Abstract
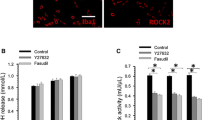
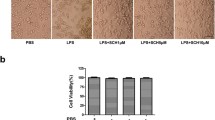
Inflammation within the central nervous system (CNS) is a major component of many neurodegenerative diseases. The underlying mechanisms of neuronal loss are not fully understood, but the activation of CNS resident phagocytic microglia seems to be a significant element contributing to neurodegeneration. At the onset of inflammation, high levels of microglial phagocytosis may serve as an essential prerequisite for creating a favorable environment for neuronal regeneration. However, the excessive and long-lasting activation of microglia and the augmented engulfment of neurons have been suggested to eventually govern widespread neurodegeneration. Here, we investigated in a functional assay of acute inflammation how the small GTPase RhoA and its main target the Rho kinase (ROCK) influence microglial phagocytosis of neuronal debris. Using BV-2 microglia and human NT2 model neurons, we demonstrate that the pain reliever Ibuprofen decreases RhoA activation and microglial phagocytosis of neuronal cell fragments. Inhibition of the downstream effector ROCK with the small-molecule agents Y-27632 and Fasudil reduces the engulfment of neuronal debris and attenuates the production of the inflammatory mediator nitric oxide during stimulation with lipopolysaccharide. Our results support a therapeutic potential for RhoA/ROCK-inhibiting agents as an effective treatment of excessive inflammation and the resulting progression of microglia-mediated neurodegeneration in the CNS.






Similar content being viewed by others
References
Andrews PW (1984) Retinoic acid induces neuronal differentiation of a cloned human embryonal carcinoma cell line in vitro. Dev Biol 103:285–293
Bishop A, Hall A (2000) Rho GTPases and their effector proteins. Biochem J 348:241–255
Blasi E, Barluzzi R, Bocchini V, Mazzolla R, Bistoni F (1990) Immortalization of murine microglial cells by a v-raf/v-myc carrying retrovirus. J Neuroimmunol 27:229–237
Block ML, Hong J-S (2005) Microglia and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Prog Neurobiol 76:77–98
Block ML, Zecca L, Hong J-S (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8:57–69
Brown GC, Neher JJ (2014) Microglial phagocytosis of live neurons. Nat Rev Neurosci 15:209–216
Caron E, Hall A (1998) Identification of two distinct mechanisms of phagocytosis controlled by different Rho GTPases. Science 282:1717–1721
Chimini G, Chavrier P (2000) Function of Rho family proteins in actin dynamics during phagocytosis and engulfment. Nat Cell Biol 2:E191–E196
Cui G, Zuo T, Zhao Q, Hu J, Jin P, Zhao H, Jing J, Zhu J, Chen H, Liu B (2013) ROCK mediates the inflammatory response in thrombin induced microglia. Neurosci Lett 554:82–87
Degasperi A, Birtwistle MR, Volinsky N, Rauch J, Kolch W, Kholodenko BN (2014) Evaluating strategies to normalise biological replicates of western blot data. PLoS ONE 9(1):e87293
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140:918–934
Hackam DJ, Rotstein OD, Schreiber A, Zhang W, Grinstein S (1997) Rho is required for the initiation of calcium signaling and phagocytosis by Fcγ receptors in macrophages. J Exp Med 186:955–966
Hall A (1998) Rho GTPases and the actin cytoskeleton. Science 279:509–514
Hanisch U-K, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10:1387–1394
Hensel N, Rademacher S, Claus P (2015) Chatting with the neighbors: crosstalk between Rho-kinase (ROCK) and other signaling pathways for treatment of neurological disorders. Front Neurosci 9:198
Hiroki J, Shimokawa H, Higashi M, Morikawa K, Kandabashi T, Kawamura N, Kubota T, Ichiki T, Amano M, Kaibuchi K (2004) Inflammatory stimuli upregulate Rho-kinase in human coronary vascular smooth muscle cells. J Mol Cell Cardiol 37:537–546
Kopp M, Liebscher T, Niedeggen A, Laufer S, Brommer B, Jungehulsing G, Strittmatter S, Dirnagl U, Schwab J (2012) Small-molecule-induced Rho-inhibition: NSAIDs after spinal cord injury. Cell Tissue Res 349:119–132
Kreutzberg GW (1996) Microglia: a sensor for pathological events in the CNS. Trends Neurosci 19:312–318
Kubo T, Yamaguchi A, Iwata N, Yamashita T (2008) The therapeutic effects of Rho-ROCK inhibitors on CNS disorders. Ther Clin Risk Manag 4:605–615
Laurenzi MA, Arcuri C, Rossi R, Marconi P, Bocchini V (2001) Effects of microenvironment on morphology and function of the microglial cell line BV-2. Neurochem Res 26:1209–1216
