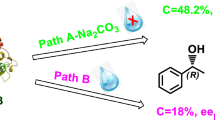
Abstract
Enantioselective deacetylation of a set of benzylic acetates via alcoholysis catalyzed by Lipase B from Candida antarctica (CAL-B), under mild conditions is described. A systematic study allows to determine the appropriate combination nucleophile/organic solvent and also to explain the influence of these parameters on the enzymatic catalytic reaction. In all cases, (R)-alcohols are obtained with high ee (up to >99 %) at conversion 36 % < C < 48 %, the selectivity reaching E > 500. The enzymatic reactivity is influenced by the hydrophobicity of solvent and the structure/nature of the nucleophile. Furthermore, CAL-B allows enantio-complementary between transesterifications in non-aqueous media: alcoholysis and acetylation.
Graphical Abstract






Similar content being viewed by others
References
Anastas PT, Warner JC (1998) Green chemistry: theory and practice. Oxford University Press, New York
Anastas PT, Li PT (2010) In: Yukawa H, Blaschek H (eds) Water as a green solvent. Wiley, New York
Anastas PT, Eghbali N (2010) Chem Soc Rev 39:301
Trost BM (1995) Angew Chem Int Ed 107:285
Carrea G, Riva S (2000) Angew Chem Int Ed 39:2226
Faber K (2011) Biotransformations in organic chemistry, 6th edn. Springer, Berlin
Patel RN (2003) Curr Opin Drug Discov Dev 6:902
Patel RN (2008) Coord Chem Rev 252:659
Ahmed M, Kelly T, Ghanem A (2012) Tetrahedron 68:6781
Pan J, Yu HL, Xu JH, Lin GQ (2011) Advances in biocatalysis: enzymatic reactions and their applications. In: Ma S (ed) Asymmetric catalysis from a Chinese perspective. Springer, Berlin, pp 67–103
Aribi-Zouioueche L, Fiaud JC (2000) Tetrahedron Lett 41:4085
Bidjou C, Aribi-Zouioueche L, Fiaud JC (2002) Tetrahedron Lett 43:3025
Bora PP, Bez G, Anal JMH (2011) J Mol Catal B 72:270
Kumaragura T, Fadnavis NW (2012) Tetrahedron 23:775
Bouzemi N, Grib I, Houiene Z, Aribi-Zouioueche L (2014) Catalysts 4:215
Turner NJ (2003) Curr Opin Biotechnol 14:401
Kamal A, Azhar MA, Krishnaji T, Malik MH, Azeeza S (2008) Coord Chem Rev 252:569
Merabet-Khelassi M, Vriamont N, Riant O, Aribi-Zouioueche L (2011) Tetrahedron 22:1790
Bouzemi N, Debbeche H, Aribi-Zouioueche L, Fiand JC (2004) Tetrahedron Lett 45:627
Merabet-Khelassi M, Bouzemi N, Fiaud JC, Riant O, Aribi-Zouioueche L (2011) C R Chim 14:978
Merabet-Khelassi M, Aribi-Zouioueche L, Riant O (2008) Tetrahedron 19:2378
Merabet-Khelassi M, Aribi-Zouioueche L, Riant O (2009) Tetrahedron 20:1371
Merabet-Khelassi M, Houiene Z, Aribi-Zouioueche L, Riant O (2012) Tetrahedron 23:823
Houiene Z, Merabet-Khelassi M, Bouzemi N, Aribi-Zouioueche L, Riant O (2013) Tetrahedron 24:290
Chen CS, Fujimoto Y, Sih CJ (1982) J Am Chem Soc 104:7294
Kagan HB, Fiaud JC (1998) Kinetic resolution. In: Eliel EL, Wilen SH (eds) Topics in stereochemistry, vol 18. Wiley & Sons, New York, pp 249–330
Baldessari A, Iglesias LE (2012) Methods Mol Biol 861:457
Zhang J, Wu J, Yang L (2004) J Mol Catal B 31:67
Tosa M, Pilbak S, Moldovan P, Paizs C, Szatzker G, Szakacs G, Novak L, Irimie FD, Poppe L (2008) Tetrahedron 19:1844
Kawasaki M, Nakamura K, Kawabata S (1999) J Mol Catal B 6:447
Zhou R, Xu JH (2005) Biochem Eng J 23:11
Kato K, Gong Y, Irimescu R, Saito T, Yokogawa Y (2002) Biotechnol Lett 24:1623
Schieweck F, Altenbach HJ (1998) Tetrahedron 9:403
Bencze LC, Paizs C, Tos MI, Trif M, Irimie FD (2010) Tetrahedron 21:1999
Brem J, Pilbak S, Paizs C, Banoczi G, Irimie FD, Tos MI, Poppe L (2011) Tetrahedron 22:916
Faigl F, Kovacs E, Balogh D, Holczbauer T, Czugler M, Simandi B (2014) Central Eur J Chem 12(1):25
Kitaguchi H, Fitzpatrick PA, Huber JE, Klibanov AM (1989) J Am Chem Soc 111:3094
Woudenberg-van Oosterom M, van Rantwijk F, Sheldon RA (1996) Biotechnol Bioeng 49:328
Fan Y, Xie Z, Zhang H, Qian J (2011) Kinet Catal 52(5):686
Suan CL, Sarmidi MR (2004) J Mol Catal B 28:111
Zilbeyaz K, Taskin M, Kurbanoglu EB, Kurbanoglu NI, Kilic H (2010) Chirality 22:543
Sheldon RA (1996) J Chem Tech Biotechnol 67:1
Pan Y, Tang K-W, He C-Q, Yi W, Zhu W, Liu Y-N (2014) Biotechnol Appl Biochem 61:274
Acknowledgments
Algerian Ministry of Higher Education and Scientific Research (MESRS, FNR 2000) and ANDRU (PNR) are gratefully acknowledged for financial support of this work. Prof. Olivier RIANT (IMCN/Louvain Catholic University -UCL- Louvain-La-Neuve, Belgium) is acknowledged for his help and the welcome of Amna ZAÏDI and Mounia MERABET to perform specific analyses.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zaïdi, A., Merabet-Khelassi, M. & Aribi-Zouioueche, L. CAL-B-Catalyzed Enantioselective Deacetylation of Some Benzylic Acetate Derivatives Via Alcoholysis in Non-aqueous Media. Catal Lett 145, 1054–1061 (2015). https://doi.org/10.1007/s10562-014-1470-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-014-1470-7