Abstract
Purpose
Chronically elevated catecholamine levels activate cardiac β-adrenergic receptors, which play a vital role in the pathogenesis of heart failure. Evidence suggests that vasostatin-1 (VS-1) exerts anti-adrenergic effects on isolated and perfused hearts in vitro. Whether VS-1 ameliorates hypertrophy/remodeling by inducing the chronic activation of β-adrenergic receptors is unknown. The present study aims to test the efficacy of using VS-1 to treat the advanced hypertrophy/remodeling that result from chronic β-adrenergic receptor activation and to determine the cellular and molecular mechanisms that underlie this response.
Methods and Result
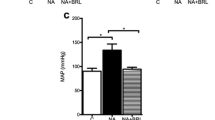
Rats were subjected to infusion with either isoprenaline (ISO, 5 mg/kg/d), ISO plus VS-1 (30 mg/kg/d) or placebo for 2 weeks. VS-1 suppressed chamber dilation, myocyte hypertrophy and fibrosis and improved in vivo heart function in the rats subjected to ISO infusion. VS-1 increased phosphorylated nitric oxide synthase levels and induced the activation of protein kinase G. VS-1 also deactivated multiple hypertrophy signaling pathways that were triggered by the chronic activation of β-adrenergic receptors, such as the phosphoinositide-3 kinase (PI3K)/Akt and Ca2+/calmodulin-dependent kinase (CaMK-II) pathways. Myocytes isolated from ISO + VS-1 hearts displayed higher Ca2+ transients, shorter Ca2+ decays, higher sarcoplasmic reticulum Ca2+ levels and higher L-type Ca2+ current densities than the ISO rat hearts. VS-1 treatment restored the protein expression of sarcoplasmic reticulum Ca2+ uptake ATPase, phospholamban and Cav1.2, indicating improved calcium handling.
Conclusions
Chronic VS-1 treatment inhibited the progression of hypertrophy, fibrosis, and chamber remodeling, and improved cardiac function in a rat model of ISO infusion. In addition, Ca2+ handling and its molecular modulation were also improved by VS-1. The beneficial effects of VS-1 on cardiac remodeling may be mediated by the enhanced activation of the eNOS-cGMP-PKG pathway.






Similar content being viewed by others
References
Samuels MA. The brain-heart connection. Circulation. 2007;116:77–84.
Goldspink DF, Burniston JG, Ellison GM, Clark WA, Tan LB. Catecholamine-induced apoptosis and necrosis in cardiac and skeletal myocytes of the rat in vivo: the same or separate death pathways? Exp Physiol. 2004;89:407–16.
Chien KR, Knowlton KU, Zhu H, Chien S. Regulation of cardiac gene expression during myocardial growth and hypertrophy: molecular studies of an adaptive physiologic response. FASEB J. 1991;5:3037–46.
Kitagawa, Y, Yamashita, D, Ito, H and Takaki, M. Reversible effects of isoproterenol-induced hypertrophy on in situ left vetricular function in rat hearts. Am J Physiol Heart Circ Physiol 2004;287:H277-HH85.
Zhang GX, Kimura S, Nishiyama A, Shokoji T, Rahman M, Yao L, et al. Cardiac oxidative stress in acute and chronic isoproterenol-infused rats. Cardiovasc Res. 2005;65:230–8.
Cohn JN, Levine TB, Olivari MT, Gaberg V, Lura D, Francis GS, et al. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure. N Engl J Med. 1984;311:819–23.
Esler M, Kaye D, Lambert G, Esler D, Jennings G. Adrenergic nervous system in heart failure. Am J Cardiol. 1997;80:7 L–14 L.
Mahapatra NR. Catestatin is a novel endogenous peptide that regulates cardiac function and blood pressure. Cardiovasc Res. 2008;80:330–8.
