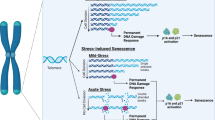
Abstract
Telomeres are the protective structures located at the ends of linear chromosomes. They were first described in the 1930s, but their biology remained unexplored until the early 70s, when Alexey M. Olovnikov, a theoretical biologist, suggested that telomeres cannot be fully copied during DNA replication. He proposed a theory that linked this phenomenon with the limit of cell proliferation capacity and the “duration of life” (theory of marginotomy), and suggested a potential of telomere lenghthening for the prevention of aging (anti-marginotomy). The impact of proliferative telomere shortening on life expectancy was later confirmed. In humans, telomere shortening is counteracted by telomerase, an enzyme that is undetectable in most adult somatic cells, but present in cancer cells and adult and embryonic stem and germ cells. Although telomere length dynamics are different in male and female gametes during gametogenesis, telomere lengths are reset at the blastocyst stage, setting the initial length of the species. The role of the telomere pathway in reproduction has been explored for years, mainly because of increased infertility resulting from delayed childbearing. Short telomere length in ovarian somatic cells is associated to decreased fertility and higher aneuploidy rates in embryos. Consequently, there is a growing interest in telomere lengthening strategies, aimed at improving fertility. It has also been observed that lifestyle factors can affect telomere length and improve fertility outcomes. In this review, we discuss the implications of telomere theory in fertility, especially in oocytes, spermatozoa, and embryos, as well as therapies to enhance reproductive success.


Similar content being viewed by others
Abbreviations
- ALT:
-
Alternative lengthening of telomeres
- ART:
-
Assisted reproductive technologies
- GC:
-
Granulosa cells
- NZ:
-
Normozoospermic
- OAZ:
-
Oligoasthenozoospermic
- PCOS:
-
Polycystic ovary syndrome
- POF:
-
Premature ovarian failure
- Q-FISH:
-
Quantitative fluorescence in-situ hybridization
- ROS:
-
Reactive oxygen species
- STL:
-
Sperm telomere length
- SNT:
-
Somatic nuclear transfer
- TA:
-
Telomerase activity
- TERRA:
-
Telomeric repeat-containing RNA
- TERT:
-
Telomerase REVERSE TRANSCRIPTASE
- TL:
-
Telomere length
- T-SCE:
-
Telomere sister chromatid exchange
- WHO:
-
World Health Organization
References
Achi MAV, Ravindranath N, Dym M (2000) Telomere length in male germ cells is inversely correlated with telomerase activity1. Biol Reprod 63:591–598. https://doi.org/10.1095/biolreprod63.2.591
Aflatoonian A, Arabjahvani F, Eftekhar M, Sayadi M (2014) Effect of vitamin D insufficiency treatment on fertility outcomes in frozen-thawed embryo transfer cycles: a randomized clinical trial. Iran J Reprod Med 12:595–600
Agarwal A, Gupta S, Sharma RK (2005) Role of oxidative stress in female reproduction. Reprod Biol Endocrinol 3:28. https://doi.org/10.1186/1477-7827-3-28
Akino N, Wada-Hiraike O, Isono W, Terao H, Honjo H, Miyamoto Y, Tanikawa M, Sone K, Hirano M, Harada M, Hirata T, Hirota Y, Koga K, Oda K, Fujii T, Osuga Y (2019) Activation of Nrf2/Keap1 pathway by oral Dimethylfumarate administration alleviates oxidative stress and age-associated infertility might be delayed in the mouse ovary. Reprod Biol Endocrinol 17:23. https://doi.org/10.1186/s12958-019-0466-y
Alahmar AT (2019) The impact of two doses of coenzyme Q10 on semen parameters and antioxidant status in men with idiopathic oligoasthenoteratozoospermia. Clin Exp Reprod Med 46:112–118. https://doi.org/10.5653/cerm.2019.00136
Al-Alousi TA, Aziz AA, Al-Allak MMA, Al Ghazali BS (2018) The effect of omega-3 on the number of retrieved ova, fertilization rate, and embryo grading in subfertile women undergoing intracytoplasmic sperm injection. Biomed Pharmacol J 11:2221–2232. https://doi.org/10.13005/bpj/1605
Alam MH, Miyano T (2020) Interaction between growing oocytes and granulosa cells in vitro. Reprod Med Biol 19:13–23. https://doi.org/10.1002/rmb2.12292
Alamo A, Condorelli RA, Mongioi LM, Cannarella R, Giacone F, Calabrese V, La Vignera S, Calogero AE (2019) Environment and male fertility: effects of benzo-alpha-pyrene and resveratrol on human sperm function in vitro. J Clin Med. https://doi.org/10.3390/jcm8040561
Alshinnawy AS, El-Sayed WM, Taha AM, Sayed AA, Salem AM (2020) Astragalus membranaceus and Punica granatum alleviate infertility and kidney dysfunction induced by aging in male rats. Turk J Biol 44:166–175. https://doi.org/10.3906/biy-2001-5
Amirzadegan M, Sadeghi N, Tavalaee M, Nasr-Esfahani MH (2021) Analysis of leukocyte and sperm telomere length in oligozoospermic men. Andrologia 53:e14204. https://doi.org/10.1111/and.14204
Armstrong E, Boonekamp J (2023) Does oxidative stress shorten telomeres in vivo? A meta-analysis. Ageing Res Rev. https://doi.org/10.1016/j.arr.2023.101854
Arora R, Azzalin CM (2015) Telomere elongation chooses TERRA ALTernatives. RNA Biol 12:938–941. https://doi.org/10.1080/15476286.2015.1065374
Aviv A, Susser E (2013) Leukocyte telomere length and the father’s age enigma: implications for population health and for life course. Int J Epidemiol 42:457–462. https://doi.org/10.1093/ije/dys236
Azzalin CM, Reichenbach P, Khoriauli L, Giulotto E, Lingner J (2007) Telomeric repeat containing RNA and RNA surveillance factors at mammalian chromosome ends. Science 318:798–801. https://doi.org/10.1126/science.1147182
Baker TG (1963) A quantitative and cytological study of germ cells in human ovaries. Proc R Soc Lond B Biol Sci 158:417–433. https://doi.org/10.1098/rspb.1963.0055
Balmori C, Varela E (2018) Should we consider telomere length and telomerase activity in male factor infertility? Curr Opin Obstet Gynecol 30:197–202. https://doi.org/10.1097/gco.0000000000000451
