Abstract
An aerobic microorganism with an ability to utilize phenol as sole carbon and energy source was isolated from phenol-contaminated wastewater samples. The isolate was identified as Bacillus amyloliquefaciens strain WJDB-1 based on morphological, physiological, and biochemical characteristics, and 16S rDNA sequence analysis. Strain WJDB-1 immobilized in alginate–chitosan–alginate (ACA) microcapsules could degrade 200 mg/l phenol completely within 36 h. The concentration of phenol was determined using differential pulse voltammetry (DPV) at glassy carbon electrode (GCE) with a linear relationship between peak current and phenol concentration ranging from 2.0 to 20.0 mg/l. Cells immobilized in ACA microcapsules were found to be superior to the free suspended ones in terms of improving the tolerance to the environmental loadings. The optimal conditions to prepare microcapsules for achieving higher phenol degradation rate were investigated by changing the concentrations of sodium alginate, calcium chloride, and chitosan. Furthermore, the efficiency of phenol degradation was optimized by adjusting various processing parameters, such as the number of microcapsules, pH value, temperature, and the initial concentration of phenol. This microorganism has the potential for the efficient treatment of organic pollutants in wastewater.










Similar content being viewed by others
References
Adav SS, Chen MY, Lee DJ, Ren NQ (2007) Degradation of phenol by Acinetobacter strain isolated from aerobic granules. Chemosphere 67:1566–1572. doi:10.1016/j.chemosphere.2006.11.067
Ballesteros E, Gallego M, Valcárcel M (1990) Gas chromatographic determination of phenol compounds with automatic continuous extraction and derivatization. J Chromatogr A 518:59–67. doi:10.1016/S0021-9673(01)93162-0
Bayly RC, Wigmore GJ (1973) Metabolism of phenol and cresols by mutants of Pseudomonas putida. J Bacteriol 113:1112–1120
Belloli R, Barletta B, Bolzacchini E, Meinardi S, Orlandi M, Rindone B (1999) Determination of toxic nitrophenols in the atmosphere by high-performance liquid chromatography. J Chromatogr A 846:277–281. doi:10.1016/S0021-9673(99)00030-8
Buswell JA (1975) Metabolism of phenol and cresols by Bacillus stearothermophilus. J Bacteriol 124:1077–1083
Carter RM, Blake RC, Nguyen TD, Bostanian LA (2003) Near real-time biosensor-based detection of 2,4-dinitrophenol. Biosens Bioelectron 18:69–72. doi:10.1016/S0956-5663(02)00107-0
Carvalho RM, Mello C, Kubota LT (2000) Simultaneous determination of phenol isomers in binary mixtures by differential pulse voltammetry using carbon fibre electrode and neural network with pruning as a multivariate calibration tool. Anal Chim Acta 420:109–121. doi:10.1016/S0003-2670(00)01012-6
Chung TP, Tseng HY, Juang RS (2003) Mass transfer effect and intermediate detection for phenol degradation in immobilized Pseudomonas putida systems. Process Biochem 38:1497–1507. doi:10.1016/S0032-9592(03)00038-4
Dąbrowski A, Podkościelny P, Hubicki Z, Barczak M (2005) Adsorption of phenolic compounds by activated carbon—a critical review. Chemosphere 58:1049–1070. doi:10.1016/j.chemosphere.2004.09.067
Deborde M, Rabouan S, Mazellier P, Duguet JP, Legube B (2008) Oxidation of bisphenol A by ozone in aqueous solution. Water Res 42:4299–4308. doi:10.1016/j.watres.2008.07.015
El-Naas MH, Al-Muhtaseb SA, Makhlouf S (2009) Biodegradation of phenol by Pseudomonas putida immobilized in polyvinyl alcohol (PVA) gel. J Hazard Mater 164:720–725. doi:10.1016/j.jhazmat.2008.08.059
