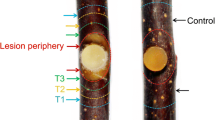
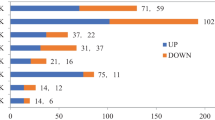
Abstracts
Downy mildew caused by Hyaloperonospora brassicae is one of the most damaging diseases of Chinese cabbage. In this study, we analyzed genome-wide gene expression profiles of a resistant line 318R and a susceptible line 361S of Chinese cabbage in response to downy mildew infection by high throughput RNA sequencing technology. More transcripts were identified in H. brassicae infected samples in both lines, especially in the susceptible line 361S. A total of 2297 and 3338 differentially expressed genes were identified in 318R and 361S, respectively, which are mainly involved in the plant-pathogen interaction pathway, plant hormone signal transduction pathway, and biosynthesis of secondary metabolites pathway. A total of 1284 genes were found to be differentially expressed in both lines, among which, 68 genes showed higher variation in the resistant line than in the susceptible line. These 68 genes, along with the 1012 genes that were only differentially expressed in the resistant line, were annotated to defense response genes, transcriptional factors, and cell wall related genes, which function in the plant-pathogen interaction pathway, protein processing in endoplasmic reticulum pathway, or metabolism pathways. These genes are important candidates for understanding the molecular mechanisms of host response to downy mildew.



Similar content being viewed by others
References
Adie, B. A., Perez-Perez, J., Perez-Perez, M. M., Godoy, M., Sanchez-Serrano, J. J., Schmelz, E. A., et al. (2007). ABA is an essential signal for plant resistance to pathogens affecting JA biosynthesis and the activation of defenses in Arabidopsis. Plant Cell, 19(5), 1665–1681.
Agarwal, P. K., Agarwal, P., Reddy, M. K., & Sopory, S. K. (2006). Role of DREB transcription factors in abiotic and biotic stress tolerance in plants. Plant Cell Reports, 25(12), 1263–1274.
Asai, S., Rallapalli, G., Piquerez, S. J. M., Caillaud, M., Furzer, O.J., Ishaque, N., et al. (2014). Expression profiling during Arabidopsis/downy mildew interaction reveals a highlyexpressed effector that attenuates responses to salicylic acid. PLoS Pathogens, 10(10), e1004443.
Audic, S., & Claverie, J. M. (1997). The significance of digital gene expression profiles. Genome Research, 7(10), 986–995.
Brutus, A., Sicilia, F., Macone, A., Cervone, F., & De Lorenzo, G. (2010). A domain swap approach reveals a role of the plant wall-associated kinase 1 (WAK1) as a receptor of oligogalacturonides. Proceedings of the National Academy of Sciences of the United States of America, 107(20), 9452–9457.
Burkhardt, A., & Day, B. (2016). Transcriptome and small RNAome Dynamicsduring a resistant and susceptible Interactionbetween cucumber and downy mildew. Plant Genome, 9(1), 1–19.
Caillaud, M., Asai, S., Rallapalli, G., Piquerez, S., Fabro, G., et al. (2013). A downy mildew effector attenuates salicylic acid–triggered immunity in Arabidopsisby interacting with the HostMediatorComplex. PLoS Biology, 11(12), e1001732.
Cantu, D., Vicente, A. R., Labavitch, J. M., Bennett, A. B., & Powell, A. L. (2008). Strangers in the matrix: Plant cell walls and pathogen susceptibility. Trends in Plant Science, 13(11), 610–617.
Chen, X. F., Hou, X. L., Zhang, J. Y., & Zheng, J. Q. (2008). Molecular characterization of two important antifungal proteins isolated by downy mildew infection in non-heading Chinese cabbage. Molecular Biology Reports, 35(4), 621–629.
Chen, X. F., Ma, Q. H., Mu, J. H., & Wang, B. C. (2015). PRs genes expression in Chinese cabbage inductionby Peronosporaparasitica. Jilin agricultural sciences, 40(4), 60–64 (In Chinese).
Coker, T. L., Cevik, V., Beynon, J. L., & Gifford, M. L. (2015). Spatial dissection of the Arabidopsis Thaliana transcriptional response to downy mildewusing fluorescence activated cell sorting. Frontiers in Plant Science, 6, 527.