Balmori C, Cordova-Oriz I, de Alba G, Medrano M, Jiménez-Tormo L, Polonio AM, Chico-Sordo L, Pacheco A, García-Velasco JA, Varela E (2022) Effects of age and oligoasthenozoospermia on telomeres of sperm and blood cells. Reprod Biomed Online 44:1090–1100. https://doi.org/10.1016/j.rbmo.2021.10.010
Bayne S, Liu JP (2005) Hormones and growth factors regulate telomerase activity in ageing and cancer. Mol Cell Endocrinol 240:11–22. https://doi.org/10.1016/j.mce.2005.05.009
Bentov Y, Hannam T, Jurisicova A, Esfandiari N, Casper RF (2014) Coenzyme Q10 supplementation and oocyte aneuploidy in women undergoing IVF-ICSI treatment. Clin Med Insights Reprod Health 8:31–36. https://doi.org/10.4137/CMRH.S14681
Bernardes De Jesus B, Schneeberger K, Vera E, Tejera A, Harley CB, Blasco MA (2011) The telomerase activator TA-65 elongates short telomeres and increases health span of adult/old mice without increasing cancer incidence. Aging Cell 10:604–621. https://doi.org/10.1111/j.1474-9726.2011.00700.x
Bernardes De Jesus B, Vera E, Schneeberger K, Tejera AM, Ayuso E, Bosch F, Blasco MA (2012) Telomerase gene therapy in adult and old mice delays aging and increases longevity without increasing cancer. EMBO Mol Med 4:691–704. https://doi.org/10.1002/emmm.201200245
Berneau SC, Shackleton J, Nevin C, Altakroni B, Papadopoulos G, Horne G, Brison DR, Murgatroyd C, Povey AC, Carroll M (2019) Associations of sperm telomere length with semen parameters, clinical outcomes and lifestyle factors in human normozoospermic samples. Andrology 8:583–593. https://doi.org/10.1111/andr.12734
Betts DH, King WA (1999) Telomerase activity and telomere detection during early bovine development. Dev Genet 25:397–403. https://doi.org/10.1002/(SICI)1520-6408(1999)25:4%3c397::AID-DVG13%3e3.0.CO;2-J
Blackburn EH, Gall JG (1978) A tandemly repeated sequence at the termini of the extrachromosomal ribosomal RNA genes in Tetrahymena. J Mol Biol 120:33–53. https://doi.org/10.1016/0022-2836(78)90294-2
Blasco MA, Lee HW, Hande MP, Samper E, Lansdorp PM, Depinho RA, Greider CW (1997) Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91:25–34. https://doi.org/10.1016/s0092-8674(01)80006-4
Borum K (1961) Oogenesis in the mouse. A study of the meiotic prophase. Exp Cell Res 24:495–507. https://doi.org/10.1016/0014-4827(61)90449-9
Brook M, Smith JW, Gray NK (2009) The DAZL and PABP families: RNA-binding proteins with interrelated roles in translational control in oocytes. Reproduction 137:595–617. https://doi.org/10.1530/REP-08-0524
Brykczynska U, Hisano M, Erkek S, Ramos L, Oakeley EJ, Roloff TC, Beisel C, Schubeler D, Stadler MB, Peters AH (2010) Repressive and active histone methylation mark distinct promoters in human and mouse spermatozoa. Nat Struct Mol Biol 17:679–687. https://doi.org/10.1038/nsmb.1821
Butts S, Riethman H, Ratcliffe S, Shaunik A, Coutifaris C, Barnhart K (2009) Correlation of telomere length and telomerase activity with occult ovarian insufficiency. J Clin Endocrinol Metab 94:4835–4843. https://doi.org/10.1210/jc.2008-2269
Calado RT, Yewdell WT, Wilkerson KL, Regal JA, Kajigaya S, Stratakis CA, Young NS (2009) Sex hormones, acting on the TERT gene, increase telomerase activity in human primary hematopoietic cells. Blood 114:2236–2243. https://doi.org/10.1182/blood-2008-09-178871
Canudas S, Becerra-Tomas N, Hernandez-Alonso P, Galie S, Leung C, Crous-Bou M, de Vivo I, Gao Y, Gu Y, Meinila J, Milte C, Garcia-Calzon S, Marti A, Boccardi V, Ventura-Marra M, Salas-Salvado J (2020) Mediterranean diet and telomere length: a systematic review and meta-analysis. Adv Nutr 11:1544–1554. https://doi.org/10.1093/advances/nmaa079
Cariati F, Jaroudi S, Alfarawati S, Raberi A, Alviggi C, Pivonello R, Wells D (2016) Investigation of sperm telomere length as a potential marker of paternal genome integrity and semen quality. Reprod Biomed Online 33:404–411. https://doi.org/10.1016/j.rbmo.2016.06.006
Cerone MA, Londono-Vallejo JA, Bacchetti S (2001) Telomere maintenance by telomerase and by recombination can coexist in human cells. Hum Mol Genet 10:1945–1952. https://doi.org/10.1093/hmg/10.18.1945
Cesare AJ, Reddel RR (2010) Alternative lengthening of telomeres: models, mechanisms and implications. Nat Rev Genet 11:319–330. https://doi.org/10.1038/nrg2763
Cheng EH, Chen SU, Lee TH, Pai YP, Huang LS, Huang CC, Lee MS (2013) Evaluation of telomere length in cumulus cells as a potential biomarker of oocyte and embryo quality. Hum Reprod 28:929–936. https://doi.org/10.1093/humrep/det004
Chico-Sordo L, Cordova-Oriz I, Polonio AM, Ls SM, Medrano M, Garcia-Velasco JA, Varela E (2021) Reproductive aging and telomeres: Are women and men equally affected? Mech Ageing Dev 198:111541. https://doi.org/10.1016/j.mad.2021.111541
Choi Y, Rajkovic A (2006) Genetics of early mammalian folliculogenesis. Cell Mol Life Sci 63:579–590. https://doi.org/10.1007/s00018-005-5394-7
Colasante A, Minasi MG, Scarselli F, Casciani V, Zazzaro V, Ruberti A, Greco P, Varricchio MT, Greco E (2019) The aging male: Relationship between male age, sperm quality and sperm DNA damage in an unselected population of 3124 men attending the fertility centre for the first time. Arch Ital Urol Androl 90:254–259. https://doi.org/10.4081/aiua.2018.4.254
Cordova-Oriz I, Chico-Sordo L, Varela E (2022) Telomeres, aging and reproduction. Curr Opin Obstet Gynecol 34:151–158. https://doi.org/10.1097/GCO.0000000000000779
Cordova-Oriz I, Kohls G, Iglesias C, Polonio AM, Chico-Sordo L, Toribio M, Meseguer M, Varela E, Pellicer A, Garcia-Velasco JA (2023) A randomized controlled intervention trial with danazol to improve telomeric and fertility parameters in women with diminished ovarian reserve: a pilot study. Women’s Health Rep 4:305–318. https://doi.org/10.1089/whr.2023.0013
