and crab-eating macaque claustrum. Front Syst Neurosci 8:99. https://doi.org/10.3389/fnsys.2014.00099
Pirone A, Magliaro C, Giannessi E, Ahluwalia A (2015) Parvalbumin expression in the claustrum of the adult dog. An immunohistochemical and topographical study with comparative notes on the structure of the nucleus. J Chem Neuroanat 64–65:33–42. https://doi.org/10.1016/j.jchemneu.2015.02.004
Pirone A, Cantile C, Miragliotta V, Lenzi C, Giannessi E, Cozzi B (2016) Immunohistochemical distribution of the cannabinoid receptor 1 and fatty acid amide hydrolase in the dog claustrum. J Chem Neuroanat 74:21–27. https://doi.org/10.1016/j.jchemneu.2016.02.002
Pirone A, Miragliotta V, Ciregia F, Giannessi E, Cozzi B (2018) The catecholaminergic innervation of the claustrum of the pig. J Anat 232(1):158–166. https://doi.org/10.1111/joa.12706
Reser DH, Majka P, Snell S, Chan JM, Watkins K, Worthy K, Quiroga MD, Rosa MG (2017) Topography of claustrum and insula projections to medial prefrontal and anterior cingulate cortices of the common marmoset (Callithrix jacchus). J Comp Neurol 525(6):1421–1441. https://doi.org/10.1002/cne.24009
Smith JB, Alloway KD (2010) Functional specificity of claustrum connections in the rat: interhemispheric communication between specific parts of motor cortex. J Neurosci 30(50):16832–16844. https://doi.org/10.1523/JNEUROSCI.4438-10.2010
Smith JB, Alloway KD (2014) Interhemispheric claustral circuits coordinate sensory and motor cortical areas that regulate exploratory behaviors. Front Syst Neurosci 8:93. https://doi.org/10.3389/fnsys.2014.00093
Smythies J, Edelstein L, Ramachandran V (eds) (2014) The claustrum. Structural, functional and clinical neuroscience. Academic Press, Cambridge
Spahn B, Braak H (1985) Percentage of projection neurons and various types of interneurons in the human claustrum. Acta Anat (Basel) 122(4):245–248. https://doi.org/10.1159/000146023
Stedehouder J, Kushner SA (2017) Myelination of parvalbumin interneurons: a parsimonious locus of pathophysiological convergence in schizophrenia. Mol Psychiatry 22(1):4–12. https://doi.org/10.1038/mp.2016.147
Tomás Pereira I, Agster KL, Burwell RD (2016) Subcortical connections of the perirhinal, postrhinal, and entorhinal cortices of the rat. I. afferents. Hippocampus 26(9):1189–1212. https://doi.org/10.1002/hipo.22603
Torgerson CM, Irimia A, Goh SY, Van Horn JD (2015) The DTI connectivity of the human claustrum. Hum Brain Mapp 36(3):827–838. https://doi.org/10.1002/hbm.22667
Valverde F (1970) The Golgi method. A tool for comparative structural analysis. In: Nauta WJH, Ebbesson SOE (eds) Contemporary research methods in neuroanatomy. Springer, Berlin, pp 12–31
Vertes RP, Hoover WB (2008) Projections of the paraventricular and paratenial nuclei of the dorsal midline thalamus in the rat. J Comp Neurol 508(2):212–237. https://doi.org/10.1002/cne.21679
Wang Q, Ng L, Harris JA, Feng D, Li Y, Royall JJ, Oh SW, Bernard A, Sunkin SM, Koch C, Zeng H (2017) Organization of the connections between claustrum and cortex in the mouse. J Comp Neurol 525(6):1317–1346. https://doi.org/10.1002/cne.24047
Watson GDR, Smith JB, Alloway KD (2017) Interhemispheric connections between the infralimbic and entorhinal cortices: the endopiriform nucleus has limbic connections that parallel the sensory and motor connections of the claustrum. J Comp Neurol 525(6):1363–1380. https://doi.org/10.1002/cne.23981
White MG, Cody PA, Bubser M, Wang HD, Deutch AY, Mathur BN (2017) Cortical hierarchy governs rat claustrocortical circuit organization. J Comp Neurol 525(6):1347–1362. https://doi.org/10.1002/cne.23970
Zingg B, Hintiryan H, Gou L, Song MY, Bay M, Bienkowski MS, Foster NN, Yamashita S, Bowman I, Toga AW, Dong HW (2014) Neural networks of the mouse neocortex. Cell 156(5):1096–1111. https://doi.org/10.1016/j.cell.2014.02.023
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hinova-Palova, D., Iliev, A., Edelstein, L. et al. Electron microscopic study of Golgi-impregnated and gold-toned neurons and fibers in the claustrum of the cat. J Mol Hist 49, 615–630 (2018). https://doi.org/10.1007/s10735-018-9799-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-018-9799-7