Abstract
Background
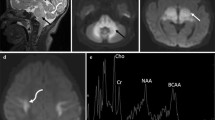
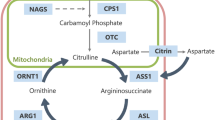
In glutaric aciduria type 1 (GA1) the neurotoxic metabolites glutaric acid (GA) and 3-hydroxyglutaric acid (3-OH-GA) accumulate within the brain. Due to limited efflux across the blood–brain-barrier biochemical monitoring of intracerebrally accumulating toxic metabolites is as yet not possible.
Aims
To investigate brain metabolic patterns in glutaric aciduria type 1 using 1H magnetic resonance spectroscopy (1H-MRS) with focus on detecting the disease-related neurotoxic metabolites GA and 3-OH-GA.
Patients and methods
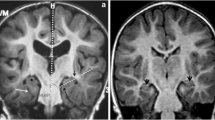
Short echo time 1H-MRS was performed in 13 treated metabolically stable patients. Twenty-one white matter and 16 basal ganglia spectra from 12 patients (age range 7 months - 22 years) were included. Subgroups based on age, biochemical phenotype and/or associated MRI changes were compared with control spectra.
Results
GA was elevated in white matter of patients. 3-OH-GA was elevated in white matter of older patients with associated signal changes on MRI, which was structurally characterized by decreased creatine and phosphocreatine (tCr) and elevated choline (Cho). Metabolite changes differed with biochemical phenotype and disease duration: Low excretors with up to 30 % residual enzyme activity had only mildly, non-significantly elevated GA and mildly subnormal N-acetylaspartate (tNAA). High excretors with complete lack of enzyme activity had significantly increased GA, tNAA was mildly subnormal in younger and decreased in older high excretors.
Conclusions
GA and 3-OH-GA are detectable by in vivo 1H-MRS, which might finally allow biochemical follow-up monitoring of intracerebrally accumulating neurotoxic metabolites in GA1. A high excreting phenotype appears to be a risk factor for cerebral GA accumulation and progressive neuroaxonal compromise despite a similar clinical course in younger high and low excreting patients. This might have consequences for long-term outcome.


Similar content being viewed by others
References
Bähr O, Mader I, Zschocke J, Dichgans J, Schulz J (2002) Adult onset glutaric aciduria type I presenting with a leukoencephalopathy. Neurology 59:1802–1804
Bal D, Gryff-Keller A (2002) 1H and 13C NMR study of 2-hydroxyglutaric acid and its lactone. Magn Reson Chem 40:533–536
Baric I, Wagner L, Feyh P, Liesert M, Buckel W, Hoffmann G (1999) Sensitivity and specificity of free and total glutaric and 3-hydroxyglutaric acid measurements of stable-sotope dilution assays for the diagnosis of glutaric aciduria type I. J Inherit Metab Dis 22:867–881
Barry M, VanSwearingen J, Albright A (1999) Reliability and responsiveness of the Barry-Albright dystonia scale. Dev Med Child Neurol 41:404–411
Bergman I, Finegold D, Gartner J, Zitelli B, Claassen D, Scarano J et al (1989) Acute profound dystonia in infants with glutaric acidemia. Pediatrics 83:228–234
Bodamer OA, Gruber S, Stöckler-Ipsiroglu S (2004) Nuclear magnetic resonance spectroscopy in glutaryl-CoA dehydrogenase deficiency. J Inherit Metab Dis 27:877–883
Boy N, Haege G, Heringer J, Assmann B, Mühlhausen C, Ensenauer R et al (2013) Low lysine diet in glutaric aciduria type I–effect on anthropometric and biochemical follow-up parameters. J Inherit Metab Dis 36:525–533
Cakmakci H, Pekcevik Y, Yis U, Unalp A, Kurul S (2010) Diagnostic value of proton MR spectroscopy and diffusion-weighted MR imaging in childhood inherited neurometabolic brain diseases and review of the literature. Eur J Radiol 74:e161–e171
Chakraborty G, Mekala P, Yahya D, Wu G, Ledeen R (2001) Intraneuronal N-acetylaspartate supplies acetyl groups for myelin lipid synthesis: evidence for myelin-associated aspartoacylase. J Neurochem 78:736–745
Choi C, Ganji S, Hulsey K, Madan A, Kovacs Z, Dimitrov I et al (2013) A comparative study of short- and long-TE 1H MRS at 3 T for in vivo detection of 2-hydroxyglutarate in brain tumors. NMR Biomed 26:1242–1250
Christensen E, Ribes A, Merinero B, Zschocke J (2004) Correlation of genotype and phenotype in glutaryl-CoA dehydrogenase deficiency. J Inherit Metab Dis 27:861–868
