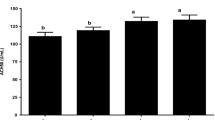
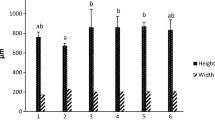
Abstract
Probiotics have evolved as a successful heavy-metal removal approach in aquaculture. In this study, probiotic Lactobacillus plantarum encapsulated with alginate/chitosan nanoparticles were evaluated for lead toxicity mitigation, growth, and hematological development and modification in the intestinal enzyme activity in rainbow trout (Oncorhynchus mykiss). Six hundred juvenile fish (15±1.2 g; Mean±SD) were divided into five groups in triplicates as follows: first and second groups (G1 and G2) were fed with encapsulated and free Lactobacillus plantarum (108 CFU g-1) respectively. The third group (G3) was fed with diets containing alginate/chitosan free probiotic, and the control groups (G4 and G5) were fed with basal diet. After 8 weeks, lead acetate salt (500 μg g-1 of feed) was used for 3 weeks in all groups except G5. Growth, hemato-immunological indices, expression of metallothionein gene in liver as well as intestinal enzymes activity were evaluated at day zero, and weeks 8, 9, 10, and 11. Results showed that almost all growth indices and some immunological significantly (P<0.05) improved in G1 compare to control group. Hematological parameters, intestinal enzymes activity, and resistance against bacterial challenge as well as metallothionein gene expression elevated in G1 and G2 compared to the other groups (P<0.05). Hence, based on the results of this experiment, it can be concluded that using microencapsulated L. plantarum with alginate/chitosan in the diet not only reduced lead destructive toxicity, but also improved the growth and hemato-immunogical indices and resistance against bacterial challenge in rainbow trout, so this method is recommended to remove lead from the polluted ecosystems





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available in below Google drive link: https://drive.google.com/drive/folders/1QqAEBepqYu-S 99FsAmdYaSF8Y0zGV34?usp=share_link
References
Abumourad IM, Abbas WT, Awaad ES, Authman MM, El-Shafei K, Sharaf OM, Ibrahim GA, Sadek ZI, El-Sayed HS (2013) Evaluation of Lactobacillus plantarum as a probiotic in aquaculture: emphasis on growth performance and innate immunity. J Appl Sci Res 9(1):572–582. https://www.researchgate.net/publication/257958772
Alves LC, Wood CM (2006) The chronic effects of dietary lead in freshwater juvenile rainbow trout (Oncorhynchus mykiss) fed elevated calcium diets. Aquat Toxicol 78(3):217–232. https://doi.org/10.1016/j.aquatox.2006.03.005
Aly SM, Ahmed YA, Ghareeb AA, Mohamed MF (2008) Studies on Bacillus subtilis and Lactobacillus acidophilus, as potential probiotics, on the immune response and resistance of Tilapia nilotica (Oreochromis niloticus) to challenge infections. Fish Shellfish Immunol 25(1-2):128–136. https://doi.org/10.1016/j.fsi.2008.03.013
Antunes AE, Liserre AM, Coelho AL, Menezes CR, Moreno I, Yotsuyanagi K, Azambuja NC (2013) Acerola nectar with added microencapsulated probiotic. LWT-Food Sci Technol 54:125–131. https://doi.org/10.1016/j.lwt.2013.04.018
Areekijseree M, Engkagul A, Kovitvadhi U, Thongpan A, Mingmuang M, Pakkong P, Rungruangsak-Torrissen K (2004) Temperature and pH characteristics of amylase and proteinase of adult freshwater pearl mussel, Hyriopsis (Hyriopsis) bialatus Simpson 1900. Aquaculture 234(1-4):575–587. https://doi.org/10.1016/j.aquaculture.2003.12.008
Ashouri G, Soofiani NM, Hoseinifar SH, Jalali SA, Morshedi V, Van Doan H, Mozanzadeh MT (2018) Combined effects of dietary low molecular weight sodium alginate and Pediococcus acidilactici MA18/5M on growth performance, haematological and innate immune responses of Asian sea bass (Lates calcalifer) juveniles. Fish Shellfish Immunol 79:34–41. https://doi.org/10.1016/j.fsi.2018.05.009
