Summary
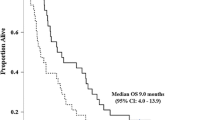
Objective This study aimed to compare the safety and efficacy of the combination therapy of gemcitabine and S-1 (GS) versus gemcitabine and cisplatin (GC) in patients with advanced biliary tract cancer (BTC). Methods In this multicenter retrospective cohort study, a total of 212 patients with advanced BTC receiving GS (n = 125) or GC (n = 87) between July 2006 and August 2015 were analyzed. The primary endpoint was overall survival (OS). The secondary endpoints were progression-free survival (PFS), objective tumor response, and safety. Results Patient characteristics were well balanced between the two groups, except for tumor size (the baseline sum of the largest diameter of the tumor: 6.3 cm in the GS group vs. 8.6 cm in the GC group, p = 0.01). Although the response rate was higher in the GS group than in the GC group (28.8% vs. 10.3%, p = 0.01), the median PFS and OS were comparable between the two groups (PFS of 5.6 vs. 7.6 months, p = 0.74; OS of 12.4 vs. 9.2 months, p = 0.20, respectively). Stomatitis and skin rash were more frequently observed in the GS group, whereas anemia, thrombocytopenia, nausea, and renal toxicity were more commonly observed in the GC group. Conclusion This study demonstrates that GS and GC are similar with regard to their safety and efficacy in patients with advanced BTC. GS could serve as an alternative treatment for advanced BTC as a first-line chemotherapy.



Similar content being viewed by others
Abbreviations
- BTC:
-
Biliary tract cancer
- GS:
-
Gemcitabine and S-1 combination therapy
- GC:
-
Gemcitabine and cisplatin combination therapy
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
References
Miyakawa S, Ishihara S, Horiguchi A et al (2009) Biliary tract cancer treatment: 5,584 results from the biliary tract cancer statistics registry from 1998 to 2004 in Japan. J Hepato-Biliary-Pancreat Surg 16:1–7
Valle J, Wasan H, Palmer DH et al (2010) Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med 362:1273–1281
Okusaka T, Nakachi K, Fukutomi A et al (2010) Gemcitabine alone or in combination with cisplatin in patients with biliary tract cancer: a comparative multicentre study in Japan. Br J Cancer 103:469–474
Furuse J, Okusaka T, Boku N et al (2008) S-1 monotherapy as first-line treatment in patients with advanced biliary tract cancer: a multicenter phase II study. Cancer Chemother Pharmacol 62:849–855
Sasaki T, Isayama H, Yashima Y et al (2009) S-1 monotherapy in patients with advanced biliary tract cancer. Oncology 77:71–74
Park I, Lee JL, Ryu MH et al (2009) Efficacy and safety of S-1 monotherapy in patients with advanced biliary tract adenocarcinoma: retrospective analysis of 162 patients. Oncology 76:126–132
Sasaki T, Isayama H, Nakai Y et al (2013) A randomized phase II study of gemcitabine and S-1 combination therapy versus gemcitabine monotherapy for advanced biliary tract cancer. Cancer Chemother Pharmacol 71:973–979
Sasaki T, Isayama H, Nakai Y et al (2010) Multicenter, phase II study of gemcitabine and S-1 combination chemotherapy in patients with advanced biliary tract cancer. Cancer Chemother Pharmacol 65:1101–1107
Morizane C, Okusaka T, Mizusawa J et al (2013) Randomized phase II study of gemcitabine plus S-1 versus S-1 in advanced biliary tract cancer: a Japan clinical oncology group trial (JCOG 0805). Cancer Sci 104:1211–1216
Kanai M, Yoshimura K, Tsumura T et al (2011) A multi-institution phase II study of gemcitabine/S-1 combination chemotherapy for patients with advanced biliary tract cancer. Cancer Chemother Pharmacol 67:1429–1434
Kim HS, Kim HY, Zang DY et al (2015) Phase II study of gemcitabine and S-1 combination chemotherapy in patients with metastatic biliary tract cancer. Cancer Chemother Pharmacol 75:711–718
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Moriwaki T, Yamamoto Y, Gosho M et al (2016) Correlations of survival with progression-free survival, response rate, and disease control rate in advanced biliary tract cancer: a meta-analysis of randomised trials of first-line chemotherapy. Br J Cancer 114:881–888
Sasaki T, Isayama H, Nakai Y et al (2011) Prognostic factors in patients with advanced biliary tract cancer receiving chemotherapy. Cancer Chemother Pharmacol 67:847–853
Lamarca A, Hubner RA, David Ryder W, Valle JW (2014) Second-line chemotherapy in advanced biliary cancer: a systematic review. Ann Oncol 25:2328–2338
Brieau B, Dahan L, De Rycke Y et al (2015) Second-line chemotherapy for advanced biliary tract cancer after failure of the gemcitabine-platinum combination: a large multicenter study by the association des gastro-Enterologues Oncologues. Cancer 121:3290–3297
Sato K, Watanabe S, Ohtsubo A et al (2016) Nephrotoxicity of cisplatin combination chemotherapy in thoracic malignancy patients with CKD risk factors. BMC Cancer 16:222
Sasaki T, Isayama H, Nakai Y et al (2014) Treatment outcomes of chemotherapy between unresectable and recurrent biliary tract cancer. World J Gastroenterol 20:18452–18457
Mizusawa J, Morizane C, Okusaka T et al (2016) Randomized phase III study of gemcitabine plus S-1 versus gemcitabine plus cisplatin in advanced biliary tract cancer: Japan clinical oncology group study (JCOG1113, FUGA-BT). Jpn J Clin Oncol 46:385–388
Acknowledgements
We thank all participating patients and their families, as well as the investigators Drs. Ryunosuke Hakuta, Gyotane Umefune, Tomotaka Saito, Takeo Watanabe, and Kaoru Takagi, and the research coordinators Miyuki Tsuchida and Chiho Takeda (Department of Gastroenterology, the University of Tokyo Hospital).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest related to this article.
Funding
None.
Ethical approval
This study was performed in accordance with the Declaration of Helsinki and was approved by each institutional review board.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Takahara, N., Isayama, H., Nakai, Y. et al. Gemcitabine and S-1 versus gemcitabine and cisplatin treatment in patients with advanced biliary tract cancer: a multicenter retrospective study. Invest New Drugs 35, 269–276 (2017). https://doi.org/10.1007/s10637-017-0430-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-017-0430-7