Abstract
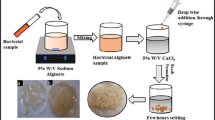
Controlled environments are pivotal in all bioconversion processes, influencing the efficacy of biocatalysts. In this study, we designed a batch bioreactor system with a packed immobilization column and a decontamination chamber to enhance phenol and 2,4-dichlorophenol degradation using the hyper-tolerant bacterium Pseudomonas aeruginosa STV1713. When free cells were employed to degrade phenol and 2,4-DCP at a concentration of 1000 mg/L, the cells completely removed the pollutants within 28 h and 66 h, respectively. Simultaneous reductions in chemical oxygen demand and biological oxygen demand were observed (phenol: 30.21 mg/L/h and 16.92 mg/L/h, respectively; 2,4-dichlorophenol: 12.85 mg/L/h and 7.21 mg/L/h, respectively). After assessing the degradation capabilities, the bacterium was immobilized on various matrices (sodium alginate, alginate-chitosan-alginate and polyvinyl alcohol-alginate) to enhance pollutant removal. Hybrid immobilized cells exhibited greater tolerance and degradation capabilities than those immobilized in a single matrix. Among them, polyvinyl alcohol-alginate immobilized cells displayed the highest degradation capacities (up to 2000 mg/L for phenol and 2500 mg/L for 2,4-dichlorophenol). Morphological analysis of the immobilized cells revealed enhanced cell preservation in hybrid matrices. Furthermore, the elucidation of the metabolic pathway through the catechol dioxygenase enzyme assay indicated higher activity of the catechol 1,2-dioxygenase enzyme, suggesting that the bacterium employed an ortho-degradation mechanism for pollutant removal. Additionally, enzyme zymography confirmed the presence of catechol 1,2-dioxygenase, with the molecular weight of the enzyme determined as 245 kDa.








Similar content being viewed by others
Data availability
The data supporting this study's findings are available from the corresponding author, Suchithra Tharamel Vasu, upon reasonable request.
References
Abarian M, Hassanshahian M, Esbah A (2019) Degradation of phenol at high concentrations using immobilization of Pseudomonas putida P53 into sawdust entrapped in sodium-alginate beads. Water Sci Technol 79:1387–1396. https://doi.org/10.2166/wst.2019.134
Afzal M, Iqbal S, Rauf S, Khalid ZM (2007) Characteristics of phenol biodegradation in saline solutions by monocultures of Pseudomonas aeruginosa and Pseudomonas pseudomallei. J Hazard Mater 149:60–66. https://doi.org/10.1016/j.jhazmat.2007.03.046
Ahmad SA, Shamaan NA, Arif NM et al (2012) Enhanced phenol degradation by immobilized Acinetobacter sp. strain AQ5NOL 1. World J Microbiol Biotechnol 28:347–352. https://doi.org/10.1007/s11274-011-0826-z
Ahmad SA, Shamaan NA, Syed MA et al (2017) Meta-cleavage pathway of phenol degradation by Acinetobacter sp. strain AQ5NOL 1. Rend Lincei 28:1–9. https://doi.org/10.1007/s12210-016-0554-2
Asimakoula S, Marinakos O, Tsagogiannis E, Koukkou AI (2023) Phenol degradation by Pseudarthrobacter phenanthrenivorans Sphe3. Microorganisms 11:524. https://doi.org/10.3390/microorganisms11020524
Bala S, Garg D, Thirumalesh BV et al (2022) Recent strategies for bioremediation of emerging pollutants: a review for a green and sustainable environment. Toxics 10:484. https://doi.org/10.3390/toxics10080484
Barik M, Das CP, Kumar Verma A et al (2021) Metabolic profiling of phenol biodegradation by an indigenous Rhodococcus pyridinivorans strain PDB9T N-1 isolated from paper pulp wastewater. Int Biodeterior Biodegrad 158:105168. https://doi.org/10.1016/j.ibiod.2020.105168
Basak B, Jeon BH, Kurade MB et al (2019) Biodegradation of high concentration phenol using sugarcane bagasse immobilized Candida tropicalis PHB5 in a packed-bed column reactor. Ecotoxicol Environ Saf 180:317–325. https://doi.org/10.1016/j.ecoenv.2019.05.020