Assan D, Kuebutornye FK, Hlordzi V, Chen H, Mraz J, Mustapha UF, Abarike ED (2022) Effects of probiotics on digestive enzymes of fish (finfish and shellfish); status and prospects: a mini review. Comparative Biochem Physiol Part B: Biochem Mol Biol 257:110653. https://doi.org/10.1016/j.cbpb.2021.110653
Badoei-Dalfard A, Khankari S, Karami Z (2020) One-pot synthesis and biochemical characterization of protease metal organic framework (protease@ MOF) and its application on the hydrolysis of fish protein-waste. Coll Surf B: Biointerf 196:111318. https://doi.org/10.1016/j.colsurfb.2020.111318
Bhattacharya S (2019) Probiotics against alleviation of lead toxicity: recent advances. Interdis Toxicol 12:89. https://doi.org/10.2478/intox-2019-0010
Boonanuntanasarn S, Ditthab K, Jangprai A, Nakharuthai C (2019) Effects of microencapsulated Saccharomyces cerevisiae on growth, hematological indices, blood chemical, and immune parameters and intestinal morphology in striped catfish, Pangasianodon hypophthalmus. Probiot Antimicrob Proteins 11:427–437. https://doi.org/10.1007/s12602-018-9404-0
Budiño B, Cal RM, Piazzon MC, Lamas J (2006) The activity of several components of the innate immune system in diploid and triploid turbot. Comparative Biochem Physiol Part A: Mol Integr Physiol 145(1):108–113. https://doi.org/10.1016/j.cbpa.2006.05.007
Burgos-Montes O, Moreno R (2009) Stability of concentrated suspensions of Al2O3–SiO2 measured by multiple light scattering. J Eur Ceram Soc 29(4):603–610. https://doi.org/10.1016/j.jeurceramsoc.2008.07.044
Chang PH, Lin CW, Lee YC (2002) Lactococcus garvieae infection of cultured rainbow trout, Oncorhynchus mykiss in Taiwan and associated biophysical characteristics and histopathology. Bull Eur Assoc Fish Pathol 22:319–327
Charteris C, Kelly K, Morelli M, Collins C (1998) Development and application of an in vitro methodology to determine the transit tolerance of potentially probiotic Lactobacillus and Bifidobacterium species in the upper human gastrointestinal tract. J Appl Microbiol 84(5):759–768. https://doi.org/10.1046/j.1365-2672.1998.00407.x
Chen R, Tu H, Chen T (2022) Potential Application of Living Microorganisms in the Detoxification of Heavy Metals. Foods 11(13):1905. https://doi.org/10.3390/foods11131905
Chowdhury MJ, Baldisserotto B, Wood CM (2005) Tissue-specific cadmium and metallothionein levels in rainbow trout chronically acclimated to waterborne or dietary cadmium. Arch Environ Contaminat Toxicol 48:381–90
Das S, Mandal S, Dhara H, Chatterjee PN (2022) Dietary amelioration of lead toxicity in fish-A review. Indian J Anim Health 61(2):173–189. https://doi.org/10.36062/ijah.2022.spl.03422
Dezfuly ZT, Alishahi M, Ghorbanpoor M, Tabandeh MR, Mesbah M (2020) Immunogenicity and protective efficacy of Yersinia ruckeri lipopolysaccharide (LPS), encapsulated by alginate-chitosan micro/nanoparticles in rainbow trout (Oncorhyncus mykiss). Fish Shellfish Immunol 1(104):25–35
El-Greisy ZA, El-Gamal AH (2015) Experimental studies on the effect of cadmium chloride, zinc acetate, their mixture and the mitigation with vitamin C supplementation on hatchability, size and quality of newly hatched larvae of common carp, Cyprinus carpio. The. Egypt J Aquat Res 41(2):219–226. https://doi.org/10.1016/j.ejar.2015.03.007
Emenike EC, Iwuozor KO, Anidiobi SU (2022) Heavy metal pollution in aquaculture: sources, impacts and mitigation techniques. Biol Trace Element Res 200:4476–4492. https://doi.org/10.1007/s12011-021-03037-x
Erlanger BF, Kokowsky N, Cohen W (1961) The preparation and properties of two new chromogenic substrates of trypsin. Arch Biochem Biophys 95(2):271–278. https://doi.org/10.1016/0003-9861(61)90145-X