Crous-Bou M, Fung TT, Prescott J, Julin B, Du M, Sun Q, Rexrode KM, Hu FB, de Vivo I (2014) Mediterranean diet and telomere length in nurses’ health study: population based cohort study. BMJ 349:g6674. https://doi.org/10.1136/bmj.g6674
Cusanelli E, Romero CA, Chartrand P (2013) Telomeric noncoding RNA TERRA is induced by telomere shortening to nucleate telomerase molecules at short telomeres. Mol Cell 51:780–791. https://doi.org/10.1016/j.molcel.2013.08.029
Cutillas-Tolin A, Minguez-Alarcon L, Mendiola J, Lopez-Espin JJ, Jorgensen N, Navarrete-Munoz EM, Torres-Cantero AM, Chavarro JE (2015) Mediterranean and western dietary patterns are related to markers of testicular function among healthy men. Hum Reprod 30:2945–2955. https://doi.org/10.1093/humrep/dev236
Darmishonnejad Z, Tavalaee M, Izadi T, Tanhaei S, Nasr-Esfahani MH (2019) Evaluation of sperm telomere length in infertile men with failed/low fertilization after intracytoplasmic sperm injection. Reprod Biomed Online 38:579–587. https://doi.org/10.1016/j.rbmo.2018.12.022
Darmishonnejad Z, Zarei-Kheirabadi F, Tavalaee M, Zarei-Kheirabadi M, Zohrabi D, Nasr-Esfahani MH (2020) Relationship between sperm telomere length and sperm quality in infertile men. Andrologia. https://doi.org/10.1111/and.13546
De Frutos C, Lopez-Cardona AP, Fonseca Balvis N, Laguna-Barraza R, Rizos D, Gutierrez-Adan A, Bermejo-Alvarez P (2016) Spermatozoa telomeres determine telomere length in early embryos and offspring. Reproduction 151:1–7. https://doi.org/10.1530/REP-15-0375
De Lange T (2015) A loopy view of telomere evolution. Front Genet 6:321. https://doi.org/10.3389/fgene.2015.00321
De-Lange T (2018) Shelterin-mediated telomere protection. Annu Rev Genet 52:223–247. https://doi.org/10.1146/annurev-genet-032918-021921
Donate LE, Blasco MA (2011) Telomeres in cancer and ageing. Philos Trans R Soc Lond B Biol Sci 366:76–84. https://doi.org/10.1098/rstb.2010.0291
Duran HE, Simsek-Duran F, Oehninger SC, Jones HW Jr, Castora FJ (2011) The association of reproductive senescence with mitochondrial quantity, function, and DNA integrity in human oocytes at different stages of maturation. Fertil Steril 96:384–388. https://doi.org/10.1016/j.fertnstert.2011.05.056
Dym M (1994) Spermatogonial stem cells of the testis. Proc Natl Acad Sci 91:11287–11289. https://doi.org/10.1073/pnas.91.24.11287
Eisenberg DTA (2011) An evolutionary review of human telomere biology: the thrifty telomere hypothesis and notes on potential adaptive paternal effects. Am J Human Biol 23:149–167. https://doi.org/10.1002/ajhb.21127
Eisenhauer KM, Gerstein RM, Chiu CP, Conti M, Hsueh AJ (1997) Telomerase activity in female and male rat germ cells undergoing meiosis and in early embryos. Biol Reprod 56:1120–1125. https://doi.org/10.1095/biolreprod56.5.1120
Erkek S, Hisano M, Liang CY, Gill M, Murr R, Dieker J, Schubeler D, van der Vlag J, Stadler MB, Peters AH (2013) Molecular determinants of nucleosome retention at CpG-rich sequences in mouse spermatozoa. Nat Struct Mol Biol 20:868–875. https://doi.org/10.1038/nsmb.2599
Espino J, Macedo M, Lozano G, Ortiz A, Rodriguez C, Rodriguez AB, Bejarano I (2019) Impact of Melatonin supplementation in women with unexplained infertility undergoing fertility treatment. Antioxidants (Basel). https://doi.org/10.3390/antiox8090338
Farahzadi R, Fathi E, Mesbah-Namin SA, Zarghami N (2017) Zinc sulfate contributes to promote telomere length extension via increasing telomerase gene expression, telomerase activity and change in the TERT gene promoter CpG island methylation status of human adipose-derived mesenchymal stem cells. PLoS ONE 12:e0188052. https://doi.org/10.1371/journal.pone.0188052
Ferlin A, Rampazzo E, Rocca MS, Keppel S, Frigo AC, de Rossi A, Foresta C (2013) In young men sperm telomere length is related to sperm number and parental age. Hum Reprod 28:3370–3376. https://doi.org/10.1093/humrep/det392
Fernando S, Wallace EM, Vollenhoven B, Lolatgis N, Hope N, Wong M, Lawrence M, Lawrence A, Russell C, Leong K, Thomas P, Rombauts L (2018) Melatonin in assisted reproductive technology: a pilot double-blind randomized placebo-controlled clinical trial. Front Endocrinol (lausanne) 9:545. https://doi.org/10.3389/fendo.2018.00545
Ferreira AF, Soares M, Almeida-Santos T, Ramalho-Santos J, Sousa AP (2023) Aging and oocyte competence: a molecular cell perspective. Wires Mech Dis. https://doi.org/10.1002/wsbm.1613
Florou P, Anagnostis P, Theocharis P, Chourdakis M, Goulis DG (2020) Does coenzyme Q(10) supplementation improve fertility outcomes in women undergoing assisted reproductive technology procedures? A systematic review and meta-analysis of randomized-controlled trials. J Assist Reprod Genet 37:2377–2387. https://doi.org/10.1007/s10815-020-01906-3
Gaspa-Toneu L, Peters AHFM (2023) Nucleosomes in mammalian sperm: conveying paternal epigenetic inheritance or subject to reprogramming between generations? Curr Opin Genet Dev. https://doi.org/10.1016/j.gde.2023.102034
Ghosh S, Feingold E, Chakraborty S, Dey SK (2010) Telomere length is associated with types of chromosome 21 nondisjunction: a new insight into the maternal age effect on Down syndrome birth. Hum Genet 127:403–409. https://doi.org/10.1007/s00439-009-0785-8
Gonzalo S, Jaco I, Fraga MF, Chen T, Li E, Esteller M, Blasco MA (2006) DNA methyltransferases control telomere length and telomere recombination in mammalian cells. Nat Cell Biol 8:416–424. https://doi.org/10.1038/ncb1386
Gou L-T, Lim D-H, Ma W, Aubol BE, Hao Y, Wang X, Zhao J, Liang Z, Shao C, Zhang X, Meng F, Li H, Zhang X, Xu R, Li D, Rosenfeld MG, Mellon PL, Adams JA, Liu M-F, Fu X-D (2020) Initiation of parental genome reprogramming in fertilized oocyte by splicing kinase SRPK1-catalyzed protamine phosphorylation. Cell 180:1212-1227.e14. https://doi.org/10.1016/j.cell.2020.02.020