Chatzisymeon E, Droumpali A, Mantzavinos D, Venieri D (2011) Disinfection of water and wastewater by UV-A and UV-C irradiation: application of real-time PCR method. Photochem Photobiol Sci 10:389–395. https://doi.org/10.1039/c0pp00161a
Chris Felshia YA, Varadharajan K, Baran A, Mandal AB, Ganesan S, Arumugam G (2014) Maximum phenol tolerance and subsequent degradation profile of a Bacillus pumilus strain mcg03an isolate from tannery wastewater contaminated soil. Int J Curr Res 6:9734–9744
Dell’ Anno F, Rastelli E, Sansone C et al (2021) Bacteria, fungi and microalgae for the bioremediation of marine sediments contaminated by petroleum hydrocarbons in the omics era. Microorganisms 9:1695. https://doi.org/10.3390/microorganisms9081695
Guo F, Chai L, Zhang S et al (2020) Computational biotransformation profile of emerging phenolic pollutants by cytochromes P450: phenol-coupling mechanism. Environ Sci Technol 54:2902–2912. https://doi.org/10.1021/acs.est.9b06897
Hosseini SH, Borghei SM (2005) The treatment of phenolic wastewater using a moving bed bio-reactor. Process Biochem 40:1027–1031. https://doi.org/10.1016/j.procbio.2004.05.002
Keweloh H, Heipieper HJ, Rehm HJ (1989) Protection of bacteria against toxicity of phenol by immobilization in calcium alginate. Appl Microbiol Biotechnol 31:383–389. https://doi.org/10.1007/BF00257609
Krastanov A, Alexieva Z, Yemendzhiev H (2013) Microbial degradation of phenol and phenolic derivatives. Eng Life Sci 13:76–87. https://doi.org/10.1002/elsc.201100227
Lee GL, Zakaria NN, Futamata H et al (2022) Metabolic pathway of phenol degradation of a cold-adapted Antarctic bacteria Arthrobacter sp. Catalysts 12:1422. https://doi.org/10.3390/catal12111422
Lin YH, Cheng YS (2020) Phenol degradation kinetics by free and immobilized Pseudomonas putida BCRC 14365 in batch and continuous-flow bioreactors. Processes 8:721. https://doi.org/10.3390/toxics10080484
Lin YH, Gu YJ (2023) Phenol degradation performance in batch and continuous reactors with immobilized cells of Pseudomonas putida. Processes 11:739. https://doi.org/10.3390/pr11030739
Lowry O, Rosebrough N, Farr AL, Randall R (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Lu D, Zhang Y, Niu S et al (2012) Study of phenol biodegradation using Bacillus amyloliquefaciens strain WJDB-1 immobilized in alginate–chitosan–alginate (ACA) microcapsules by electrochemical method. Biodegradation 23:209–219. https://doi.org/10.1007/s10532-011-9500-2
Mahiudddin M, Fakhruddin ANM, Abdullah-Al M (2012) Degradation of phenol via meta cleavage pathway by Pseudomonas fluorescens PU1. ISRN Microbiol 2012:741820. https://doi.org/10.5402/2012/741820
Mishra V (2017) Microbial degradation of phenol: a review. J Water Pollut Purif Res 32:12–28. https://doi.org/10.1504/IJEP.2008.016895
Mollaei M, Abdollahpour S, Atashgahi S et al (2010) Enhanced phenol degradation by Pseudomonas sp. SA01: gaining insight into the novel single and hybrid immobilizations. J Hazard Mater 175:284–292. https://doi.org/10.1016/j.jhazmat.2009.10.002
Nikel PI, Pérez-Pantoja D, de Lorenzo V (2013) Why are chlorinated pollutants so difficult to degrade aerobically? Redox stress limits 1,3-dichloprop-1-ene metabolism by Pseudomonas pavonaceae. Philos Trans R Soc B Biol Sci 368:20120377. https://doi.org/10.1098/rstb.2012.0377
Oluwasanu AA (2018) Fate and toxicity of chlorinated phenols of environmental implications: a review. Med Anal Chem Int J. https://doi.org/10.23880/macij-16000126
Panigrahy N, Priyadarshini A, Sahoo MM et al (2022) A comprehensive review on eco-toxicity and biodegradation of phenolics: recent progress and future outlook. Environ Technol Innov 27:102423. https://doi.org/10.1016/j.eti.2022.102423