D’Adamo A, Gidez L, Yatsu F (1968) Acetyl transport mechanisms. Involvement of N-Acetyl aspartic acid in de novo fatty acid biosynthesis in the developing Rat brain. Exp Brain Res 5:267–273
Elster A (2004) Glutaric aciduria type I: value of diffusion-weighted magnetic resonance imaging for diagnosing acute striatal necrosis. J Comput Assist Tomogr 28:98–100
Funk CBR, Prasad AN, Frosk P, Sauer S, Kölker S, Greenberg CRDB, Marc R (2005) Neuropathological, biochemical and molecular findings in a glutaric acidemia type 1 cohort. Brain 128:711–722
Gerstner B, Gratopp A, Marcinkowski M, Sifringer M, Obladen M, Buhrer C (2005) Glutaric acid and its metabolites cause apoptosis in immature oligodendrocytes: a novel mechanism of white matter degeneration in glutaryl-CoA dehydrogenase deficiency. Pediatr Res 57:771–776
Gitiaux C, Roze E, Kinugawa K, Flamand-Rouvière C, Boddaert N, Apartis E et al (2008) Spectrum of movement disorders associated with glutaric aciduria type 1: a study of 16 patients. Mov Disord 23:2392–2397
Goodman S, Norenberg M, Shikes R, Breslich D, Moe P (1977) Glutaric aciduria: biochemical and morphologic considerations. J Pediatr 90:746–750
Harting I, Neumaier-Probst E, Seitz A, Maier EM, Assmann B, Baric I et al (2009) Dynamic changes of striatal and extrastriatal abnormalities in glutaric aciduria type I. Brain 132:1764–1782
Heringer J, Boy S, Ensenauer R, Assmann B, Zschocke J, Harting I et al (2010) Use of guidelines improves the neurological outcome in glutaric aciduria type I. Ann Neurol 68:743–752
HMDB: Human Metabolome Database. http://www.hmdb.ca., access year 2013
Kölker S, Hoffmann G, Schor D, Feyh P, Wagner L, Jeffrey I et al (2003) Glutaryl-CoA dehydrogenase deficiency: region-specific analysis of organic acids and acylcarnitines in post mortem brain predicts vulnerability of the putamen. Neuropediatrics 34:253–260
Kölker S, Koeller D, Sauer S, Hörster F, Schwab M, Hoffmann G et al (2004) Excitotoxicity and bioenergetics in glutaryl-CoA dehydrogenase deficiency. J Inherit Metab Dis 27:805–812
Kölker S, Garbade S, Greenberg C, Leonard J, Saudubray J, Ribes A et al (2006) Natural history, outcome and therapeutic efficacy in children and adults with glutaryl-CoA dehydrogenase deficiency. Pediatr Res 59:840–847
Kölker S, Christensen E, Leonard J, Greenberg C, Burlina A, Burlina A et al (2007a) Guideline for the diagnosis and management of glutaryl-CoA dehydrogenase deficiency (glutaric aciduria type I). J Inherit Metab Dis 30:5–22
Kölker S, Garbade S, Boy N, Maier E, Meissner T, Mühlhausen C et al (2007b) Decline of acute encephalopathic crises in children with glutaryl-CoA dehydrogenase deficiency identified by newborn screening in Germany. Pediatr Res 62:357–363
Kölker S, Christensen E, Leonard J, Greenberg C, Boneh A, Burlina A et al (2011) Diagnosis and management of glutaric aciduria type I - revised recommendations. J Inherit Metab Dis 34:677–694
Kreis R, Emst T, Ross BD (1993) Development of the human brain: in vivo quantification of metabolite and water content with proton magnetic resonance spectroscopy. Magn Reson Med 30:424–437
Külkens S, Harting I, Sauer S, Zschocke J, Hoffmann GF, Gruber S et al (2005) Late-onset neurologic disease in glutaryl-CoA dehydrogenase deficiency. Neurology 64:2142–2144
Kurul S, Çakmakçi H, Dırık E (2004) Glutaric aciduria type 1: proton magnetic resonance spectroscopy findings. Pediatr Neurol 31:228–231
Lamp J, Keyser B, Koeller D, Ullrich K, Braulke T, Mühlhausen C (2011) Glutaric aciduria type 1 metabolites impair the succinate transport from astrocytic to neuronal cells. J Biol Chem 286:17777–17784
Leibel R, Shih V, Goodman S et al (1980) Glutaric acidemia: a metabolic disorder causing progressive choreoathetosis. Neurology 30:1163–1168
Oguz K, Ozturk A, Cila A (2005) Diffusion-weighted MR imaging and MR spectroscopy in glutaric aciduria type 1. Neuroradiology 47:229–234
Olivera-Bravo S, Fernández A, Sarlabós M, Rosillo J, Casanova G, Jiménez M et al (2011) Neonatal astrocyte damage is sufficient to trigger progressive striatal degeneration in a rat model of glutaric acidemia-I. PLoS One 6:e20831
Olivera-Bravo S, Isasi E, Fernández A, Rosillo J, Jiménez M, Casanova G et al (2014) White matter injury induced by perinatal exposure to glutaric acid. Neurotox Res 25:381–391