Lucin KM, Wyss-Coray T (2009) Immune activation in brain aging and neurodegeneration: too much or too little? Neuron 64:110–122
Minghetti L, Levi G (1998) Microglia as effector cells in brain damage and repair: focus on prostanoids and nitric oxide. Prog Neurobiol 54:99–125
Mueller BK, Mack H, Teusch N (2005) Rho kinase, a promising drug target for neurological disorders. Nat Rev Drug Discov 4:387–398
Neher JJ, Neniskyte U, Zhao J-W, Bal-Price A, Tolkovsky AM, Brown GC (2011) Inhibition of microglial phagocytosis is sufficient to prevent inflammatory neuronal death. J Immunol 186:4973–4983
Nimmerjahn A, Kirchhoff F, Helmchen F (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308:1314–1318
Olazabal IM, Caron E, May RC, Schilling K, Knecht DA, Machesky LM (2002) Rho-kinase and myosin-II control phagocytic cup formation during CR, but not FcγR, phagocytosis. Curr Biol 12:1413–1418
Orlando KA, Stone NL, Pittman RN (2006) Rho kinase regulates fragmentation and phagocytosis of apoptotic cells. Exp Cell Res 312:5–15
Paquet-Durand F, Bicker G (2007) Human model neurons in studies of brain cell damage and neural repair. Curr Mol Med 7:541–554
Paquet-Durand F, Tan S, Bicker G (2003) Turning teratocarcinoma cells into neurons: rapid differentiation of NT-2 cells in floating spheres. Dev Brain Res 142:161–167
Podrygajlo G, Tegenge MA, Gierse A, Paquet-Durand F, Tan S, Bicker G, Stern M (2009) Cellular phenotypes of human model neurons (NT2) after differentiation in aggregate culture. Cell Tissue Res 336:439–452
Raad M, El Tal T, Gul R, Mondello S, Zhang Z, Boustany R, Guingab J, Wang KK, Kobeissy F (2012) Neuroproteomics approach and neurosystems biology analysis: ROCK inhibitors as promising therapeutic targets in neurodegeneration and neurotrauma. Electrophoresis 33:3659–3668
Ren X, Kiosses WB, Schwartz MA (1999) Regulation of the small GTP-binding protein Rho by cell adhesion and the cytoskeleton. EMBO J 18:578–585
Roloff F, Scheiblich H, Dewitz C, Dempewolf S, Stern M, Bicker G (2015) Enhanced neurite outgrowth of human model (NT2) neurons by small-molecule inhibitors of Rho/ROCK signaling. PLoS ONE 10:e0118536
Scheiblich H, Bicker G (2015a) Regulation of microglial migration, phagocytosis, and neurite outgrowth by HO-1/CO signaling. Dev Neurobiol 75:854–876
Scheiblich H, Bicker G (2015b) Nitric oxide regulates antagonistically phagocytic and neurite outgrowth inhibiting capacities of microglia. Dev Neurobiol. doi:10.1002/dneu.22333 in press
Schmandke A, Schmandke A, Strittmatter SM (2007) ROCK and Rho: biochemistry and neuronal functions of Rho-associated protein kinases. Neuroscientist 13:454–469
Streit WJ, Walter SA, Pennell NA (1999) Reactive microgliosis. Prog Neurobiol 57:563–581
Takeuchi H (2010) Neurotoxicity by microglia: mechanisms and potential therapeutic strategy. Clin Exp Neuroimmunol 1:12–21
Tegenge M, Roloff F, Bicker G (2011) Rapid differentiation of human embryonal carcinoma stem cells (NT2) into neurons for neurite outgrowth analysis. Cell Mol Neurobiol 31:635–643
Tönges L, Koch J-C, Bähr M, Lingor P (2011) ROCKing regeneration: Rho kinase inhibition as molecular target for neurorestoration. Front Mol Neurosci 4:39
Vicente AM, Guillén MI, Alcaraz MJ (2001) Modulation of haem oxygenase-1 expression by nitric oxide and leukotrienes in zymosan-activated macrophages. Br J Pharmacol 133:920–926
Wang C, Song S, Zhang Y, Ge Y, Fang X, Huang T, Du J, Gao J (2015) Inhibition of the Rho/Rho kinase pathway prevents lipopolysaccharide-induced hyperalgesia and the release of TNF-α and IL-1β in the mouse spinal cord. Sci Rep. 5:14553
Zielasek J, Hartung H-P (1996) Molecular mechanisms of microglial activation. Adv Neuroimmunol 6:191–222
Acknowledgments
This work was supported by a grant of the German Research Foundation: BI 262/16-2. The authors would like to thank Saime Tan for generating the human NT2 neurons and our colleagues from the Forschergruppe 1103 for their contributions regarding glial culture. We are grateful to Frank Roloff and Michael Stern for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scheiblich, H., Bicker, G. Regulation of Microglial Phagocytosis by RhoA/ROCK-Inhibiting Drugs. Cell Mol Neurobiol 37, 461–473 (2017). https://doi.org/10.1007/s10571-016-0379-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-016-0379-7