Patel A, Sartaj K, Arora N et al (2017) Biodegradation of phenol via meta cleavage pathway triggers de novo TAG biosynthesis pathway in oleaginous yeast. J Hazard Mater 340:47–56. https://doi.org/10.1016/j.jhazmat.2017.07.013
Pinheiro PF, Menini LAP, Bernardes PC et al (2018) Semisynthetic phenol derivatives obtained from natural phenols: antimicrobial activity and molecular properties. J Agric Food Chem 66:323–330. https://doi.org/10.1021/acs.jafc.7b04418
Ruan B, Wu P, Chen M et al (2018) Immobilization of Sphingomonas sp. GY2B in polyvinyl alcohol–alginate–kaolin beads for efficient degradation of phenol against unfavorable environmental factors. Ecotoxicol Environ Saf 162:103–111. https://doi.org/10.1016/j.ecoenv.2018.06.058
Samudro G, Mangkoedihardjo S (2010) Review on BOD, COD and BOD/COD ratio: a triangle zone for toxic, biodegradable and stable levels. Int J Acad Res 2:235–239
Sasi R, Suchithra TV (2023) Transcriptome analysis of a novel and highly resistant hydrocarbon-degrading bacteria during the removal of phenol and 2,4-dichlorophenol. Biomass Convers Biorefinery 11:1–21. https://doi.org/10.1007/s13399-023-04509-x
Sengupta A, Jebur M, Kamaz M, Wickramasinghe SR (2022) Removal of emerging contaminants from wastewater streams using membrane bioreactors: a review. Membranes 12:60. https://doi.org/10.3390/membranes12010060
Seung HS, Suk SC, Park K, Yoo YJ (2005) Novel hybrid immobilization of microorganisms and its applications to biological denitrification. Enzyme Microb Technol 37:567–573. https://doi.org/10.1016/j.enzmictec.2005.07.012
Shourian M, Noghabi KA, Zahiri HS et al (2009) Efficient phenol degradation by a newly characterized Pseudomonas sp. SA01 isolated from pharmaceutical wastewaters. Desalination 246:577–594. https://doi.org/10.1016/j.desal.2008.07.015
Subramaniam K, Athirrah T, Mazuki T et al (2019) Isolation and optimisation of phenol degradation by antarctic isolate using one factor at time. Malaysian J Biochem Mol Biol 2019:79–86
Subramaniam K, Shaharuddin NA, Tengku-Mazuki TA et al (2020) Statistical optimisation for enhancement of phenol biodegradation by the Antarctic soil bacterium Arthrobacter sp. strain AQ5–15 using response surface methodology py. J Environ Biol 41:1560–1569. https://doi.org/10.22438/JEB/41/6/MRN-1496
Surendra SV, Mahalingam BL, Velan M (2017) Degradation of monoaromatics by Bacillus pumilus MVSV3. Braz Arch Biol Technol 60:1–18. https://doi.org/10.1590/1678-4324-2017160319
Tripathi M, Garg SK (2013) Co-remediation of pentachlorophenol and Cr6+ by free and immobilized cells of native Bacillus cereus isolate: spectrometric characterization of PCP dechlorination products, bioreactor trial and chromate reductase activity. Process Biochem 48:496–509. https://doi.org/10.1016/j.procbio.2013.02.009
Acknowledgements
We want to acknowledge the Kerala State Council for Science, Technology, and Environment for the financial support throughout this study. Also, we would like to acknowledge Dr. K. Haribabu for his valuable suggestions during the design of the batch bioreactor.
Funding
Kerala State Council for Science Technology and Environment (Sasthra Bhavan, Pattom, Thiruvananthapuram, Kerala, India-695004) (Research Fellowship- Sanction order No.29/FSHP/2016/KSCSTE dated 24.03.2017).
Author information
Authors and Affiliations
Contributions
All authors, RS and STV, contributed to the study's conception and design. RS performed material preparation, data collection and experimental analyses. RS wrote the first draft of the manuscript and all authors commented on previous versions. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sasi, R., Vasu, S.T. Batch-mode degradation of high-strength phenolic pollutants by Pseudomonas aeruginosa strain STV1713 immobilized on single and hybrid matrices. Biodegradation (2024). https://doi.org/10.1007/s10532-023-10067-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10532-023-10067-w