Halimi M, Alishahi M, Abbaspour MR, Ghorbanpoor M, Tabandeh MR (2020) High efficacy and economical procedure of oral vaccination against Lactococcus garvieae/Streptococcus iniae in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol 99:505–513. https://doi.org/10.1016/j.fsi.2020.02.033
Hassani F, Peyghan R, Alishahi M, Ghorbanpour M, Ahangarzadeh M (2021) Molecular and biochemical investigation of the role of streptococcus iniae in mortality of Lates calcarifer cages culturing in the Persian Gulf. Exp Animal Biol 9(3):19–27
He N, Wang S, Lv Z, Zhao W, Li S (2020) Low molecular weight chitosan oligosaccharides (LMW-COSs) prevent obesity-related metabolic abnormalities in association with the modification of gut microbiota in high-fat diet (HFD)-fed mice. Food Funct 11(11):9947–9959. https://doi.org/10.1039/D0FO01871F
Homayouni A, Azizi A, Ehsani MR, Yarmand MS, Razavi SH (2008) Effect of microencapsulation and resistant starch on the probiotic survival and sensory properties of synbiotic ice cream. Food Chem 111(1):50–55. https://doi.org/10.1016/j.foodchem.2008.03.036
Hooshyar Y, Abedian Kenari A, Paknejad H, Gandomi H (2020) Effects of Lactobacillus rhamnosus ATCC 7469 on different parameters related to health status of rainbow trout (Oncorhynchus mykiss) and the protection against Yersinia ruckeri. Probiot Antimicrob Proteins 12(4):1370–1384
Hotel AC, Cordoba A (2001) Health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Prevention 5(1):1
Hummel BC (1959) A modified spectrophotometric determination of chymotrypsin, trypsin, and thrombin. Canad J Biochem Physiol 37(12):1393–1399. https://doi.org/10.1139/o59-157
Kandati K, Reddy AS, Nannepaga JS, Viswanath B (2023) The Influence of Probiotics in Reducing Cisplatin-Induced Toxicity in Zebrafish (Danio rerio). Curr Microbiol 80(4):109. https://doi.org/10.1007/s00284-023-03203-5
Kirillova AV, Danilushkina AA, Irisov DS, Bruslik NL, Fakhrullin RF, Zakharov YA, Bukhmin VS, Yarullina DR (2017) Assessment of resistance and bioremediation ability of Lactobacillus strains to lead and cadmium. Int J Microbiol 4:2017. https://dspace.kpfu.ru/xmlui/handle/net/144112
Kuzmina VV, Gavrovskaya LK, Ryzhova OV, Taurine (2010) Effect on exotrophia and metabolism in mammals and fish. J Evol Biochem Physiol 46:19–27. https://doi.org/10.1134/S0022093010010020
Lee B, Paek E, Mitlin D, Lee SW (2019) Sodium metal anodes: emerging solutions to dendrite growth. Chem Rev 119(8):5416–5460. https://doi.org/10.1021/acs.chemrev.8b00642
Li Z, Junaid M, Chen G, Wang J (2022) Interactions and associated resistance development mechanisms between microplastics, antibiotics and heavy metals in the aquaculture environment. Rev Aquacult 14(2):1028–1045. https://doi.org/10.1111/raq.12639
Liu M, Kakade A, Liu P, Wang P, Tang Y, Li X (2019) Hg2+-binding peptide decreases mercury ion accumulation in fish through a cell surface display system. Sci Total Environ 659:540–547. https://doi.org/10.1016/j.scitotenv.2018.12.406
Matsuyama T, Fujiwara A, Nakayasu C, Kamaishi T, Oseko N, Hirono I, Aoki T (2007) Gene expression of leucocytes in vaccinated Japanese flounder (Paralichthys olivaceus) during the course of experimental infection with Edwardsiella tarda. Fish Shellfish Immunol 22(6):598–607. https://doi.org/10.1016/j.fsi.2006.08.006
Michida H, Tamalampudi S, Pandiella SS, Webb C, Fukuda H, Kondo A (2006) Effect of cereal extracts and cereal fiber on viability of Lactobacillus plantarum under gastrointestinal tract conditions. Biochem Eng J 28(1):73–78. https://doi.org/10.1016/j.bej.2005.09.004