Damme, M., Andel, A., Huibers, R. P., Panstruga, R., Weisbeek, P. J., & Ackerveken, G. V. (2005). Identification of Arabidopsis loci required for susceptibility to the downy mildew pathogen. Hyaloperonosporaparasitica. Molecular Plant-Microbe Interactions, 18(6), 583–592.
Di, C., Zhang, M., Xu, S., Cheng, T., & An, L. (2006). Role of poly-galacturonase inhibiting protein in plant defense. Critical Reviews in Microbiology, 32(2), 91–100.
Dong, X., Hong, Z., Chatterjee, J., Kim, S., & Verma, D. P. (2008). Expression of callose synthase genes and its connection with Npr1 signaling pathway during pathogen infection. Planta, 229(1), 87–98.
Ellinger, D., Glöckner, A., Koch, J., Naumann, M., Stürtz, V., Schütt, K., et al. (2014). Interaction of the Arabidopsis GTPase RabA4c with its effector PMR4 results in complete penetration resistance to powdery mildew. Plant Cell, 26(7), 3185–3200.
Eulgem, T., & Somssich, I. E. (2007). Networks of WRKY transcription factors in defense signaling. Current Opinion in Plant Biology, 10(4), 366–371.
Fan, M., Bai, M. Y., Kim, J. G., Wang, T., Oh, E., Chen, L., et al. (2014). The bHLH transcription factor HBI1 mediates the trade-off between growth and pathogen-associated molecular pattern-triggered immunity in Arabidopsis. Plant Cell, 26(2), 828–841.
Farinhó, M., Coelho, P., Carlier, J., Svetleva, D., Monteiro, A., & Leitao, J. (2004). Mapping of a locus for adult plant resistance to downy mildew in broccoli (Brassica Oleraceaconvar. Italica). Theoretical and Applied Genetics, 109(7), 1392–1398.
Gao, T., Yu, S., Zhang, F., Chen, X., Yu, Y., Zhang, D., et al. (2014). Expression analysis of major genes involved in signaling pathways during infection of Chinese cabbage with Hyaloperonosporabrassicae. ScientiaHorticulturae, 167(3), 27–35.
Giovannelli, J. L., Farnham, M. W., Wang, M., & Strand, A. E. (2002). Development of sequence characterized amplified region markers linked to downy mildew resistance in broccoli. Journal of the American Society for Horticulturalence, 127(4), 597–601.
Glazebrook, J. (2005). Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annual Review of Phytopathology, 43, 205–227.
Greenberg, J. T., & Yao, N. (2004). The role and regulation of programmed cell death in plant-pathogen interactions. Cellular Microbiology, 6(3), 201–211.
Huckelhoven, R. (2007). Cell wall-associated mechanisms of disease resistance and susceptibility. Annual Review of Phytopathology, 45, 101–127.
Ishiga, Y., Ishiga, T., Uppalapati, S. R., & Mysore, K. S. (2013). Jasmonate ZIM-domain (JAZ) protein regulates host and nonhost pathogen-induced cell death in tomato and Nicotiana Benthamiana. PLoS One, 8(9), e75728.
Jung, H. W., Kim, K. D., & Hwang, B. K. (2005). Identification of pathogen-responsive regions in the promoter of a pepper lipid transfer protein gene (CALTPI) and the enhanced resistance of the CALTPI transgenic Arabidopsis against pathogen and environmental stresses. Planta, 221(3), 361–373.
Kim, S., Song, Y. H., Lee, J. Y., Choi, S. R., Dhandapani, V., Jang, C. S., et al. (2011). Identification of the BrRHP1 locus that confers resistance to downy mildew in Chinese cabbage (Brassica Rapa Ssp. Pekinensis) and development of linked molecular markers. Theoretical and Applied Genetics, 123(7), 1183–1192.
Li, J., Brader, G., & Palva, E. T. (2004). The WRKY70 transcription factor: A node of convergence for jasmonate-mediated and salicylate-mediated signals in plant defense. Plant Cell, 16(2), 319–331.