Frenzel W, Krekler S (1995) Spectrophotometric determination of total phenolics by solvent extraction and sorbent extraction optosensing using flow injection methodology. Anal Chim Acta 310:437–446. doi:10.1016/0003-2670(95)00141-L
Gernjak W, Krutzler T, Glaser A, Malato S, Caceres J, Bauer R, Fernández-Alba AR (2003) Photo-Fenton treatment of water containing natural phenolic pollutants. Chemosphere 50:71–78. doi:10.1016/s0045-6535(02)00403-4
Gohel MC, Sheth MN, Patel MM, Jani GK, Patel H (1994) Design of chitosan microspheres containing diclofenac sodium. Indian J Pharm Sci 56:210–214
Gong XC, Luo GS, Yang WW, Wu FY (2006) Separation of organic acids by newly developed polysulfone microcapsules containing triotylamine. Sep Purif Technol 48:235–243. doi:10.1016/j.seppur.2005.07.030
Hsieh FM, Huang C, Lin TF, Chen YM, Lin JC (2008) Study of sodium tripolyphosphate-crosslinked chitosan beads entrapped with Pseudomonas putida for phenol degradation. Process Biochem 43:83–92. doi:10.1016/j.procbio.2007.10.016
Hughes EJL, Bayly RC, Skurray RA (1984) Evidence for isofunctional enzymes in the degradation of phenol, m- and p-toluate, and p-cresol via catechol meta-cleavage pathways in Alcaligenes eutrophus. J Bacteriol 158:79–83
Jia X, Wen J, Jiang Y, Bai J, Cheng X (2005) Modeling for batch phenol biodegradation with immobilized Alcaligenes faecalis. AIChE J 52:1294–1303. doi:10.1002/aic.10744
Karigar C, Mahesh A, Nagenahalli M, Yun DJ (2006) Phenol degradation by immobilized cells of Arthrobacter citreus. Biodegradation 17:47–55. doi:10.1007/s10532-005-3048-y
Kim JH, Jeong WH, Karegoudar TB, Kim CK (2002) Stable degradation of benzoate by Klebsiella oxytoca C302 immobilized in alginate and polyurethane. Biotechnol Bioprocess Eng 7:347–351. doi:10.1007/BF02933519
King A, Hellerstrom C, Sandler S, Kulseng B, Andersson A, Skjak-Braek G (1999) Glucose metabolism in vitro of cultured and transplanted mouse pancreatic islets microencapsulated by means of a high-voltage electrostatic field. Diabetes Care 22:B121–B126
Koyama K, Seki M (2004) Cultivation of yeast and plant cells entrapped in the low-viscous liquid core of an alginate membrane capsule prepared using polyethylene glycol. J Biosci Bioeng 97:111–118. doi:10.1016/S1389-1723(04)70177-2
Lim LY, Wan LSC, Thai PY (1997) Chitosan microspheres prepared by emulsification and ionotropic gelation. Drug Dev Ind Pharm 23:981–985. doi:10.3109/03639049709149150
Lin K, Liu W, Gan J (2009) Oxidative removal of bisphenol A by manganese dioxide: efficacy, products, and pathways. Environ Sci Technol 43:3860–3864. doi:10.1021/es900235f
Mahmoud SS, Ahmed MM (2009) Electrocatalytic oxidation of phenol using Ni-Al2O3 composite-coating electrodes. J Alloys Compd 477:570–575. doi:10.1016/j.jallcom.2008.10.160
Manohar S, Kim CK, Karegoudar TB (2001) Enhanced degradation of naphthalene by immobilization of Pseudomonas sp. strain NGK1 in polyurethane foam. Appl Microbiol Biotechnol 55:311–316. doi:10.1007/s002530000488
Marrot B, Barrios-Martinez A, Moulin P, Roche N (2006) Biodegradation of high phenol concentration by activated sludge in an immersed membrane bioreactor. Biochem Eng J 30:174–183. doi:10.1016/j.bej.2006.03.006
Mollaei M, Abdollahpour S, Atashgahi S, Abbasi H, Masoomi F, Rad I, Lotfi AS, Zahiri HS, Vali H, Noghabi KA (2010) Enhanced phenol degradation by Pseudomonas sp. SA01: gaining insight into the novel single and hybrid immobilizations. J Hazard Mater 175:284–292. doi:10.1016/j.jhazmat.2009.10.002