Corti A, Mannarino C, Mazza R, Angelone T, Longhi R, Tota B, Chromogranin AN. Terminal fragments vasostatin-1 and the synthetic CGA 7-57 peptide act as cardiostatins on the isolated working frog heart. Gen Comp Endocrinol. 2004;136:217–24.
Gallo MP, Levi R, Ramella R, Brero A, Boero O, Tota B, et al. Endothelium-derived nitric oxide mediates the antiadrenergic effect of human vasostatin-1 in rat ventricular myocardium. Am J Physiol Heart Circ Physiol. 2007;292:2906–12.
Cerra MC, De Iuri L, Angelone T, Corti A, Tota B, Recombinant N. Terminal fragments of chromogranin-a modulate cardiac function of the Langendorff-perfused rat heart. Basic Res Cardiol. 2006;101:43–52.
Cappello S, Angelone T, Tota B, Pagliaro P, Penna C, Rastaldo R, et al. Human recombinant chromogranin A-derived vasostatin-1 mimics preconditioning via an adenosine/nitric oxide signaling mechanism. Am J Physiol Heart Circ Physiol. 2007;293:719–27.
Glattard E, Angelone T, Strub JM, Corti A, Aunis D, Tota B, et al. Characterization of natural vasostatin-containing peptides in rat heart. FEBS J. 2006;273:3311–21.
Corti A, Perez Sanchez L, Gasparri A, Curnis F, Longhi R, Branaazza A, et al. Production and structure characterization of recombinant chromograin a N-terminal fragment (vasostatin. Eur J Biochem. 1997;248:692–9.
Huang CX, Yuan MJ, Huang H, Wu G, Liu Y, SB Y, et al. Ghrelin inhibits post-infarct myocardial remodeling and improves cardiac function through anti-inflammation effect. Peptides. 2009;30:2286–91.
Yamamoto K, Dang QN, Kennedy SP, Osathanondh R, Kelly RA, Lee RT. Induction of tenascin-C in cardiac myocytes by mechanical deformation. J Biol Chem. 1999;274:21840–6.
Takinmoto E, Champion HC, Li M, Belardi D, Ren S, Rodriguez ER, et al. Chronic inhibition of cyclic GMP phosphodiesterase 5 A prevents and reverses cardiac hypertrophy. Nat Med. 2005;11:214–22.
Liu B, Ho HT, Velez-Cortes F, Lou Q, Valdivia CR, Knollmann BC, et al. Genetic ablation of ryanodine receptor 2 phosphorylation at Ser-2808 aggravates Ca(2+)-dependent cardiomyopathy by exacerbating diastolic Ca2+ release. J Physiol. 2014;592:1957–73.
Soltysinska E, Olesen SP, Osadchii OE. Myocardial structural, contractile, and electrophysiological changes in the Guinea-pig heart failure model induced by chronic sympathetic activation. Exp Physiol. 2011;96:647–63.
Osadchii OE. Cardiac hypertrophy induced by sustained beta-adrenoreceptor activation: pathophysiological aspects. Heart Fail Rev. 2007;12:66–86.
Naohiro Yano VI, Ting C. Zhao, Andy Tseng, James F. Padbury, and Yi-tang Tseng. A novel signaling pathway for beta-adrenergic receptor-mediated activation of phosphoinositide 3-kinase in H9c2 cardiomyocytes. Am J Physiol Heart Circ Physiol. 2007;293:H385–H93.
Grimm M, Brown JH. Beta-adrenergic receptor signaling in the heart: role of CaMKII. J Mol Cell Cardiol. 2010;48:322–30.
Lincoln T, Potter TC, Vallee L. Willman, David E. Wolfe. Synthesis, binding, release, and metabolism of norepinephrine in normal and transplanted dog hearts. Circ Res. 1965;16:468–81.
Bold AJd. Atrial natriuretic factor: A hormone produced by the heart. Science. 1985;230:767–70.
Tota B, Angelone T, Mazza R, Cerra M. The chromogranin A-derived vasostatins new players in the endocrine heart. Curr Med Chem. 2008;15:1444–51.