Li, H., Zhou, S. Y., Zhao, W. S., Su, S. C., & Peng, Y. L. (2009a). A novel wall-associated receptor-like protein kinase gene, OsWAK1, plays important roles in rice blast disease resistance. Plant Molecular Biology, 69(3), 337–346.
Li, R., Yu, C., Li, Y., Lam, T. W., Yiu, S. M., Kristiansen, K., et al. (2009b). SOAP2: An improved ultrafast tool for short read alignment. Bioinformatics, 25(15), 1966–1967.
Li, H., Yu, S. C., Zhang, F. L., Yu, Y. J., Zhao, X. Y., Zhang, D. S., et al. (2011). Development of molecular markers linked to the resistant QTL for downy mildew in Brassica Rapa L. Ssp. Pekinensis. Hereditas, 33(11), 1271–1278.
Li, X., Wu, J., Yin, L., Zhang, Y. L., Qu, J. J., & Lu, J. (2015). Comparative transcriptome analysis reveals defense-related genes and pathways against downy mildew in Vitisamurensis grapevine. Plant Physiology and Biochemistry, 95, 1.
Liu, G., Holub, E. B., Alonso, J. M., Ecker, J. R., & Fobert, P. R. (2005). An Arabidopsis NPR1-like gene, NPR4, is required for disease resistance. Plant Journal, 41(2), 304–318.
Loake, G., & Grant, M. (2007). Salicylic acid in plant defence--the players and protagonists. Current Opinion in Plant Biology, 10(5), 466–472.
Lorenzo, O., Piqueras, R., Sanchez-Serrano, J. J., & Solano, R. (2003). ETHYLENE RESPONSE FACTOR1 integrates signals from ethylene and jasmonate pathways in plant defense. Plant Cell, 15(1), 165–178.
Lorenzo, O., Chico, J. M., Sanchez-Serrano, J. J., & Solano, R. (2004). JASMONATE-INSENSITIVE1 encodes a MYC transcription factor essential to discriminate between different jasmonate-regulated defense responses in Arabidopsis. Plant Cell, 16(7), 1938–1950.
Manners, J. M., Penninckx, I. A., Vermaere, K., Kazan, K., Brown, R. L., Morgan, A., et al. (1998). The promoter of the plant defensin gene PDF1.2 from Arabidopsis is systemically activated by fungal pathogens and responds to methyl jasmonate but not to salicylic acid. Plant Molecular Biology, 38(6), 1071–1080.
Marcos, R., Izquierdo, Y., Vellosillo, T., Kulasekaran, S., Cascon, T., Hamberg, M., et al. (2015). 9-lipoxygenase-derived Oxylipins activate Brassinosteroid signaling to promote Cell Wall-based defense and limit pathogen infection. Plant Physiology, 169(3), 2324–2334.
McDowell, J. M., Cuzick, A., Can, C., Beynon, J., Dang, J. L., & Holub, E. B. (2000). Downy mildew (Peronosporaparasitica) resistance genes in Arabidopsis vary in functional requirements for NDR1, EDS1, NPR1 and salicylic acid accumulation. Plant Journal, 22(6), 523–529.
Mortazavi, A., Williams, B. A., McCue, K., Schaeffer, L., & Wold, B. (2008). Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nature Methods, 5(7), 621–628.
Mou, Z., Fan, W., & Dong, X. (2003). Inducers of plant systemic acquired resistance regulate NPR1 function through redox changes. Cell, 113(7), 935–944.
Oh, S. K., Park, J. M., Joung, Y. H., Lee, S., Chung, E., Kim, S. Y., et al. (2005). A plant EPF-type zinc-finger protein, CaPIF1, involved in defence against pathogens. Molecular Plant Pathology, 6(3), 269–285.
Penninckx, I. A., Thomma, B. P., Buchala, A., Metraux, J. P., & Broekaert, W. F. (1998). Concomitant activation of jasmonate and ethylene response pathways is required for induction of a plant defensin gene in Arabidopsis. Plant Cell, 10(12), 2103–2113.
Penninckx, I. A., Eggermont, K., Schenk, P. M., Van den Ackerveken, G., Cammue, B. P., & Thomma, B. P. (2003). The Arabidopsis mutant iop1 exhibits induced over-expression of the plant defensin gene PDF1.2 and enhanced pathogen resistance. Molecular Plant Pathology, 4(6), 479–486.