Gray KE, Schiff MA, Fitzpatrick AL, Kimura M, Aviv A, Starr JR (2014) Leukocyte telomere length and age at menopause. Epidemiology 25:139–146. https://doi.org/10.1097/EDE.0000000000000017
Greco E, Iacobelli M, Rienzi L, Ubaldi F, Ferrero S, Tesarik J (2005) Reduction of the incidence of sperm DNA fragmentation by oral antioxidant treatment. J Androl 26:349–353. https://doi.org/10.2164/jandrol.04146
Greider CW, Blackburn EH (1985) Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43:405–413
Greider CW, Blackburn EH (1987) The telomere terminal transferase of Tetrahymena is a ribonucleoprotein enzyme with two kinds of primer specificity. Cell 51:887–898
Griffith JD, Comeau L, Rosenfield S, Stansel RM, Bianchi A, Moss H, de Lange T (1999) Mammalian telomeres end in a large duplex loop. Cell 97:503–514. https://doi.org/10.1016/s0092-8674(00)80760-6
Guo C, Armbruster BN, Price DT, Counter CM (2003) In vivo regulation of hTERT expression and telomerase activity by androgen. J Urol 170:615–618. https://doi.org/10.1097/01.ju.0000074653.22766.c8
Harley CB, Futcher AB, Greider CW (1990) Telomeres shorten during ageing of human fibroblasts. Nature 345:458–460. https://doi.org/10.1038/345458a0
Harley CB, Liu W, Blasco M, Vera E, Andrews WH, Briggs LA, Raffaele JM (2011) A natural product telomerase activator as part of a health maintenance program. Rejuvenation Res 14:45–56. https://doi.org/10.1089/rej.2010.1085
Hayflick L, Moorhead PS (1961) The serial cultivation of human diploid cell strains. Exp Cell Res 25:585–621
Hemann MT, Strong MA, Hao LY, Greider CW (2001) The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell 107:67–77. https://doi.org/10.1016/s0092-8674(01)00504-9
Herrera E, Samper E, Martin-Caballero J, Flores JM, Lee HW, Blasco MA (1999) Disease states associated with telomerase deficiency appear earlier in mice with short telomeres. EMBO J 18:2950–2960. https://doi.org/10.1093/emboj/18.11.2950
Illiano E, Trama F, Zucchi A, Iannitti RG, Fioretti B, Costantini E (2020) Resveratrol-based multivitamin supplement increases sperm concentration and motility in idiopathic male infertility: a pilot clinical study. J Clin Med. https://doi.org/10.3390/jcm9124017
Jahromi BN, Sadeghi S, Alipour S, Parsanezhad ME, Alamdarloo SM (2017) Effect of melatonin on the outcome of assisted reproductive technique cycles in women with diminished ovarian reserve: a double-blinded randomized clinical trial. Iran J Med Sci 42:73–78
Jaskelioff M, Muller FL, Paik JH, Thomas E, Jiang S, Adams AC, Sahin E, Kost-Alimova M, Protopopov A, Cadinanos J, Horner JW, Maratos-Flier E, Depinho RA (2011) Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature 469:102–106. https://doi.org/10.1038/nature09603
Jiménez Tuñón JM, Trilles PP, Molina MG, Duvison MH, Pastor BM, Martín PS, Sánchez-Borrego R (2017) A double-blind, randomized prospective study to evaluate the efficacy of previous therapy with melatonin, myo-inositol, folic acid, and selenium in improving the results of an assisted reproductive treatment. Clin Med Insights Therap 9:1. https://doi.org/10.1177/1179559X17742902
Jungwirth A, Giwercman A, Tournaye H, Diemer T, Kopa Z, Dohle G, Krausz C (2012) European association of urology guidelines on male infertility: the 2012 update. Eur Urol 62:324–332. https://doi.org/10.1016/j.eururo.2012.04.048
Kalmbach KH, Fontes Antunes DM, Dracxler RC, Knier TW, Seth-Smith ML, Wang F, Liu L, Keefe DL (2013) Telomeres and human reproduction. Fertil Steril 99:23–29. https://doi.org/10.1016/j.fertnstert.2012.11.039
Karayiannis D, Kontogianni MD, Mendorou C, Douka L, Mastrominas M, Yiannakouris N (2017) Association between adherence to the Mediterranean diet and semen quality parameters in male partners of couples attempting fertility. Hum Reprod 32:215–222. https://doi.org/10.1093/humrep/dew288
Karayiannis D, Kontogianni MD, Mendorou C, Mastrominas M, Yiannakouris N (2018) Adherence to the Mediterranean diet and IVF success rate among non-obese women attempting fertility. Hum Reprod 33:494–502. https://doi.org/10.1093/humrep/dey003
Keefe DL (2020) Telomeres and genomic instability during early development. Eur J Med Genet 63:103638. https://doi.org/10.1016/j.ejmg.2019.03.002
Keefe DL, Marquard K, Liu L (2006) The telomere theory of reproductive senescence in women. Curr Opin Obstet Gynecol 18:280–285. https://doi.org/10.1097/01.gco.0000193019.05686.49
Keefe DL, Liu L, Marquard K (2007) Telomeres and aging-related meiotic dysfunction in women. Cell Mol Life Sci 64:139–143. https://doi.org/10.1007/s00018-006-6466-z
Kermack AJ, Lowen P, Wellstead SJ, Fisk HL, Montag M, Cheong Y, Osmond C, Houghton FD, Calder PC, Macklon NS (2020) Effect of a 6-week “Mediterranean” dietary intervention on in vitro human embryo development: the preconception dietary supplements in assisted reproduction double-blinded randomized controlled trial. Fertil Steril 113:260–269. https://doi.org/10.1016/j.fertnstert.2019.09.041
Kiecolt-Glaser JK, Epel ES, Belury MA, Andridge R, Lin J, Glaser R, Malarkey WB, Hwang BS, Blackburn E (2013) Omega-3 fatty acids, oxidative stress, and leukocyte telomere length: a randomized controlled trial. Brain Behav Immun 28:16–24. https://doi.org/10.1016/j.bbi.2012.09.004
Kossowska-Tomaszczuk K, de Geyter C (2013) Cells with stem cell characteristics in somatic compartments of the ovary. Biomed Res Int 2013:310859. https://doi.org/10.1155/2013/310859
Lafuente R, Gonzalez-Comadran M, Sola I, Lopez G, Brassesco M, Carreras R, Checa MA (2013) Coenzyme Q10 and male infertility: a meta-analysis. J Assist Reprod Genet 30:1147–1156. https://doi.org/10.1007/s10815-013-0047-5
Lafuente R, Bosch-Rue E, Ribas-Maynou J, Alvarez J, Brassesco C, Amengual MJ, Benet J, Garcia-Peiro A, Brassesco M (2018) Sperm telomere length in motile sperm selection techniques: a qFISH approach. Andrologia. https://doi.org/10.1111/and.12840
