Abstract
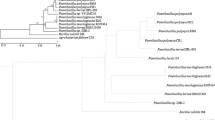
Despite being one of the most abundant elements in soil, phosphorus (P) often becomes a limiting macronutrient for plants due to its low bioavailability, primarily locked away in insoluble organic and inorganic forms. Phosphate solubilizing and mineralizing bacteria, also called phosphobacteria, isolated from P-deficient soils have emerged as a promising biofertilizer alternative, capable of converting these recalcitrant P forms into plant-available phosphates. Three such phosphobacteria strains—Serratia sp. RJAL6, Klebsiella sp. RCJ4, and Enterobacter sp. 198—previously demonstrated their particular strength as plant growth promoters for wheat, ryegrass, or avocado under abiotic stresses and P deficiency. Comparative genomic analysis of their draft genomes revealed several genes encoding key functionalities, including alkaline phosphatases, isonitrile secondary metabolites, enterobactin biosynthesis and genes associated to the production of indole-3-acetic acid (IAA) and gluconic acid. Moreover, overall genome relatedness indexes (OGRIs) revealed substantial divergence between Serratia sp. RJAL6 and its closest phylogenetic neighbours, Serratia nematodiphila and Serratia bockelmanii. This compelling evidence suggests that RJAL6 merits classification as a novel species. This in silico genomic analysis provides vital insights into the plant growth-promoting capabilities and provenance of these promising PSRB strains. Notably, it paves the way for further characterization and potential application of the newly identified Serratia species as a powerful bioinoculant in future agricultural settings.




Similar content being viewed by others
References
Aguena M, Yagil E, Spira B (2002) Transcriptional analysis of the pst operon of Escherichia coli. Mol Genet Genomics 268:518–524. https://doi.org/10.1007/s00438-002-0764-4
Auch AF, Henz SR, Holland BR, Göker M (2006) Genome BLAST distance phylogenies inferred from whole plastid and whole mitochondrion genome sequences. BMC Bioinform 7:350. https://doi.org/10.1186/1471-2105-7-350
Aziz RK, Devoid S, Disz T, Edwards RA, Henry CS, Olsen GJ et al (2012) SEED Servers: High-Performance Access to the SEED Genomes, Annotations, and Metabolic Models. PLoS ONE 7:e48053. https://doi.org/10.1371/journal.pone.0048053
Barra PJ, Inostroza NG, Acuña JJ, Mora ML, Crowley DE, Jorquera MA (2016) Formulation of bacterial consortia from avocado (Persea americana Mill.) and their effect on growth, biomass and superoxide dismutase activity of wheat seedlings under salt stress. Appl Soil Ecol 102:80–91. https://doi.org/10.1016/j.apsoil.2016.02.014
Barra PJ, Inostroza NG, Mora ML, Crowley DE, Jorquera MA (2017) Bacterial consortia inoculation mitigates the water shortage and salt stress in an avocado (Persea americana Mill.) nursery. Appl Soil Ecol 111:39–47. https://doi.org/10.1016/j.apsoil.2016.11.012
Barra PJ, Pontigo S, Delgado M, Parra-Almuna L, Duran P, Valentine AJ et al (2019) Phosphobacteria inoculation enhances the benefit of P–fertilization on Lolium perenne in soils contrasting in P–availability. Soil Biol Biochem 136:107516. https://doi.org/10.1016/j.soilbio.2019.06.012
Barra PJ, Duran P, Delgado M, Viscardi S, Claverol S, Larama G et al (2023) Proteomic response to phosphorus deficiency and aluminum stress of three aluminum-tolerant phosphobacteria isolated from acidic soils. iScience 26:107910. https://doi.org/10.1016/j.isci.2023.107910
Barra, P. J., Viscardi, S., Jorquera, M. A., Duran, P. A., Valentine, A. J., and de la Luz Mora, M. (2018). Understanding the Strategies to Overcome Phosphorus–Deficiency and Aluminum–Toxicity by Ryegrass Endophytic and Rhizosphere Phosphobacteria. Front Microbiol 9. https://doi.org/10.3389/fmicb.2018.01155.