Pieroni, M, Corti, A, Tota, B, Curnis, F, Angelone, T, Colombo, B, Cerra, MC, Bellocci, F, Crea, F and Maseri, A. Myocardial production of chromogranin a in human heart a new regulator of cardiac function. Eur Heart J 2007;28:1117–1127.
Brodde OE. Beta-adrenoceptor blocker treatment and the cardiac beta-adrenoceptor-G-protein(s)-adenylyl cyclase system in chronic heart failure. Naunyn Schmiedeberg's Arch Pharmacol. 2007;374:361–72.
Buys ES, Raher MJ, Blake SL, Neilan TG, Graveline AR, Passeri JJ, et al. Cardiomyocyte-restricted restoration of nitric oxide synthase 3 attenuates left ventricular remodeling after chronic pressure overload. Am J Physiol Heart Circ Physiol. 2007;293:H620–H7.
Ichinose F, Bloch KD, JC W, Hataishi R, Aretz HT, Picard MH, et al. Pressure overload-induced LV hypertrophy and dysfunction in mice are exacerbated by congenital NOS3 deficiency. Am J Physiol Heart Circ Physiol. 2004;286:H1070–H5.
Janssens S, Pokreisz P, Schoonjans L, Pellens M, Vermeersch P, Tjwa M, et al. Cardiomyocyte-specific overexpression of nitric oxide synthase 3 improves left ventricular performance and reduces compensatory hypertrophy after myocardial infarction. Circ Res. 2004;94:1256–62.
Ramella R, Boero O, Alloatti G, Angelone T, Levi R, Gallo MP. Vasostatin 1 activates eNOS in endothelial cells through a proteoglycan-dependent mechanism. J Cell Biochem. 2010;110:70–9.
Cerra MC, Gallo MP, Angelone T, Quintieri AM, Pulera E, Filice E, et al. The homologous rat chromogranin A1-64 (rCGA1-64) modulates myocardial and coronary function in rat heart to counteract adrenergic stimulation indirectly via endothelium-derived nitric oxide. FASEB J. 2008;22:3992–4004.
Morisco C, Zebrowski D, Condorelli G, Tsichlis P, Vatner SF, Sadoshima J. The Akt-glycogen synthase kinase 3beta pathway regulates transcription of atrial natriuretic factor induced by beta-adrenergic receptor stimulation in cardiac myocytes. J Biol Chem. 2000;275:14466–75.
Condorelli G, Drusco A, Stassi G, Bellacosa A, Roncarati R, Iaccarino G, et al. Akt induces enhanced myocardial contractility and cell size in vivo in transgenic mice. Proc Natl Acad Sci U S A. 2002;99:12333–8.
Matsui T, Li L, JC W, Cook SA, Nagoshi T, Picard MH, et al. Phenotypic spectrum caused by transgenic overexpression of activated Akt in the heart. J Biol Chem. 2002;277:22896–901.
Hsu S, Nagayama T, Koitabashi N, Zhang M, Zhou L, Bedja D, et al. Phosphodiesterase 5 inhibition blocks pressure overload-induced cardiac hypertrophy independent of the calcineurin pathway. Cardiovasc Res. 2009;81:301–9.
Hasenfuss G, Reinecke H, Studer R, Meyer M, Pieske B, Holtz J, et al. Relation between myocardial function and expression of sarcoplasmic reticulum Ca2 + −ATPase in failing and nonfailing human myocardium. Circ Res. 1994;75:434–42.
Nicolaou P, Kranias EG. Role of PP1 in the regulation of Ca cycling in cardiac physiology and pathophysiology. Front Biosci. 2009;14:3571–85.
Schwinger RHG, Munch G, Bolck B, Karczewski P, Krause E-G, Erdmann E. Reduced Ca2 + −sensitivity of SERCA2a in failing human myocardium due to reduced serin-16 phospholamban phoshorylation. J Mol Cell Cardiol. 1999;31:479–91.