Lansdorp PM (2022) Telomeres, telomerase and cancer. Arch Med Res 53:741–746. https://doi.org/10.1016/j.arcmed.2022.10.004
Lara-Molina EE, Franasiak JM, Marin D, Tao X, Diaz-Gimeno P, Florensa M, Martin M, Seli E, Pellicer A (2020) Cumulus cells have longer telomeres than leukocytes in reproductive-age women. Fertil Steril 113:217–223. https://doi.org/10.1016/j.fertnstert.2019.08.089
Lavranos TC, Mathis JM, Latham SE, Kalionis B, Shay JW, Rodgers RJ (1999) Evidence for ovarian granulosa stem cells: telomerase activity and localization of the telomerase ribonucleic acid component in bovine ovarian follicles. Biol Reprod 61:358–366. https://doi.org/10.1095/biolreprod61.2.358
Lee HW, Blasco MA, Gottlieb GJ, Horner JW II, Greider CW, Depinho RA (1998) Essential role of mouse telomerase in highly proliferative organs. Nature 392:569–574. https://doi.org/10.1038/33345
Li Y, Deng B, Ouyang N, Yuan P, Zheng L, Wang W (2017) Telomere length is short in PCOS and oral contraceptive does not affect the telomerase activity in granulosa cells of patients with PCOS. J Assist Reprod Genet 34:849–859. https://doi.org/10.1007/s10815-017-0929-z
Li SJ, Su WD, Qiu LJ, Wang X, Liu J (2018) Resveratrol protects human sperm against cryopreservation-induced injury. Zhonghua Nan Ke Xue 24:499–503
Liu L, Blasco M, Trimarchi J, Keefe D (2002) An essential role for functional telomeres in mouse germ cells during fertilization and early development. Dev Biol 249:74–84. https://doi.org/10.1006/dbio.2002.0735
Liu L, Bailey SM, Okuka M, Munoz P, Li C, Zhou L, Wu C, Czerwiec E, Sandler L, Seyfang A, Blasco MA, Keefe DL (2007) Telomere lengthening early in development. Nat Cell Biol 9:1436–1441. https://doi.org/10.1038/ncb1664
Liu S, Li Y, Gao X, Yan JH, Chen ZJ (2010) Changes in the distribution of mitochondria before and after in vitro maturation of human oocytes and the effect of in vitro maturation on mitochondria distribution. Fertil Steril 93:1550–1555. https://doi.org/10.1016/j.fertnstert.2009.03.050
Liu J, Liu M, Ye X, Liu K, Huang J, Wang L, Ji G, Liu N, Tang X, Baltz JM, Keefe DL, Liu L (2012) Delay in oocyte aging in mice by the antioxidant N-acetyl-l-cysteine (NAC). Hum Reprod 27:1411–1420. https://doi.org/10.1093/humrep/des019
Liu JJ, Prescott J, Giovannucci E, Hankinson SE, Rosner B, Han J, de Vivo I (2013a) Plasma vitamin D biomarkers and leukocyte telomere length. Am J Epidemiol 177:1411–1417. https://doi.org/10.1093/aje/kws435
Liu M, Yin Y, Ye X, Zeng M, Zhao Q, Keefe DL, Liu L (2013b) Resveratrol protects against age-associated infertility in mice. Hum Reprod 28:707–717. https://doi.org/10.1093/humrep/des437
Lopez De Silanes I, Grana O, De Bonis ML, Dominguez O, Pisano DG, Blasco MA (2014) Identification of TERRA locus unveils a telomere protection role through association to nearly all chromosomes. Nat Commun 5:4723. https://doi.org/10.1038/ncomms5723
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153:1194–1217. https://doi.org/10.1016/j.cell.2013.05.039
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G (2023) Hallmarks of aging: an expanding universe. Cell 186:243–278. https://doi.org/10.1016/j.cell.2022.11.001
Lundblad V, Szostak JW (1989) A mutant with a defect in telomere elongation leads to senescence in yeast. Cell 57:633–643. https://doi.org/10.1016/0092-8674(89)90132-3
Majzoub A, Agarwal A (2018) Systematic review of antioxidant types and doses in male infertility: benefits on semen parameters, advanced sperm function, assisted reproduction and live-birth rate. Arab J Urol 16:113–124. https://doi.org/10.1016/j.aju.2017.11.013
Mania A, Mantzouratou A, Delhanty JD, Baio G, Serhal P, Sengupta SB (2014) Telomere length in human blastocysts. Reprod Biomed Online 28:624–637. https://doi.org/10.1016/j.rbmo.2013.12.010
Marion RM, Strati K, Li H, Tejera A, Schoeftner S, Ortega S, Serrano M, Blasco MA (2009) Telomeres acquire embryonic stem cell characteristics in induced pluripotent stem cells. Cell Stem Cell 4:141–154. https://doi.org/10.1016/j.stem.2008.12.010
Martinez P, Blasco MA (2017) Telomere-driven diseases and telomere-targeting therapies. J Cell Biol 216:875–887. https://doi.org/10.1083/jcb.201610111
Mcclintock B (1939) The behavior in successive nuclear divisions of a chromosome broken at meiosis. Proc Natl Acad Sci USA 25:405–416. https://doi.org/10.1073/pnas.25.8.405
Meerdo LN, Reed WA, White KL (2005) Telomere-to-centromere ratio of bovine clones, embryos, gametes, fetal cells, and adult cells. Cloning Stem Cells 7:62–73. https://doi.org/10.1089/clo.2005.7.62
Michaeli J, Smoom R, Serruya N, El Ayoubi H, Rotshenker-Olshinka K, Srebnik N, Michaeli O, Eldar-Geva T, Tzfati Y (2022) Leukocyte telomere length correlates with extended female fertility. Cells. https://doi.org/10.3390/cells11030513
Min J, Wright WE, Shay JW (2017) Alternative lengthening of telomeres can be maintained by preferential elongation of lagging strands. Nucl Acids Res 45:2615–2628. https://doi.org/10.1093/nar/gkw1295
Minhas S, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, Cocci A, Corona G, Dimitropoulos K, Gül M, Hatzichristodoulou G, Jones TH, Kadioglu A, Martínez Salamanca JI, Milenkovic U, Modgil V, Russo GI, Serefoglu EC, Tharakan T, Verze P, Salonia A (2021) European association of urology guidelines on male sexual and reproductive health: 2021 update on male infertility. Eur Urol 80:603–620. https://doi.org/10.1016/j.eururo.2021.08.014
Morgan HD, Santos F, Green K, Dean W, Reik W (2005) Epigenetic reprogramming in mammals. Hum Mol Genet 14(11):R47-58. https://doi.org/10.1093/hmg/ddi114
Morin SJ, Tao X, Marin D, Zhan Y, Landis J, Bedard J, Scott RT, Seli E (2018) DNA methylation-based age prediction and telomere length in white blood cells and cumulus cells of infertile women with normal or poor response to ovarian stimulation. Aging (albany) 10:3761–3773. https://doi.org/10.18632/aging.101670