Mordocco A, Kuek C, Jenkins R (1999) Continuous degradation of phenol at low concentration using immobilized Pseudomonas putida. Enzyme Microb Technol 25:530–536. doi:10.1016/S0141-0229(99)00078-2
Neujahr HY, Varga JM (1970) Degradation of phenols by intact cells and cell-free preparations of Trichosporon cutaneum. Eur J Biochem 13:37–44. doi:10.1111/j.1432-1033.1970.tb00896.x
Nuhoglu A, Yalcin B (2005) Modeling of phenol removal in a batch reactor. Process Biochem 40:1233–1239. doi:10.1016/j.procbio.2004.04.003
Parameswarappa S, Karigar C, Nagenahalli M (2008) Degradation of ethylbenzene by free and immobilized Pseudomonas fluorescens-CS2. Biodegradation 19:137–144. doi:10.1007/s10532-007-9121-y
Polymenakou PN, Stephanou EG (2005) Effect of temperature and additional carbon sources on phenol degradation by an indigenous soil Pseudomonad. Biodegradation 16:403–413. doi:10.1007/s10532-004-3333-1
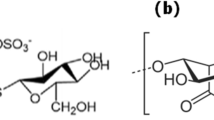
Qi WB, Ma J, Yu WT, Xie YB, Wang W, Ma XJ (2006) Behavior of microbial growth and metabolism in alginate–chitosan–alginate (ACA) microcapsules. Enzyme Microb Technol 38:697–704. doi:10.1016/j.enzmictec.2005.10.003
Rivas FJ, Beltrán FJ, Gimeno O, Alvarez P (2003) Optimisation of Fenton’s reagent usage as a pre-treatment for fermentation brines. J Hazard Mater B96:277–290. doi:10.1016/s0304-3894(02)00217-0
Rodriguez IN, Zamora MB, Salvador JMB, Leyva JAM, Hernandez-Artiga MP, Cisneros JLHH (1997) Voltammetric determination of 2-nitrophenol at a bentonite-modified carbon paste electrode. Mikrochim Acta 126:87–92. doi:10.1007/BF01242667
Saha NC, Bhunia F, Kaviraj A (1999) Toxicity of phenol to fish and aquatic ecosystems. Bull Environ Contam Toxicol 63:195–202. doi:10.1007/s001289900966
Santos VL, Monteiro AS, Braga DT, Santoro MM (2009) Phenol degradation by Aureobasidium pullulans FE13 isolated from industrial effluents. J Hazard Mater 161:1413–1420. doi:10.1016/j.jhazmat.2008.04.112
Straube G (1987) Phenol hydroxylase from Rhodococcus sp. P1. J Basic Microbiol 27:229–232. doi:10.1002/jobm.3620270415
Veenagayathri K, Vasudevan N (2010) Effect of pH, nitrogen sources and salts on the degradation of Phenol by the bacterial consortium under saline conditions. Int J Biotechnol Biochem 6:783–791
Wang XG, Wu QS, Ding YP (2008) Enhancement of surfactants in the electrochemical determination of phenols and the mechanisms research. Colloids Surf A 329:119–124. doi:10.1016/j.colsurfa.2008.07.003
Yu C, Cunningham M, Rogers C, Dinbergs ID, Edelman ER (1998) The biological effects of growth factor-toxin conjugates in models of vascular injury depend on dose, mode of delivery, and animal species. J Pharm Sci 87:1300–1304. doi:10.1021/js980086i
Zhang TY (2004) Study on extracting and determination polyphenols in red bean. China Food Additives 5:99–100. doi:CNKI:SUN:ZSTJ.0.2004-05-025
Acknowledgments
This study is supported by the National Natural Science Foundation of China under Grant Nos. 20577017 and J0730425.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lu, D., Zhang, Y., Niu, S. et al. Study of phenol biodegradation using Bacillus amyloliquefaciens strain WJDB-1 immobilized in alginate–chitosan–alginate (ACA) microcapsules by electrochemical method. Biodegradation 23, 209–219 (2012). https://doi.org/10.1007/s10532-011-9500-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-011-9500-2