Pérez-Duenas B, De La Osaa A, Capdevilab A, Navarro-Sastred A, Leistc A, Ribesd A et al (2009) Brain injury in glutaric aciduria type I: the value of functional techniques in magnetic resonance imaging. Eur J Paediatr Neurol 13:534–540
Pouwels PJW, Brockmann K, Kruse B, Wilken B, Wick M, Hanefeld F et al (1999) Regional age dependence of human brain metabolites from infancy to adulthood as detected by quantitative localized proton MRS. Pediatr Res 46:474–485
Provencher S (1993) Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn Reson Med 30:672–679
Rash J (2010) Molecular disruption of the panglialsyncytium block potassium siphoning and axonal saltatory conduction: pertinence to neuromyelitis optica and other demyelinating diseases of the central nervous system. Neuroscience 168:982–1008
Santos C, Roach E (2005) Glutaric aciduria type I: a neuroimaging diagnosis? J Child Neurol 20:588–590
Sauer S, Okun J, Schwab M, Crnic L, Hoffmann G, Goodman S et al (2005) Bioenergetics in glutaryl-coenzyme a dehydrogenase deficiency: a role for glutaryl-coenzyme a. J Biol Chem 280:21830–21836
Sauer S, Okun J, Fricker G, Mahringer A, Crnic L, Mühlhausen C et al (2006) Intracerebral accumulation of glutaric and 3-hydroxyglutaric acids in glutaryl-coenzyme a dehydrogenase deficiency, a biochemical risk factor for neurodegeneration. J Neurochem 97:899–910
Sauer S, Opp S, Mahringer A, Kaminski M, Thiel C, Okun J et al (2010) Glutaric aciduria type I and methylmalonic aciduria: simulation of cerebral import and export of accumulating neurotoxic dicarboxylic acids in in vitro models of the blood–brain barrier and the choroid plexus. Biochim Biophys Acta 1802:552–560
Seminotti B, Amaral A, da Rosa M, Fernandes C, Leipnitz G, Olivera-Bravo S et al (2013) Disruption of brain redox homeostasis in glutaryl-CoA dehydrogenase deficient mice treated with high dietary lysine supplementation. Mol Genet Metab 108:30–39
Sijens P, Smit G, Meiners L, Oudkerk M, van Spronsen F (2006) Cerebral 1H MR spectroscopy evealing white matter NAA decreases in glutaric aciduria type I. Mol Genet Metab 88:285–289
Soffer D, Amir N, Elpeleg O, Gomori J, Shalev R, Gottschalk-Sabag S (1992) Striatal degeneration and spongy myelinopathy in glutaric acidemia. J Neurol Sci 107:199–204
Sonmez G, Mutlu H, Ozturk E, Sildiroglu H, Keskin A, Basekim C et al (2007) Magnetic resonance imaging findings of adult-onset glutaric aciduria type I. Acta Radiol 48:557–559
Stellmer F, Keyser B, Burckhardt B, Koepsell H, Streichert T, Glatzel M et al (2007) 3-Hydroxyglutaric acid is transported via the sodium-dependent dicarboxylate transporter NaDC3. J Mol Med 85:763–770
VeSPA: Versatile Simulation, Pulses, and Analysis for Magnetic Resonance Spectroscopy. http://scion.duhs.duke.edu/vespa/project
Zinnanti W, Lazovic J, Housman C, LaNoue K, O’Callaghan J, Simpson I et al (2007) Mechanism of age-dependent susceptibility and novel treatment strategy in glutaric acidemia type I. J Clin Invest 117:3258–3270
Acknowledgments
We thank the patients with GA1 and their parents for participation in this study. The study was supported by a grant from the Kindness for Kids Foundation, Munich, Germany, to IH.
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Jutta Gaertner
Electronic supplementary material
Below is the link to the electronic supplementary material.
Suppl.Table 1
Previously reported 1H-MRS of cerebral white matter and basal ganglia. (n: number of patients with MRS; AEC: acute encephalopathic crisis; bgl: basal ganglia; d: day(s); mo: months; yrs: years; TE: echo time) (DOCX 66 kb)
Suppl. Fig. 1
Concentration differences of tNAA and MMLip20 due to inclusion of GA in the basis set (std&GA -std basis sets) plotted against corresponding GA values together with results of linear fitting (tNAA: y=0.16*x+0.023; MMLip20: y=-1.09*x+-0.08). (GIF 7 kb)
Rights and permissions
About this article
Cite this article
Harting, I., Boy, N., Heringer, J. et al. 1H-MRS in glutaric aciduria type 1: impact of biochemical phenotype and age on the cerebral accumulation of neurotoxic metabolites. J Inherit Metab Dis 38, 829–838 (2015). https://doi.org/10.1007/s10545-015-9826-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-015-9826-8