Motta PM, Makabe S, Nottola SA (1997) The ultrastructure of human reproduction. I. The natural history of the female germ cell: origin, migration and differentiation inside the developing ovary. Hum Reprod Update 3:281–295. https://doi.org/10.1093/humupd/3.3.281
Muller HJ (1936) Bar duplication. Science 83:528–530. https://doi.org/10.1126/science.83.2161.528-a
Nadjarzadeh A, Shidfar F, Amirjannati N, Vafa MR, Motevalian SA, Gohari MR, Nazeri Kakhki SA, Akhondi MM, Sadeghi MR (2014) Effect of Coenzyme Q10 supplementation on antioxidant enzymes activity and oxidative stress of seminal plasma: a double-blind randomised clinical trial. Andrologia 46:177–183. https://doi.org/10.1111/and.12062
Nadri P, Ansari-Mahyari S, Jafarpour F, Mahdavi AH, Tanhaei Vash N, Lachinani L, Dormiani K, Nasr Esfahani MH (2022) Melatonin accelerates the developmental competence and telomere elongation in ovine SCNT embryos. PLoS ONE 17:e0267598. https://doi.org/10.1371/journal.pone.0267598
Neto FT, Bach PV, Najari BB, Li PS, Goldstein M (2016) Spermatogenesis in humans and its affecting factors. Semin Cell Dev Biol 59:10–26. https://doi.org/10.1016/j.semcdb.2016.04.009
Olovnikov AM (1971) Principle of marginotomy in template synthesis of polynucleotides. Dokl Akad Nauk SSSR 201:1496–1499
Olovnikov AM (1973) A theory of marginotomy. The incomplete copying of template margin in enzymic synthesis of polynucleotides and biological significance of the phenomenon. J Theor Biol 41:181–190. https://doi.org/10.1016/0022-5193(73)90198-7
Olovnikov AM (1996) Telomeres, telomerase, and aging: origin of the theory. Exp Gerontol 31:443–448. https://doi.org/10.1016/0531-5565(96)00005-8
Opstad TB, Alexander J, Aaseth JO, Larsson A, Seljeflot I, Alehagen U (2022) Selenium and Coenzyme Q(10) intervention prevents telomere attrition, with association to reduced cardiovascular mortality-sub-study of a randomized clinical trial. Nutrients. https://doi.org/10.3390/nu14163346
Ozkan S, Jindal S, Greenseid K, Shu J, Zeitlian G, Hickmon C, Pal L (2010) Replete vitamin D stores predict reproductive success following in vitro fertilization. Fertil Steril 94:1314–1319. https://doi.org/10.1016/j.fertnstert.2009.05.019
Passos JF, Saretzki G, von Zglinicki T (2007) DNA damage in telomeres and mitochondria during cellular senescence: is there a connection? Nucl Acids Res 35:7505–7513. https://doi.org/10.1093/nar/gkm893
Patel S (2018) Polycystic ovary syndrome (PCOS), an inflammatory, systemic, lifestyle endocrinopathy. J Steroid Biochem Mol Biol 182:27–36. https://doi.org/10.1016/j.jsbmb.2018.04.008
Pedroso DCC, Santana VP, Donaires FS, Picinato MC, Giorgenon RC, Santana BA, Pimentel RN, Keefe DL, Calado RT, Ferriani RA, Furtado CLM, Reis RM (2020) Telomere length and telomerase activity in immature oocytes and cumulus cells of women with polycystic ovary syndrome. Reprod Sci 27:1293–1303. https://doi.org/10.1007/s43032-019-00120-6
Polonio AM, Chico-Sordo L, Cordova-Oriz I, Medrano M, Garcia-Velasco JA, Varela E (2020) Impact of ovarian aging in reproduction: from telomeres and mice models to ovarian rejuvenation. Yale J Biol Med 93:561–569
Polonio AM, Medrano M, Chico-Sordo L, Cordova-Oriz I, Cozzolino M, Montans J, Herraiz S, Seli E, Pellicer A, Garcia-Velasco JA, Varela E (2023) Impaired telomere pathway and fertility in senescence-accelerated mice prone 8 females with reproductive senescence. Aging (albany) 15:4600–4624. https://doi.org/10.18632/aging.204731
Reddel RR, Bryan TM, Murnane JP (1997) Immortalized cells with no detectable telomerase activity. A review. Biochemistry (mosc) 62:1254–1262
Richards JB, Valdes AM, Gardner JP, Paximadas D, Kimura M, Nessa A, Lu X, Surdulescu GL, Swaminathan R, Spector TD, Aviv A (2007) Higher serum vitamin D concentrations are associated with longer leukocyte telomere length in women. Am J Clin Nutr 86:1420–1425. https://doi.org/10.1093/ajcn/86.5.1420
Riveiro AR, Brickman JM (2020) From pluripotency to totipotency: an experimentalist’s guide to cellular potency. Development. https://doi.org/10.1242/dev.189845
Rocca MS, Speltra E, Menegazzo M, Garolla A, Foresta C, Ferlin A (2016) Sperm telomere length as a parameter of sperm quality in normozoospermic men. Hum Reprod 31:1158–1163. https://doi.org/10.1093/humrep/dew061
Rocca MS, Foresta C, Ferlin A (2018) Telomere length: lights and shadows on their role in human reproduction. Biol Reprod. https://doi.org/10.1093/biolre/ioy208
Rocca MS, Foresta C, Ferlin A (2019) Telomere length: lights and shadows on their role in human reproduction. Biol Reprod 100:305–317. https://doi.org/10.1093/biolre/ioy208
Rudick B, Ingles S, Chung K, Stanczyk F, Paulson R, Bendikson K (2012) Characterizing the influence of vitamin D levels on IVF outcomes. Hum Reprod 27:3321–3327. https://doi.org/10.1093/humrep/des280
Russo V, Berardinelli P, Martelli A, di Giacinto O, Nardinocchi D, Fantasia D, Barboni B (2006) Expression of telomerase reverse transcriptase subunit (TERT) and telomere sizing in pig ovarian follicles. J Histochem Cytochem 54:443–455. https://doi.org/10.1369/jhc.4A6603.2006
Salvador L, Singaravelu G, Harley CB, Flom P, Suram A, Raffaele JM (2016) A natural product telomerase activator lengthens telomeres in humans: a randomized, double blind, and placebo controlled study. Rejuvenation Res 19:478–484. https://doi.org/10.1089/rej.2015.1793
Sanchez F, Smitz J (2012) Molecular control of oogenesis. Biochim Biophys Acta 1822:1896–1912. https://doi.org/10.1016/j.bbadis.2012.05.013
Sato E (2015) Intraovarian control of selective follicular growth and induction of oocyte maturation in mammals. Proc Jpn Acad Ser B Phys Biol Sci 91:76–91. https://doi.org/10.2183/pjab.91.76