Polesani, M., Bortesi, L., Ferrarini, A., Zamboni, A., Fasoli, M., Zadra, C., et al. (2010). General and species-specific transcriptional responses to downy mildew infection in a susceptible (Vitis Vinifera) and a resistant (V. Riparia) grapevine species. BMC Genomics, 11, 117.
Pre, M., Atallah, M., Champion, A., De Vos, M., Pieterse, C. M., & Memelink, J. (2008). The AP2/ERF domain transcription factor ORA59 integrates jasmonic acid and ethylene signals in plant defense. Plant Physiology, 147(3), 1347–1357.
Rimmer, S. R., Shattuck, V. I., & Buchwalt, L. (2007). Compendium of brassica diseases. The American Phytological Society, 28–30.
Rojo, E., Solano, R., & Sánchez-Serrano, J. J. (2003). Interactions between signaling compounds involved in plant defense. Journal of Plant Growth Regulation, 22(1), 82–98.
Sarowar, S., Kim, Y. J., Kim, K. D., Hwang, B. K., Ok, S. H., & Shin, J. S. (2009). Overexpression of lipid transfer protein (LTP) genes enhances resistance to plant pathogens and LTP functions in long-distance systemic signaling in tobacco. Plant Cell Reports, 28(3), 419–427.
Slusarenko, A. J., & Schlaich, N. L. (2003). Downy mildew of Arabidopsis Thaliana caused by Hyaloperonosporaparasitica (formerly Peronosporaparasitica). Molecular Plant Pathology, 4(3), 159–170.
Stassen, J. H. M., Seid, M. F., Vergeer, P. W. J., Nijman, I. J., Snel, B., Cuppen, E., et al. (2012). Effector identification in the lettuce downy mildew Bremiaactucae by massively parallel transcriptome sequencing. Molecular Plant Pathology, 13(7), 719–731.
Sun, C., Wang, L., Hu, D., Riquicho, A. R., Liu, T., Hou, X., et al. (2014). Proteomic analysis of non-heading Chinese cabbage infected with Hyaloperonospora parasitica. Journal of Proteomics, 98(4), 15–30.
Tao, Y., Xie, Z., Chen, W., Glazebrook, J., Chang, H. S., Han, B., et al. (2003). Quantitative nature of Arabidopsis responses during compatible and incompatible interactions with the bacterial pathogen pseudomonas syringae. Plant Cell, 15(2), 317–330.
Underwood, W., & Somerville, S. C. (2008). Focal accumulation of defences at sites of fungal pathogen attack. Journal of Experimental Botany, 59(13), 3501–3508.
van Loon, L. C., Rep, M., & Pieterse, C. M. (2006). Significance of inducible defense-related proteins in infected plants. Annual Review of Phytopathology, 44, 135–162.
Vidhyasekaran, P. (2015). Brassinosteroid signaling in plant immune system. Signaling & Communication in Plants, 2, 403–444.
Vogel, J. P., Raab, T. K., Schiff, C., & Somerville, S. C. (2002). PMR6, a pectate lyase-like gene required for powdery mildew susceptibility in Arabidopsis. Plant Cell, 14(9), 2095–2106.
Vogel, J. P., Raab, T. K., Somerville, C. R., & Somerville, S. C. (2004). Mutations in PMR5 result in powdery mildew resistance and altered cell wall composition. Plant Journal, 40(6), 968–978.
Wang, W., Liu, F., Ren, L., Li, X., Chen, W., Zeng, L. Y., et al. (2016). Resistance SNP development to downy mildew in Brassica Napus using SLAF-seq technique. Chinese journal of oil Corp. Science, 38(5), 555.
Wu, J., Zhang, Y., Zhang, H., Huang, H., Folta, K. M., & Lu, J. (2010). Whole genome wide expression profiles of Vitis Amurensis grape responding to downy mildew by using Solexa sequencing technology. BMC Plant Biology, 10, 234.
Xiao, S., Brown, S., Patrick, E., Brearley, C., & Turner, J. G. (2003). Enhanced transcription of the Arabidopsis disease resistance genes RPW8.1 and RPW8.2 via a salicylic acid-dependent amplification circuit is required for hypersensitive cell death. Plant Cell, 15(1), 33–45.
