Abstract
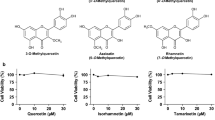
Hyperuricemia is characterized by the high uric acid (UA) level in serum (or plasma) and has been considered to be an important risk factor for gout. In the present study, we have attempted to construct an assay system for UA production in vitro employing cultured AML12 hepatocytes. UA levels in balanced salt solution (BSS) in the presence of UA precursor nucleosides, adenosine, inosine, guanosine and xanthine, at 12.5, 25, and 100 µM were significantly higher than BSS alone and their effects were dose-dependent, while all the UA precursors did not significantly increase intracellular UA levels. Hence, UA levels in BSS were thereafter adopted as an index of UA production. UA production from nucleosides was significantly higher than that from nucleotides (GMP, IMP and AMP). UA production from guanosine and inosine in combination (GI mixture) as well as nucleosides increased time-dependently and almost linearly up to 2 h. Selecting GI mixture, effects of allopurinol, a widely used anti-hyperuricemic agent, and quercetin, a well-known polyphenol in onion and strawberry, on UA production were examined. Both allopurinol and quercetin dose-dependently (0.1, 0.3 and 1 μM for allopurinol and 10, 30, and 100 μM for quercetin) and significantly reduced UA production in the hepatocytes. They also significantly reduced hyperuricemia induced by intraperitoneal injection of UA precursor purine bodies to mice at a single oral dose of 10 (allopurinol) or 200 (quercetin) mg/kg body weight. This assay system for UA production in cultured hepatocytes is considered to be useful to search for novel anti-hyperuricemic compounds in foods and natural resources with possibility to have human health benefits.





Similar content being viewed by others
Abbreviations
- AMP:
-
Adenosine-5′-monophosphate
- BSS:
-
Balanced salt solution
- GMP:
-
Guanosine-5′-monophosphate
- IMP:
-
Inosine-5′-monophosphate
- UA:
-
Uric acid
- XO:
-
Xanthine oxidase
References
Bardin T, Richette P (2014) Definition of hyperuricemia and gouty conditions. Curr Opin Rheumatol 26:186–191
Becker MA, Schumacher HR Jr, Wortmann RL, MacDonald PA, Eustace D, Palo WA, Streit J, Joseph-Ridge N (2005) Febuxostat compared with allopurinol in patients with hyperuricemia and gout. N Engl J Med 353:2450–2461
Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G (2004) Purine-rich foods, daily and protein intake, and the risk of gout in men. N Engl J Med 350:1093–1103
Choi HK, Mount DB, Reginato AM (2005) Pathogenesis of gout. Ann Intern Med 143:499–516
Ishikawa T, Aw W, Kaneko K (2013) Metabolic interactions of purine derivatives with human ABC transporter ABCG2: genetic testing to assess gout risk. Pharmaceuticals (Basel) 6:1347–1360
Kondo M, Hirano Y, Nishio M, Furuya Y, Nakamura H, Watanabe T (2013) Xanthine oxidase inhibitory activity and hypouricemic effect of aspalathin from unfermented rooibos. J Food Sci 78:H1935–H1939
Minakawa M, Miura Y, Yagasaki K (2012) Piceatannol, a resveratrol derivative, promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and suppresses blood glucose levels in type 2 diabetic model db/db mice. Biochem Biophys Res Commun 422:469–475
Miura Y, Shiomi H, Sakai F, Yagasaki K (1997) Assay systems for screening food components that have anti-proliferative and anti-invasive activity to rat ascites hepatoma cells: in vitro and ex vivo effects of green tea extract. Cytotechnology 23:127–132
Miura D, Miura Y, Yagasaki K (2004) Resveratrol inhibits hepatoma cell invasion by suppressing gene expression of hepatocyte growth factor via its reactive oxygen species-scavenging property. Clin Exp Metastasis 21:445–451
Mo SF, Zhou F, Lv YZ, Hu QH, Zhang DM, Kong LD (2007) Hypouricemic action of selected flavonoids in mice: structure-activity relationships. Biol Pharm Bull 30:1551–1556
Nguyen MT, Awale S, Tezuka Y, Tran QL, Watanabe H, Kadota S (2004) Xanthine oxidase inhibitory activity of Vietnamese medicinal plants. Biol Pharm Bull 27:1414–1421
Noro T, Oda Y, Miyase T, Ueno A, Fukushima S (1983) Inhibitors of xanthine oxidase from the flowers and buds of Daphne genkwa. Chem Pharm Bull 31:3984–3987
Petrie JL, Patman GL, Sinha I, Alexander TD, Reeves HL, Agius L (2013) The rate of production of uric acid by hepatocytes is a sensitive index of compromised cell ATP homeostasis. Am J Physiol Endocrinol Metab 305:E1255–E1265
Richette P, Bardin T (2012) Purine-rich foods: an innocent bystander of gout attacks? Ann Rheum Dis 71:1435–1436
Shi Y, Williamson G (2016) Quercetin lowers plasma uric acid in pre-hyperuricaemic males: a randomised, double-blinded, placebo-controlled, cross-over trial. Br J Nutr 115:800–806
Yamaguchi S, Ninomiya K (2000) Umami and food palatability. J Nutr 130:921S–926S
Zhao X, Zhu JX, Mo SF, Pan Y, Kong LD (2006) Effects of cassia oil on serum and hepatic uric acid levels in oxonate-induced mice and xanthine dehydrogenase and xanthine oxidase activities in mouse liver. J Ethnopharmacol 103:357–365
Acknowledgments
This work was supported by the Regional Innovation Strategy Support Program, MEXT, Japan. Authors are grateful to Shinichiro Koike, Yuki Takami and Miku Toyozaki for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
This study was conducted on the basis of general ethical norms indicated by the Japan Society for the Promotion of Science (JSPS).
Conflict of interest
Authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Adachi, Si., Yoshizawa, F. & Yagasaki, K. Assay systems for screening food and natural substances that have anti-hyperuricemic activity: uric acid production in cultured hepatocytes and purine bodies-induced hyperuricemic model mice. Cytotechnology 69, 435–442 (2017). https://doi.org/10.1007/s10616-016-0005-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-016-0005-z