Chu G, Lester JW, Young KB, Luo W, Zhai J, Kranias EGA. Single site (Ser16) phosphorylation in phospholamban is sufficient in mediating its maximal cardiac responses to β-agonists. J Biol Chem. 2000;275:38938–43.
Bers D. Calcium cycling and signaling in cardiac myocytes. Annu Rev Physiol. 2008;70:23–49.
Ling H, Zhang T, Pereira L, Means CK, Cheng H, Gu Y, et al. Requirement for Ca2+/calmodulin-dependent kinase II in the transition from pressure overload-induced cardiac hypertrophy to heart failure in mice. J Clin Invest. 2009;119:1230–40.
Heineke J, Molkentin JD. Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat Rev Mol Cell Biol. 2006;7:589–600.
Jiang LH, Gawler DJ, Hodson N, Milligan CJ, Pearson HA, Porter V, et al. Regulation of cloned cardiac L-type calcium channels by cGMP-dependent protein kinase. J Biol Chem. 2000;275:6135–43.
Schroder F, Single L. Type Ca2+ channel regulation by cGMP-dependent protein kinase type I in adult cardiomyocytes from PKG I transgenic mice. Cardiovasc Res. 2003;60:268–77.
Yeves AM, Garciarena CD, Nolly MB, Chiappe de Cingolani GE, Cingolani HE, Ennis IL. Decreased activity of the Na+/H+ exchanger by phosphodiesterase 5 A inhibition is attributed to an increase in protein phosphatase activity. Hypertension. 2010;56:690–5.
Kilic A, Velic A, De Windt LJ, Fabritz L, Voss M, Mitko D, et al. Enhanced activity of the myocardial Na+/H+ exchanger NHE-1 contributes to cardiac remodeling in atrial natriuretic peptide receptor-deficient mice. Circulation. 2005;112:2307–17.
Perez NG, Piaggio MR, Ennis IL, Garciarena CD, Morales C, Escudero EM, et al. Phosphodiesterase 5 A inhibition induces Na+/H+ exchanger blockade and protection against myocardial infarction. Hypertension. 2007;49:1095–103.
Nagayama T, Hsu S, Zhang M, Koitabashi N, Bedja D, Gabrielson KL, et al. Sildenafil stops progressive chamber, cellular, and molecular remodeling and improves calcium handling and function in hearts with pre-existing advanced hypertrophy caused by pressure overload. J Am Coll Cardiol. 2009;53:207–15.
Klaiber M, Kruse M, Völker K, Schröter J, Feil R, Freichel M, et al. Novel insights into the mechanisms mediating the local antihypertrophic effects of cardiac atrial natriuretic peptide: role of cGMP-dependent protein kinase and RGS2. Basic Res Cardiol. 2010;105:583–95.
Kilic A, Bubikat A, Gassner B, Baba HA, Kuhn M. Local actions of atrial natriuretic peptide counteract angiotensin II stimulated cardiac remodeling. Endocrinology. 2007;148:4162–9.
Acknowledgments
This work was supported by the Natural Science Foundation of Hubei Province, China (no. 2013CFA117) the Fundamental Research Funds for the Central Universities of China (no. 302274052), and Project of Wuhan Municipal Science and Technology (no.2014060101010033). The authors are grateful to the Wuhan University School of Basic Medical Science Medical Research Center for Structural biology for assisting in the performance of the experiments to test Ca2+ transients.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None
Additional information
Dandan Wang and Yinguang Shan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, D., Shan, Y., Huang, Y. et al. Vasostatin-1 Stops Structural Remodeling and Improves Calcium Handling via the eNOS-NO-PKG Pathway in Rat Hearts Subjected to Chronic β-Adrenergic Receptor Activation. Cardiovasc Drugs Ther 30, 455–464 (2016). https://doi.org/10.1007/s10557-016-6687-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-016-6687-9