Xiao, D., Liu, S. T., Wei, Y. P., Zhou, D. Y., Hou, X. L., Li, Y., et al. (2016). cDNA-AFLP analysis reveals differential gene expression in incompatible interaction between infected non-heading Chinese cabbage and Hyaloperonosporaparasitica. Horticulture Research, 3, 16034.
Xu, Y., Chang, P., Liu, D., Narasimhan, M. L., Raghothama, K. G., Hasegawa, P. M., et al. (1994). Plant defense genes are synergistically induced by ethylene and methyl Jasmonate. Plant Cell, 6(8), 1077–1085.
Yu, S., Zhang, F., Yu, R., Zou, Y., Qi, J., & Zhao, X. (2009). Genetic mapping and localization of a major QTL for seedling resistance to downy mildew in Chinese cabbage (Brassica Rapa Ssp. Pekinensis). Molecular Breeding, 23, 573–590.
Yu, S. C., Su, T. B., Zhi, S. H., Zhang, F. L., Wang, W. H., Zhang, D. S., et al. (2016). Construction of a sequence-based bin map and mapping of QTLs for downy mildew resistance at four developmental stages in Chinese cabbage (Brassica Rapa L. Ssp. Pekinensis). Molecular Breeding, 36(4), 1–12.
Zhang, Y., Fan, W., Kinkema, M., Li, X., & Dong, X. (1999). Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene. Proceedings of the National Academy of Sciences PNAS, 96(11), 6523–6528.
Zhang, H., Yang, Y. Z., Wang, C. Y., Liu, M., Li, H., Fu, Y., et al. (2014). Large-scale transcriptome comparison reveals distinct gene activations in wheat responding to stripe rust and powdery mildew. BMC Genomics, 15(1), 898.
Zhi, S. H., Su, T. B., Yu, S. C., Zhang, F. L., Yu, Y. J., Zhang, D. S., et al. (2016). Genetic characteristics of A01-located resistant loci to downy mildew in Chinese cabbage by genome-wide association studies. Plant Physiology Journal, 52(5), 693–702 (In Chinese).
Acknowledgements
This work was supported by the Modern Agricultural Industrial Technology System Funding of Shandong Province, China (SDAIT-05-04); the National Natural Science Foundation of China (31401869); the China Agriculture Research System (CARS-25); the Project for Cultivation of Major Achievements in Science and Technology in SAAS, China (2015CGPY09); the Young Talents Training Program of Shandong Academy of Agricultural Science, China (NKYSCS-03); Taishan Scholar Program of Vegetable Genomics, China (2016-2020); and the Natural Science Foundation of Shandong Province, China (ZR2015YL071); and the Natural Science Foundation of Shandong Province, China (ZR2016CB16).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The paper has not been submitted elsewhere for publication, in whole or in part.
Conflict of interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Human and animal studies
The research did not involve any Human Participants and/or Animals.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Table s1
(XLSX 11 kb)
Table s2
(XLSX 11 kb)
Table s3
(XLSX 593 kb)
Table s4
(XLSX 846 kb)
Table s5
(XLSX 23 kb)
Table s6
(XLSX 13 kb)
Figure s1
Saturation analysis of the four DEGs libraries. A, B, C, and D were saturation analysis of318R–CK, 318R–T, 361S–CK, 361S–T library, respectively. (PDF 171 kb)
Figure s2
Randomness assessments of the four DEGs libraries. A, B, C, and D were randomness assessments of 318R–CK, 318R–T, 361S–CK, 361S–T library, respectively. (PDF 200 kb)
Figure s3
Gene classification based on gene ontology (GO) for DEGs after downy mildew infection. A, Gene classification based on GO for DEGs in 318R; B, Gene classification based on GO for DEGs in 361S. (PDF 664 kb)
Rights and permissions
About this article
Cite this article
Li, J., Ding, Q., Wang, F. et al. Genome-wide gene expression profiles in response to downy mildew in Chinese cabbage (Brassica rapa L. ssp. pekinensis). Eur J Plant Pathol 151, 861–873 (2018). https://doi.org/10.1007/s10658-018-1427-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-018-1427-6