Mohammadian T, Alishahi M, Tabandeh MR, Ghorbanpoor M, Gharibi D, Tollabi M, Rohanizade S (2016) Probiotic effects of Lactobacillus plantarum and L. delbrueckii ssp. bulguricus on some immune-related parameters in Barbus grypus. Aquacult Int 24(1):225–242. https://doi.org/10.1007/s10499-015-9921-8
Mohammadian T, Dezfuly ZT, Motlagh RG, Jangaran-Nejad A, Hosseini SS, Khaj H, Alijani N (2020) Effect of encapsulated lactobacillus bulgaricus on innate immune system and hematological parameters in rainbow trout (Oncorhynchus mykiss), post-administration of Pb. Probiot Antimicrob Proteins 12(2):375–388
Mohammadian T, Ghanei-Motlagh R, Jalali M, Nasirpour M, Mohtashamipour H, Osroush E, Nejad AJ (2022) Protective effects of non-encapsulated and microencapsulated Lactobacillus delbrueckii subsp. bulgaricus in rainbow trout (Oncorhynchus mykiss) exposed to lead (Pb) via diet. Ann Animal Sci 22:325–348. https://doi.org/10.2478/aoas-2021-0026
Mohanty BR, Sahoo PK (2010) Immune responses and expression profiles of some immune-related genes in Indian major carp, Labeo rohita to Edwardsiella tarda infection. Fish Shellfish Immunol 1(28):613–621. https://doi.org/10.1016/j.fsi.2009.12.025
Musawi AM, Johari WL, Ikhsan NF, Ahmad SA, Yasid NA, Shukor MY (2019) The Growth Potential and Bioaccumulation Ability of Probiotics under the Exposure of Different Heavy Metals. Pertanika Journal of Tropical Agricultural. Pertanika J Trop Agri 42:1511-3701 e-ISSN: 2231-8542
Narain AS, Srivastava PN (1989) Anemia in the freshwater teleost, Heteropneustes fossilis, under the stress of environmental pollution. Bull Environ Contam Toxicol 43(4):627–634
NavinChandran M, Iyapparaj P, Moovendhan S, Ramasubburayan R, Prakash S, Immanuel G, Palavesam A (2014) Influence of probiotic bacterium Bacillus cereus isolated from the gut of wild shrimp Penaeus monodon in turn as a potent growth promoter and immune enhancer in P. monodon. Fish Shellfish Immunol 36(1):38–45. https://doi.org/10.1016/j.fsi.2013.10.004
Nayak SK (2010) Probiotics and immunity: a fish perspective. Fish Shellfish Immunol 29(1):2–14. https://doi.org/10.1016/j.fsi.2010.02.017
Nezamdoost-Sani N, Khaledabad MA, Amiri S, Khaneghah AM (2023) Alginate and derivatives hydrogels in encapsulation of probiotic bacteria: An updated review. Food Biosci 27:102433. https://doi.org/10.1016/j.fbio.2023.102433
Pinto MG, Franz CM, Schillinger U, Holzapfel WH (2006) Lactobacillus spp. with in-vitro probiotic properties from human faeces and traditional fermented products. International journal of food microbiology 109(3):205–214. https://doi.org/10.1016/j.ijfoodmicro.2006.01.029
Rahman A, Khan KM, Al-Khaledi G, Khan I, Al-Shemary T (2012) Over activation of hippocampal serine/threonine protein phosphatases PP1 and PP2A is involved in lead-induced deficits in learning and memory in young rats. Neurotoxicology 33(3):370–383. https://doi.org/10.1016/j.neuro.2012.02.014
Sarkar A, Ray D, Shrivastava AN, Sarker S (2006) Molecular biomarkers: their significance and application in marine pollution monitoring. Ecotoxicology 15:333–340
Shi G, Zhai M, Mao G, Li M, Hu D (2014) Chemical compositions and antifungi activities of pyroligneous acids from walnut shell of two species. Acta Botanica Boreali-Occidentalia Sinica 34(10):2109–2117
Soltani M, Shenavar Masouleh A, Ahmadi M, Pourkazemi M, Taherimirghaed A (2016) Antibacterial activity, antibiotic susceptibility and probiotic use of lactic acid bacteria (LAB) in Persian sturgeon (Acipenser persicus). Sustain Aquacult Health Manag J 2(1):54–65. http://ijaah.ir/article-1-100-en.html
Song H, Yu W, Gao M, Liu X, Ma X (2013) Microencapsulated probiotics using emulsification technique coupled with internal or external gelation process. Carbohydr Polym 96(1):181–189. https://doi.org/10.1016/j.carbpol.2013.03.068