Berriman M (2003) Viewing and annotating sequence data with Artemis. Brief Bioinform 4:124–132. https://doi.org/10.1093/bib/4.2.124
Bibb MJ, Findlay PR, Johnson MW (1984) The relationship between base composition and codon usage in bacterial genes and its use for the simple and reliable identification of protein-coding sequences. Gene 30:157–166. https://doi.org/10.1016/0378-1119(84)90116-1
Brady SF, Clardy J (2005) Cloning and Heterologous Expression of Isocyanide Biosynthetic Genes from Environmental DNA. Angew Chem Int Ed 44:7063–7065. https://doi.org/10.1002/anie.200501941
Castagno LN, Sannazzaro AI, Gonzalez ME, Pieckenstain FL, Estrella MJ (2021) Phosphobacteria as key actors to overcome phosphorus deficiency in plants. Annals of Applied Biology 178:256–267. https://doi.org/10.1111/aab.12673
Caza M, Garénaux A, Lépine F, Dozois CM (2015) Catecholate siderophore esterases Fes, IroD and IroE are required for salmochelins secretion following utilization, but only IroD contributes to virulence of extra-intestinal pathogenic Escherichia coli. Mol Microbiol 97:717–732. https://doi.org/10.1111/mmi.13059
Chen YP, Rekha PD, Arun AB, Shen FT, Lai W-A, Young CC (2006) Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl Soil Ecol 34:33–41. https://doi.org/10.1016/j.apsoil.2005.12.002
Chen T, Chen J, Tang Y, Zhou J, Guo Y, Chang W (2021) Current Understanding toward Isonitrile Group Biosynthesis and Mechanism. Chin J Chem 39:463–472. https://doi.org/10.1002/cjoc.202000448
Chenault SS, Earhart CF (1991) Organization of genes encoding membrane proteins of the Escherichia coli ferrienterobactin permease. Mol Microbiol 5:1405–1413. https://doi.org/10.1111/j.1365-2958.1991.tb00787.x
Crawford JM, Kontnik R, Clardy J (2010) Regulating Alternative Lifestyles in Entomopathogenic Bacteria. Curr Biol 20:69–74. https://doi.org/10.1016/j.cub.2009.10.059
de la Mora LL, Demanet R, Acuña JJ, Viscardi S, Jorquera M, Rengel Z et al (2017) Aluminum-tolerant bacteria improve the plant growth and phosphorus content in ryegrass grown in a volcanic soil amended with cattle dung manure. Appl Soil Ecol 115:19–26. https://doi.org/10.1016/j.apsoil.2017.03.013
Furrer JL, Sanders DN, Hook-Barnard IG, McIntosh MA (2002) Export of the siderophore enterobactin in Escherichia coli: involvement of a 43 kDa membrane exporter. Mol Microbiol 44:1225–1234. https://doi.org/10.1046/j.1365-2958.2002.02885.x
Gardner S G, and McCleary WR (2019) Control of the phoBR Regulon in Escherichia coli. EcoSal Plus 8. https://doi.org/10.1128/ecosalplus.esp-0006-2019
Glick BR (2012) Plant Growth-Promoting Bacteria: Mechanisms and Applications. Scientifica (cairo) 2012:1–15. https://doi.org/10.6064/2012/963401
Hoffmann H, Stindl S, Stumpf A, Mehlen A, Monget D, Heesemann J et al (2005) Description of Enterobacter ludwigii sp. nov., a novel Enterobacter species of clinical relevance. Syst Appl Microbiol 28:206–212. https://doi.org/10.1016/j.syapm.2004.12.009
Hu X, Boyer GL (1996) Siderophore-Mediated Aluminum Uptake by Bacillus megaterium ATCC 19213. Appl Environ Microbiol 62:4044–4048. https://doi.org/10.1128/aem.62.11.4044-4048.1996
Inostroza NG, Barra PJ, Wick LY, Mora ML, Jorquera MA (2017) Effect of rhizobacterial consortia from undisturbed arid- and agro-ecosystems on wheat growth under different conditions. Lett Appl Microbiol 64:158–163. https://doi.org/10.1111/lam.12697
Jackman SD, Vandervalk BP, Mohamadi H, Chu J, Yeo S, Hammond SA et al (2017) ABySS 2.0: resource-efficient assembly of large genomes using a Bloom filter. Genome Res 27:768–777. https://doi.org/10.1101/gr.214346.116
