Abstract
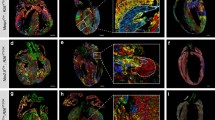
Emerging evidence suggests that mammalian hearts maintain the capacity for cardiac regeneration. Rapid and sensitive identification of cardiac cellular proliferation is prerequisite for understanding the underlying mechanisms and strategies of cardiac regeneration. The following immunologically related markers of cardiac cells were analyzed: cardiac transcription factors Nkx2.5 and Gata 4; specific marker of cardiomyocytes TnT; endothelial cell marker CD31; vascular smooth muscle marker smooth muscle myosin IgG; cardiac resident stem cells markers IsL1, Tbx18, and Wt1. Markers were co-localized in cardiac tissues of embryonic, neonatal, adult, and pathological samples by 5-ethynyl-2′-deoxyuridine (EdU) staining. EdU was also used to label isolated neonatal cardiomyocytes in vitro. EdU robustly labeled proliferating cells in vitro and in vivo, co-immunostaining with different cardiac cells markers. EdU can rapidly and sensitively label proliferating cardiac cells in developmental and pathological states. Cardiac cell proliferation assessed by EdU is a novel analytical tool for investigating the mechanism and strategies of cardiac regeneration in response to injury.




Similar content being viewed by others
References
Anversa P, Kajstura J (1998) Ventricular myocytes are not terminally differentiated in the adult mammalian heart. Circ Res 13:1–14
Beltrami AP, Urbanek K, Kajstura J, Yan SM, Finato N, Bussani R, Nadal-Ginard B, Silvestri F, Leri A, Beltrami CA, Anversa P (2001) Evidence that human cardiac myocytes divided after myocardial infarction. N Eng J Med 344:1750–1757
Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabé-Heider F, Walsh S, Zupicich J, Alkass K, Buchholz BA, Druid H, Jovinge S, Frisén J (2009) Evidence for cardiomyocyte renewal in humans. Science 324:98–102
Buck SB, Bradford J, Gee KR, Agnew BJ, Clarke ST, Salic A (2008) Detection of S-phase cell cycle progression using 5-ethynyl-2′-deoxyuridine incorporation with click chemistry, an alternative to using 5-bromo-2′-deoxyuridine antibodies. Biotechniques 44:927–929
Cappella P, Gasparri F, Pulici M, Moll J (2008) A novel method based on click chemistry, which overcomes limitations of cell cycle analysis byclassical determination of BrdU incorporation, allowing multiplex antibody staining. Cytometry A 73:626–636
Chehrehasa F, Meedeniya AC, Dwyer P, Abrahamsen G, Mackay-Sim A (2009) EdU, a new thymidine analogue for labelling proliferating cells in the nervous system. J Neurosci Methods 15:122–130
Chen K, Bai H, Arzigian M, Gao YX, Bao J, Wu WS, Shen WF, Wu L, Wang ZZ (2010) Endothelial cells regulate cardiomyocyte development from embryonic stem cells. J Cell Biochem 111:29–39
Dodou E, Verzi MP, Anderson JP, Xu SM, Black BL (2004) Mef2c is a direct transcriptional target of ISL1 and GATA factors in the anterior heart field during mouse embryonic development. Development 131:3931–3942
Formigli L, Francini F, Nistri S, Margheri M, Luciani G, Naro F, Silvertown JD, Orlandini SZ, Meacci E, Bani D (2009) Skeletal myoblasts overexpressing relaxin improve differentiation and communication of primary murine cardiomyocyte cell cultures. J Mol Cell Cardiol 47:335–345
Guner-Ataman B, Paffett-Lugassy N, Adams MS, Nevis KR, Jahangiri L, Obregon P, Kikuchi K, Poss KD, Burns CE, Burns CG (2013) Zebrafish second heart field development relies on progenitor specification in anterior lateral plate mesoderm and nkx2.5 function. Development 140:1353–1363
Harvey RP (1996) NK-2 homeobox genes and heart development. Dev Biol 178:203–216
Hsieh PC, Segers VF, Davis ME, MacGillivray C, Gannon J, Molkentin JD, Robbins J, Lee RT (2007) Evidence from a genetic fate-mapping study that stem cells refresh adult mammalian cardiomyocytes after injury. Nat Med 13:970–974
Hsu TL, Hanson SR, Kishikawa K, Wang SK, Sawa M, Wong CH (2007) Alkynyl sugar analogs for the labeling and visualization of glycoconjugates in cells. Proc Natl Acad Sci USA 20:2614–2619
Jopling C, Sleep E, Raya M, Martí M, Raya A, Izpisúa Belmonte JC (2010) Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 25:606–609
Kikuchi K, Holdway JE, Werdich AA, Anderson RM, Fang Y, Egnaczyk GF, Evans T, Macrae CA, Stainier DY, Poss KD (2010) Primary contribution to zebrafish heart regeneration by gata4(+) cardiomyocytes. Nature 25:601–605