Schaetzlein S, Rudolph KL (2005) Telomere length regulation during cloning, embryogenesis and ageing. Reprod Fertil Dev 17:85–96. https://doi.org/10.1071/rd04112
Schaetzlein S, Lucas-Hahn A, Lemme E, Kues WA, Dorsch M, Manns MP, Niemann H, Rudolph KL (2004) Telomere length is reset during early mammalian embryogenesis. Proc Natl Acad Sci USA 101:8034–8038. https://doi.org/10.1073/pnas.0402400101
Schmid TE, Eskenazi B, Marchetti F, Young S, Weldon RH, Baumgartner A, Anderson D, Wyrobek AJ (2012) Micronutrients intake is associated with improved sperm DNA quality in older men. Fertil Steril 98:1130–1137. https://doi.org/10.1016/j.fertnstert.2012.07.1126
Schoeftner S, Blasco MA (2008) Developmentally regulated transcription of mammalian telomeres by DNA-dependent RNA polymerase II. Nat Cell Biol 10:228–236. https://doi.org/10.1038/ncb1685
Schoeftner S, Blasco MA (2009) A ‘higher order’ of telomere regulation: telomere heterochromatin and telomeric RNAs. EMBO J 28:2323–2336. https://doi.org/10.1038/emboj.2009.197
Sharqawi M, Hantisteanu S, Bilgory A, Aslih N, Shibli Abu Raya Y, Atzmon Y, Estrada D, Limonad O, Meisel-Sharon S, Shalom-Paz E (2022) The impact of lifestyle on sperm function, telomere length, and IVF outcomes. Ame J Men’s Health 1:6. https://doi.org/10.1177/15579883221119931
Shay JW, Wright WE (2000) Hayflick, his limit, and cellular ageing. Nat Rev Mol Cell Biol 1:72–76. https://doi.org/10.1038/35036093
Showell MG, Mackenzie-Proctor R, Jordan V, Hart RJ (2020) Antioxidants for female subfertility. Cochrane Database Syst Rev 8:CD007807. https://doi.org/10.1002/14651858.CD007807.pub4
Silver EW, Eskenazi B, Evenson DP, Block G, Young S, Wyrobek AJ (2005) Effect of antioxidant intake on sperm chromatin stability in healthy nonsmoking men. J Androl 26:550–556. https://doi.org/10.2164/jandrol.04165
Stindl R (2016) The paradox of longer sperm telomeres in older men’s testes: a birth-cohort effect caused by transgenerational telomere erosion in the female germline. Mol Cytogenet 9:1. https://doi.org/10.1186/s13039-016-0224-1
Szostak JW, Blackburn EH (1982) Cloning yeast telomeres on linear plasmid vectors. Cell 29:245–255. https://doi.org/10.1016/0092-8674(82)90109-x
Tamura H, Kawamoto M, Sato S, Tamura I, Maekawa R, Taketani T, Aasada H, Takaki E, Nakai A, Reiter RJ, Sugino N (2017) Long-term melatonin treatment delays ovarian aging. J Pineal Res. https://doi.org/10.1111/jpi.12381
Tarkowski AK (1959) Experiments on the development of isolated blastomeres of mouse eggs. Nature 184:1286–1287. https://doi.org/10.1038/1841286a0
Thakur AS, Littarru GP, Funahashi I, Painkara US, Dange NS, Chauhan P (2015) Effect of ubiquinol therapy on sperm parameters and serum testosterone levels in oligoasthenozoospermic infertile men. J Clin Diagn Res 9:BC01-3. https://doi.org/10.7860/JCDR/2015/13617.6424
Thilagavathi J, Venkatesh S, Dada R (2013) Telomere length in reproduction. Andrologia 45:289–304. https://doi.org/10.1111/and.12008
Tomas-Loba A, Flores I, Fernandez-Marcos PJ, Cayuela ML, Maraver A, Tejera A, Borras C, Matheu A, Klatt P, Flores JM, Vina J, Serrano M, Blasco MA (2008) Telomerase reverse transcriptase delays aging in cancer-resistant mice. Cell 135:609–622. https://doi.org/10.1016/j.cell.2008.09.034
Torra-Massana M, Barragán M, Bellu E, Oliva R, Rodríguez A, Vassena R (2018) Sperm telomere length in donor samples is not related to ICSI outcome. J Assist Reprod Genet 35:649–657. https://doi.org/10.1007/s10815-017-1104-2
Townsley DM, Dumitriu B, Young NS (2016) Danazol treatment for telomere diseases. N Engl J Med 375:1095–1096. https://doi.org/10.1056/NEJMc1607752
Treff NR, Su J, Taylor D, Scott RT Jr (2011) Telomere DNA deficiency is associated with development of human embryonic aneuploidy. PLoS Genet 7:e1002161. https://doi.org/10.1371/journal.pgen.1002161
Turner S, Hartshorne GM (2013) Telomere lengths in human pronuclei, oocytes and spermatozoa. Mol Hum Reprod 19:510–518. https://doi.org/10.1093/molehr/gat021
Turner S, Wong HP, Rai J, Hartshorne GM (2010) Telomere lengths in human oocytes, cleavage stage embryos and blastocysts. Mol Hum Reprod 16:685–694. https://doi.org/10.1093/molehr/gaq048
Turner K, Lynch C, Rouse H, Vasu V, Griffin DK (2019) Direct single-cell analysis of human polar bodies and cleavage-stage embryos reveals no evidence of the telomere theory of reproductive ageing in relation to aneuploidy generation. Cells. https://doi.org/10.3390/cells8020163
Uysal F, Kosebent EG, Toru HS, Ozturk S (2021) Decreased expression of TERT and telomeric proteins as human ovaries age may cause telomere shortening. J Assist Reprod Genet 38:429–441. https://doi.org/10.1007/s10815-020-01932-1
Varela E, Schneider RP, Ortega S, Blasco MA (2011) Different telomere-length dynamics at the inner cell mass versus established embryonic stem (ES) cells. Proc Natl Acad Sci USA 108:15207–15212. https://doi.org/10.1073/pnas.1105414108
Varela E, Munoz-Lorente MA, Tejera AM, Ortega S, Blasco MA (2016) Generation of mice with longer and better preserved telomeres in the absence of genetic manipulations. Nat Commun 7:11739. https://doi.org/10.1038/ncomms11739
Vujkovic M, de Vries JH, Lindemans J, Macklon NS, van der Spek PJ, Steegers EA, Steegers-Theunissen RP (2010) The preconception Mediterranean dietary pattern in couples undergoing in vitro fertilization/intracytoplasmic sperm injection treatment increases the chance of pregnancy. Fertil Steril 94:2096–2101. https://doi.org/10.1016/j.fertnstert.2009.12.079
Wang CW, Yao DS, Horng SG, Chiu HC, Chen CK, Lee CL, Huang HY, Wang HS, Soong YK, Pao CC (2004) Feasibility of human telomerase reverse transcriptase mRNA expression in individual blastomeres as an indicator of early embryo development. J Assist Reprod Genet 21:163–168. https://doi.org/10.1023/b:jarg.0000031249.37862.3a