Kagan J, Sharon I, Beja O, Kuhn JC (2008) The tryptophan pathway genes of the Sargasso Sea metagenome: new operon structures and the prevalence of non-operon organization. Genome Biol 9:R20. https://doi.org/10.1186/gb-2008-9-1-r20
Khan AR, Park G-S, Asaf S, Hong S-J, Jung BK, Shin J-H (2017) Complete genome analysis of Serratia marcescens RSC-14: A plant growth-promoting bacterium that alleviates cadmium stress in host plants. PLoS ONE 12:e0171534. https://doi.org/10.1371/journal.pone.0171534
Klinman JP, Bonnot F (2014) Intrigues and Intricacies of the Biosynthetic Pathways for the Enzymatic Quinocofactors: PQQ, TTQ, CTQ, TPQ, and LTQ. Chem Rev 114:4343–4365. https://doi.org/10.1021/cr400475g
Kögl F, Kostermans DGFR (1934) Hetero-auxin als Stoffwechselprodukt niederer pflanzlicher Organismen. Isolierung aus Hefe. 13. Mitteilung über pflanzliche Wachstumsstoffe. Hoppe Seylers Z Physiol Chem 228:113–121. https://doi.org/10.1515/bchm2.1934.228.3-6.113
Leduc D, Battesti A, Bouveret E (2007) The Hotdog Thioesterase EntH (YbdB) Plays a Role In Vivo in Optimal Enterobactin Biosynthesis by Interacting with the ArCP Domain of EntB. J Bacteriol 189:7112–7126. https://doi.org/10.1128/JB.00755-07
Meier-Kolthoff JP, Göker M (2019) TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat Commun 10:2182. https://doi.org/10.1038/s41467-019-10210-3
Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14:60. https://doi.org/10.1186/1471-2105-14-60
Meier-Kolthoff JP, Hahnke RL, Petersen J, Scheuner C, Michael V, Fiebig A et al (2014) Complete genome sequence of DSM 30083T, the type strain (U5/41T) of Escherichia coli, and a proposal for delineating subspecies in microbial taxonomy. Stand Genomic Sci 9:2. https://doi.org/10.1186/1944-3277-9-2
Merla C, Rodrigues C, Passet V, Corbella M, Thorpe H A, Kallonen TVS et al (2019) Description of Klebsiella spallanzanii sp. nov. and of Klebsiella pasteurii sp. nov. Front Microbiol 10. https://doi.org/10.3389/fmicb.2019.02360
Morffy N, Strader LC (2020) Old Town Roads: routes of auxin biosynthesis across kingdoms. Curr Opin Plant Biol 55:21–27. https://doi.org/10.1016/j.pbi.2020.02.002
Prabhu N, Borkar S, and Garg S (2019) “Phosphate solubilization by microorganisms,” in Advances in Biological Science Research, (Elsevier), 161–176. https://doi.org/10.1016/B978-0-12-817497-5.00011-2.
Puehringer S, Metlitzky M, Schwarzenbacher R (2008) The pyrroloquinoline quinone biosynthesis pathway revisited: A structural approach. BMC Biochem 9:8. https://doi.org/10.1186/1471-2091-9-8
Rao NN, Torriani A (1990) Molecular aspects of phosphate transport in Escherichia coli. Mol Microbiol 4:1083–1090. https://doi.org/10.1111/j.1365-2958.1990.tb00682.x
Reitz ZL, Sandy M, Butler A (2017) Biosynthetic considerations of triscatechol siderophores framed on serine and threonine macrolactone scaffolds. Metallomics 9:824–839. https://doi.org/10.1039/C7MT00111H
Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci 106:19126–19131. https://doi.org/10.1073/pnas.0906412106
Rusnak F, Faraci WS, Walsh CT (1989) Subcloning, expression, and purification of the enterobactin biosynthetic enzyme 2,3-dihydroxybenzoate-AMP ligase: demonstration of enzyme-bound (2,3-dihydroxybenzoyl) adenylate product. Biochemistry 28:6827–6835. https://doi.org/10.1021/bi00443a008
Saha R, Farrance CE, Verghese B, Hong S, Donofrio RS (2013) Klebsiella michiganensis sp. nov., A new Bacterium isolated from a tooth brush holder. Curr Microbiol 66:72–78. https://doi.org/10.1007/s00284-012-0245-x
Schalk IJ, Hannauer M, Braud A (2011) New roles for bacterial siderophores in metal transport and tolerance. Environ Microbiol 13:2844–2854. https://doi.org/10.1111/j.1462-2920.2011.02556.x