Lagrue E, Abe H, Lavanya M, Touhami J, Bodard S, Chalon S, Battini JL, Sitbon M, Castelnau P (2010) Regional characterization of energy metabolism in the brain of normal and MPTP-intoxicated mice using new markers of glucose and phosphate transport. J Biomed Sci 17:91
Lepilina A, Coon AN, Kikuchi K, Holdway JE, Roberts RW, Burns CG, Poss KD (2006) A dynamic epicardial injury response supports progenitor cell activity during zebrafish heart regeneration. Cell 127:607–619
Linask KK, Lash JW (1993) Early heart development: dynamics of endocardial cell sorting suggests a common origin with cardiomyocytes. Dev Dyn 196:62–69
Narmoneva DA, Vukmirovic R, Davis ME, Kamm RD, Lee RT (2004) Endothelial cells promote cardiac myocyte survival and spatial reorganization: implications for cardiac regeneration. Circulation 110:962–968
Nassiri SM, Khaki Z, Soleimani M, Ahmadi SH, Jahanzad I, Rabbani S, Sahebjam M, Ardalan FA, Fathollahi MS (2007) The similar effect of transplantation of marrow-derived mesenchymal stem cells with or without prior differentiation induction in experimental myocardial infarction. J Biomed Sci 14:745–755
Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, Sadek HA (2011) Transient regenerative potential of the neonatal mouse heart. Science 25:1078–1080
Qu D, Wang G, Wang Z, Zhou L, Chi W, Cong S, Ren X, Liang P, Zhang B (2011) 5-Ethynyl-2′-deoxycytidine as a new agent for DNA labeling: detection of proliferating cells. Anal Biochem 417:112–121
Salic A, Mitchison TJ (2008) A chemical method for fast and sensitive detection of DNA synthesis in vivo. Proc Natl Acad Sci USA 19:2415–2420
Saravanakumar M, Devaraj H (2012) Notch signalling in cardiovasculogenesis: insight into their role in early cardiovasculardevelopment. Mol Biol Rep 40:3537–3547
Shu T, Zeng B, Ren X, Li Y (2010) HO-1 modified mesenchymal stem cells modulate MMPs/TIMPs system and adverse remodeling in infarcted myocardium. Tissue Cell 42:217–222
Studzinski GP, Harrison LE (1999) Differentiation-related changes in the cell cycle traverse. Int Rev Cytol 189:1–58
Wada R, Muraoka N, Inagawa K, Yamakawa H, Miyamoto K, Sadahiro T, Umei T, Kaneda R, Suzuki T, Kamiya K, Tohyama S, Yuasa S, Kokaji K, Aeba R, Yozu R, Yamagishi H, Kitamura T, Fukuda K, Ieda M (2013) Induction of human cardiomyocyte-like cells from fibroblasts by defined factors. Proc Natl Acad Sci USA 110:12667–12672
Yang J, Xia J, He Y, Zhao J, Zhang G (2013) MSCs transplantation with application of G-CSF reduces apoptosis or increases VEGF in rabbit model of myocardial infarction. Cytotechnology. doi:10.1007/s10616-013-9655-2
Zeng B, Chen H, Zhu C, Ren X, Lin G, Cao F (2008) Effects of combined mesenchymal stem cells and heme oxygenase-1 therapy on cardiac performance. Eur J Cardiothorac Surg 34:850–856
Zeng B, Lin G, Ren X, Zhang Y, Chong H (2010a) Over-expression of HO-1 on mesenchymal stem cells promotes angiogenesis and improves myocardial function in infarcted myocardium. J Biomed Sci 17:80
Zeng C, Pan F, Jones LA, Lim MM, Griffin EA, Sheline YI, Mintun MA, Holtzman DM, Mach RH (2010b) Evaluation of 5-ethynyl-2′-deoxyuridine staining as a sensitive and reliable method for studying cell proliferation in the adult nervous system. Brain Res 1319:21–32
Zeng B, Ren XF, Cao F, Zhou XY, Zhang J (2011) Developmental patterns and characteristics of epicardial cell markers Tbx18 and Wt1 in murine embryonic heart. J Biomed Sci 18:67
Acknowledgments
This work was supported by the Chinese National Nature Science Foundation (30900609, 81270271).
Conflict of interest
The authors do not have any possible conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
10616_2014_9827_MOESM1_ESM.ppt
Supplementary Fig. 1: EdU-labeled cardiac proliferating cells in adult hearts at E10.5, E17.5, and neonatal mice at 7 days (×40). The highest red fluorescence in the atrial and ventricular walls and ventricular septum (VS) in the fetus; staining was noticeably reduced after birth (PPT 1000 kb)
Rights and permissions
About this article
Cite this article
Zeng, B., Tong, S., Ren, X. et al. Cardiac cell proliferation assessed by EdU, a novel analysis of cardiac regeneration. Cytotechnology 68, 763–770 (2016). https://doi.org/10.1007/s10616-014-9827-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-014-9827-8