Wang F, Pan X, Kalmbach K, Seth-Smith ML, Ye X, Antumes DM, Yin Y, Liu L, Keefe DL, Weissman SM (2013) Robust measurement of telomere length in single cells. Proc Natl Acad Sci USA 110:E1906–E1912. https://doi.org/10.1073/pnas.1306639110
Watson JD (1972) Origin of concatemeric T7 DNA. Nat New Biol 239:197–201. https://doi.org/10.1038/newbio239197a0
Wei D, Xie J, Yin B, Hao H, Song X, Liu Q, Zhang C, Sun Y (2017) Significantly lengthened telomere in granulosa cells from women with polycystic ovarian syndrome (PCOS). J Assist Reprod Genet 34:861–866. https://doi.org/10.1007/s10815-017-0945-z
WHO (2023) 1 in 6 people globally affected by infertility: WHO. Saudi Med J 44:524–525. http://www.ncbi.nlm.nih.gov/pubmed/37182913
Wright WE, Piatyszek MA, Rainey WE, Byrd W, Shay JW (1996a) Telomerase activity in human germline and embryonic tissues and cells. Dev Genet 18:173–179. https://doi.org/10.1002/(sici)1520-6408(1996)18:2%3c173::Aid-dvg10%3e3.0.Co;2-3
Wright DL, Jones EL, Mayer JF, Oehninger S, Gibbons WE, Lanzendorf SE (2001) Characterization of telomerase activity in the human oocyte and preimplantation embryo. Mol Hum Reprod 7:947–955. https://doi.org/10.1093/molehr/7.10.947
Xu J, Yang X (2000) Telomerase activity in bovine embryos during early development. Biol Reprod 63:1124–1128. https://doi.org/10.1095/biolreprod63.4.1124
Xu X, Chen X, Zhang X, Liu Y, Wang Z, Wang P, Du Y, Qin Y, Chen ZJ (2017) Impaired telomere length and telomerase activity in peripheral blood leukocytes and granulosa cells in patients with biochemical primary ovarian insufficiency. Hum Reprod 32:201–207. https://doi.org/10.1093/humrep/dew283
Xu Y, Nisenblat V, Lu C, Li R, Qiao J, Zhen X, Wang S (2018) Pretreatment with coenzyme Q10 improves ovarian response and embryo quality in low-prognosis young women with decreased ovarian reserve: a randomized controlled trial. Reprod Biol Endocrinol 16:29. https://doi.org/10.1186/s12958-018-0343-0
Yadav T, Zhang JM, Ouyang J, Leung W, Simoneau A, Zou L (2022) TERRA and RAD51AP1 promote alternative lengthening of telomeres through an R- to D-loop switch. Mol Cell 82:3985-4000 e4. https://doi.org/10.1016/j.molcel.2022.09.026
Yamada-Fukunaga T, Yamada M, Hamatani T, Chikazawa N, Ogawa S, Akutsu H, Miura T, Miyado K, Tarin JJ, Kuji N, Umezawa A, Yoshimura Y (2013) Age-associated telomere shortening in mouse oocytes. Reprod Biol Endocrinol 11:108. https://doi.org/10.1186/1477-7827-11-108
Yang Q, Zhao F, Dai S, Zhang N, Zhao W, Bai R, Sun Y (2015) Sperm telomere length is positively associated with the quality of early embryonic development. Hum Reprod 30:1876–1881. https://doi.org/10.1093/humrep/dev144
Yuan Y, Tan Y, Qiu X, Luo H, Li Y, Li R, Yang X (2023) Sperm telomere length as a novel biomarker of male infertility and embryonic development: a systematic review and meta-analysis. Front Endocrinol. https://doi.org/10.3389/fendo.2022.1079966
Zalenskaya IA, Zalensky AO (2002) Telomeres in mammalian male germline cells. A Surv Cell Biol 2002:1
Zeng S, Liu L, Sun Y, Xie P, Hu L, Yuan D, Chen D, Ouyang Q, Lin G, Lu G (2014) Telomerase-mediated telomere elongation from human blastocysts to embryonic stem cells. J Cell Sci 127:752–762. https://doi.org/10.1242/jcs.131433
Zhao Y, Sfeir AJ, Zou Y, Buseman CM, Chow TT, Shay JW, Wright WE (2009) Telomere extension occurs at most chromosome ends and is uncoupled from fill-in in human cancer cells. Cell 138:463–475. https://doi.org/10.1016/j.cell.2009.05.026
Zhou L, Li L, Hao G, Li B, Yang S, Wang N, Liang J, Sun H, Ma S, Yan L, Zhao C, Wei Y, Niu Y, Zhang R (2021) Sperm mtDNA copy number, telomere length, and seminal spermatogenic cells in relation to ambient air pollution: results of a cross-sectional study in Jing-Jin-Ji region of China. J Hazardous Mater 4:6. https://doi.org/10.1016/j.jhazmat.2020.124308
Zhu L, Hathcock KS, Hande P, Lansdorp PM, Seldin MF, Hodes RJ (1998) Telomere length regulation in mice is linked to a novel chromosome locus. Proc Natl Acad Sci USA 95:8648–8653. https://doi.org/10.1073/pnas.95.15.8648
Funding
The Laboratory of Telomeres and Reproduction is supported by the Instituto de Salud Carlos III (Spanish Government) and co-funded by the European Union, through PI20/00252 to E.V. and by Merck (FER-ISS-2021-0243) to J.A.G.V. The Spanish Ministry of Science, Innovation and Universities is funding I.C.-O. (FPU 18/04068) and A.M.P. (FPU 18/02904) along with I.C.-T. (FPU 19/03226). L.C.-S. is supported by the Instituto de Salud Carlos III (Spanish Government) and co-funded by the European Union (FI19/00008). E.V. was funded by the Spanish Ministry of Science and Innovation through a Torres Quevedo grant (PTQ-16-08242).
Author information
Authors and Affiliations
Contributions
All authors have contributed with specific parts of the text and/ or have revised the final manuscript. In particular: I.C-O. wrote the part "strategies to improve fertility" and made the table. AM.P. wrote the part "telomeres in embryo development". I.C-T. worte the part "telomeres in male reproductice cells". L.C-S. wrote the part "telomeres in female reproductive cells". E.V. wrote the introduction. I.C-O. AM.P. and I.C-T. made the figures. I.C-O. AM.P. and I.C-T, L.C-S. M.M., JA.G-V. and E.V. made a critical reading of the manuscript. JA.G-V. and E.V. obtained funding for the laboratory.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflict of interest that could influence or affect the integrity of this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Córdova-Oriz, I., Polonio, A.M., Cuadrado-Torroglosa, I. et al. Chromosome ends and the theory of marginotomy: implications for reproduction. Biogerontology 25, 227–248 (2024). https://doi.org/10.1007/s10522-023-10071-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-023-10071-w