Vardy DW, Santore R, Ryan A, Giesy JP, Hecker M (2014) Acute toxicity of copper, lead, cadmium, and zinc to early life stages of white sturgeon (Acipenser transmontanus) in laboratory and Columbia River water. Environ Sci Pollut Res 21(13):8176–8187. https://doi.org/10.1007/s11356-014-2754-6
Veisi RS, Taghdir M, Abbaszadeh S, Hedayati A (2023) Dietary Effects of Probiotic Lactobacillus casei on Some Immunity Indices of Common Carp (Cyprinus carpio) Exposed to Cadmium. Biol Trace Elem Res 24:1–9
Vine NG, Leukes WD, Kaiser H (2006) Probiotics in marine larviculture. FEMS Microbiol Rev 30(3):404–427. https://doi.org/10.1111/j.1574-6976.2006.00017.x
Wangsongsak A, Utarnpongsa S, Kruatrachue M, Ponglikitmongkol M, Pokethitiyook P, Sumranwanich T (2007) Alterations of organ histopathology and metallolhionein mRNA expression in silver barb, Puntius gonionotus during subchronic cadmium exposure. J Environ Sci 19(11):1341–1348. https://doi.org/10.1016/S1001-0742(07)60219-8
Worthington DG, Westoby M, Bell JD (1991) Fish larvae settling in seagrass: effects of leaf density and an epiphytic alga. Aust J Ecol 16(3):289–293. https://doi.org/10.1111/j.1442-9993.1991.tb01056.x
Yano T, Hatayama Y, Matsuyama H, Nakao M (1988) Titration of the alternative complement pathway activity of representative cultured fishes. Nippon Suisan Gakkaishi (Japanese Edition) 54(6):1049–1054. https://doi.org/10.2331/suisan.54.1049
Yudiati E, Subagiyo S, Djarod MS (2020) Preliminary study of polysaccharide and oligosaccharide alginate (AOS) as prebiotic of probiotic bacteria. Jurnal Kelautan Tropis 23(2):234–238. https://doi.org/10.14710/jkt.v23i2.7674
Feldman BV, Zinkl JG, Jain NC, Schalm OW (2000) Schalm's veterinary hematology/editors, Bernard V. Feldman, Joseph G. Zinkl, Nemi C. Jain. Lippincott Williams & Wilkins.
Acknowledgements
This work financially supported by research council of Shahid Chamran University of Ahvaz and Centre of Excellence for Warm Water Fish Health and disease, Shahid Chamran University of Ahvaz, Ahvaz, Iran
Funding
This work was supported by Research council, Shahid Chamran University of Ahvaz and Centre of Excellence for Warm Water Fish Health and disease, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
Author information
Authors and Affiliations
Contributions
The authors of the manuscript certify that they have participated sufficiently in the work as mentioned below:
Maryam Ahmadmoradi (1st author) As a Ph.D. student was directly involved with the experiment, laboratory tests, data analyzing, and writing the article. Mojtaba Alishahi (2nd author) designed and planned the experiment and handled laboratory tests and encapsulation procedures, Siavash Soltanian (third author) performed molecular and cellular parts of the work. Ali Shahriari (4th author ), contributes to the work by assaying the intestinal enzyme activity and hematological indices and analyzing the results, and writing the manuscript. Azadeh Yektaseresht (5th author) performed bacteriological and immunological parts of the experiment.
Corresponding author
Ethics declarations
Ethics approval
In vivo phase of this experiment has been conducted as the guidelines of the Institutional Animal Ethics Committee, Faculty of Veterinary, Shahid Chamran University, Iran (Approved NO: EE/1401.2.24.78971/SCU.ac.ir).
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Brian Austin
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ahmadmoradi, M., Alishahi, M., Soltanian, S. et al. Effects of encapsulation of Lactobacillus plantarum on probiotic potential and reducing lead toxicity in rainbow trout (Oncorhynchus mykiss Walbaum). Aquacult Int 32, 337–359 (2024). https://doi.org/10.1007/s10499-023-01164-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01164-x