Viruel E, Lucca ME, Siñeriz F (2011) Plant growth promotion traits of phosphobacteria isolated from Puna, Argentina. Arch Microbiol 193:489–496. https://doi.org/10.1007/s00203-011-0692-y
Yoon S-H, Ha S, Lim J, Kwon S, Chun J (2017a) A large-scale evaluation of algorithms to calculate average nucleotide identity. Antonie Van Leeuwenhoek 110:1281–1286. https://doi.org/10.1007/s10482-017-0844-4
Yoon S-H, Ha S-M, Kwon S, Lim J, Kim Y, Seo H et al (2017b) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Zane HK, Naka H, Rosconi F, Sandy M, Haygood MG, Butler A (2014) Biosynthesis of amphi-enterobactin siderophores by Vibrio harveyi BAA-1116: identification of a bifunctional nonribosomal peptide synthetase condensation domain. J Am Chem Soc 136:5615–5618. https://doi.org/10.1021/ja5019942
Zhang C-X, Yang S-Y, Xu M-X, Sun J, Liu H, Liu J-R et al (2009) Serratia nematodiphila sp. nov., associated symbiotically with the entomopathogenic nematode Heterorhabditidoides chongmingensis (Rhabditida: Rhabditidae). Int J Syst Evol Microbiol 59:1603–1608. https://doi.org/10.1099/ijs.0.65718-0
Acknowledgements
The authors acknowledge to Scientific and Technological Bioresource Nucleus of Universidad de La Frontera (BIOREN–UFRO) and Service Management Analytical Research and Training Center (SmartC-BIOREN). The authors also acknowledge to the supercomputing infrastructure of Soroban (SATREPS MACH—JPM/JSA1705), Centro de Modelación y Computación Científica, Universidad de La Frontera, Temuco
Funding
This research was funded by the Agencia Nacional de Investigación y Desarrollo (ANID) of Chilean government through FONDECYT initiation project No. 11200377 (P.J.B.), FONDECYT Postdoctorado project No. 3220546 (C.P-N); FONDECYT Regular Projects No. 1241293 (P.J.B.); 1210684 (M.D.); 1201196 (P.D.) and 1230084 (M.d.l.L.M.). This study was also funding by Proyecto REDES ETAPA INICIAL, Convocatoria 2017, REDI170334 (P.J.B.), by Concurso Anillos de Investigación en Áreas Temáticas, ANID ATE220038 (P.D., M.D., P.J.B.) and by Universidad de La Frontera (DiUFRO), Proyectos de Investigación Vinculados a la Red Nexer No. DNX22-0009 (P.J.B.) and DNX22-0005 (P.D.).
Author information
Authors and Affiliations
Contributions
Conceptualization, P.J.B., M.D. and P.D.; methodology, P.J.B., C.C-A. and G.L.; validation, P.J.B., M.D., M.d.l.L.M. and P.D; formal analysis, C.C-A., C.P-N. and P.J.B; investigation, C.C-A. and P.J.B.,; resources, P.J.B., M.D. and P.D.; data curation, P.J.B., C.C-A. and G.L.; writing—original draft preparation, C.C. and P.J.B.; writing—review and editing, P.J.B., M.D., C.P-N., M.d.l.L.M. and P.D.; visualization, P.J.B., C.C-A. and G.L.; supervision, P.J.B.; project administration, P.J.B.; funding acquisition, P.J.B., M.D. and P.D. All authors have read and agreed to the published version of the manuscript
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
It is the original work of the authors. The work described has not been submitted elsewhere for publication, in whole or in part, and all authors listed carry out the data analysis and manuscript writing.
Conflict of interest
The authors declare that they have no direct or indirect conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cortés-Albayay, C., Delgado-Torres, M., Larama, G. et al. Comparative genomics of plant growth promoting phosphobacteria isolated from acidic soils. Antonie van Leeuwenhoek 117, 76 (2024). https://doi.org/10.1007/s10482-024-01961-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10482-024-01961-1