Abstract
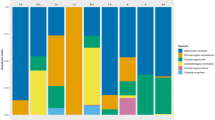
Tick-borne diseases are a major epidemiological problem worldwide. The aim of this study was to investigate the bacterial composition of saliva obtained from engorged adult Rhipicephalus microplus females. Saliva samples collected from partially or fully engorged adult female ticks were analysed using an ultra-high-throughput Illumina HiSeq 2500 sequencing system. To elucidate the possible routes of bacterial transmission, the bacterial flora from whole ticks were also investigated. Proteobacteria, Firmicutes, and Actinobacteria were the predominant phyla in all samples, and Acinetobacter, Rickettsia, Escherichia and Coxiella were the major genera. Microbial diversity in saliva samples from partially engorged ticks was more complex than that of samples from fully engorged individuals. The comparison of saliva and whole-tick samples suggests that bacteria in saliva also colonize the tick’s body. We believe that some bacterial genera, such as Dermacoccus, Achromia, SMB53, Sutterella, Providencia, Mycoplana, Oscillospira, and Agrobacterium, were found and reported in ticks for the first time. The Coxiella and Rickettsia detected in this study might be tick-borne pathogens, suggesting health risks associated with exposure to R. microplus in humans and animals. These findings may serve as the basis for developing strategies to control ticks and tick-borne diseases.




Similar content being viewed by others
References
Amann RI, Ludwig W, Schleifer KH (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Anderson JF, Magnarelli LA (2008) Biology of ticks. Infect Dis Clin North Am 22:195–215. doi:10.1016/j.idc.2007.12.006
Andreotti R, Pérez de León AA, Dowd SE, Guerrero FD, Bendele KG, Scoles GA (2011) Assessment of bacterial diversity in the cattle tick Rhipicephalus (boophilus) microplus through tag-encoded pyrosequencing. BMC Microbiol 11:6. doi:10.1186/1471-2180-11-6
Basso AP, Martins PD, Nachtigall G, Sand SV, Moura TM, Frazzon APG (2014) Antibiotic resistance and enterotoxin genes in Staphylococcus sp. isolates from polluted water in Southern Brazil. Anais da Academia Brasileira de Ciências 86:1813–1820. doi:10.1590/0001-3765201420130512
Bergogne-Bérézin E (1994) Acinetobacter spp. Saprophytic organisms of increasing pathogenic importance. Zentralbl Bakteriol 281:389–405. doi:10.1016/S0934-8840(11)80326-9
Budachetri K, Browning RE, Adamson SW, Dowd SE, Chao CC, Ching WM, Karim S (2014) An insight into the microbiome of the Amblyomma maculatum (acari: Ixodidae). J Med Entomol 51:119–129. doi:10.1603/ME12223
Choi E, Pyzocha NJ, Maurer DM (2016) Tick-borne illnesses. Curr Sports Med Rep 15:98–104. doi:10.1249/JSR.0000000000000238
De Silva AM, Fikrig E (1995) Growth and migration of Borrelia burgdorferi in ixodes ticks during blood feeding. Am J Trop Med Hyg 53:397–404
Frischknecht F (2007) The skin as interface in the transmission of arthropod-borne pathogens. Cell Microbiol 9:1630–1640. doi:10.1111/j.1462-5822.2007.00955.x
Halajian A, Palomar AM, Portillo A, Heyne H, Luus-Powell WJ, Oteo JA (2016) Investigation of rickettsia, Coxiella burnetii and bartonella in ticks from animals in South Africa. Ticks Tick-Borne Dis 7:361–366. doi:10.1016/j.ttbdis.2015.12.008
Heise SR, Elshahed MS, Little SE (2010) Bacterial diversity in Amblyomma americanum (acari: Ixodidae) with a focus on members of the genus Rickettsia. J Med Entomol 47:258–268. doi:10.1093/jmedent/47.2.258
Howell JM, Ueti MW, Palmer GH, Scoles GA, Knowles DP (2007) Persistently infected calves as reservoirs for acquisition and transovarial transmission of Babesia bovis by Rhipicephalus (boophilus) microplus. J Clin Microbiol 45:3155–3159. doi:10.1128/JCM.00766-07
Jongejan F, Uilenberg G (2004) The global importance of ticks. Parasitology 129(Suppl 1):S3–S14. doi:10.1017/S0031182004005967
Kazimírová M, Štibrániová I (2013) Tick salivary compounds: their role in modulation of host defences and pathogen transmission. Front Cell Infect Microbiol 3:43. doi:10.3389/fcimb.2013.00043
Klubal R, Kopecky J, Nesvorna M, Sparagano OA, Thomayerova J, Hubert J (2016) Prevalence of pathogenic bacteria in Ixodes ricinus ticks in Central Bohemia. Exp Appl Acarol 68:127–137. doi:10.1007/s10493-015-9988-y
Kocan KM, Stiller D, Goff WL, Claypool PL, Edwards W, Ewing SA, McGuire TC, Hair JA, Barron SJ (1992) Development of Anaplasma marginale in male Dermacentor andersoni transferred from parasitemic to susceptible cattle. Am J Vet Res 53:499–507
Kovaleva J, Degener JE, van der Mei HC (2014) Methylobacterium and its role in health care-associated infection. J Clin Microbiol 52:1317–1321. doi:10.1128/JCM.03561-13
Kurtti TJ, Simser JA, Baldridge GD, Palmer AT, Munderloh UG (2005) Factors influencing in vitro infectivity and growth of Rickettsia peacockii (Rickettsiales: Rickettsiaceae), an endosymbiont of the Rocky Mountain wood tick, dermacentor andersoni (acari, Ixodidae). J Invertebr Pathol 90:177–186. doi:10.1016/j.jip.2005.09.001
Lalzar I, Friedmann Y, Gottlieb Y (2014) Tissue tropism and vertical transmission of coxiella in Rhipicephalus sanguineus and Rhipicephalus turanicus ticks. Environ Microbiol 16:3657–3668. doi:10.1111/1462-2920.12455
Liu C, Pan F, Guo J, Yan W, Jin Y, Liu C, Qin L, Fang X (2016) Hospital acquired pneumonia due to Achromobacter spp. in a geriatric ward in China: clinical characteristic, genome variability, biofilm production, antibiotic resistance and integron in isolated strains. Front Microbiol 7:621
Mayo B, Rachid CT, Alegría A, Leite AM, Peixoto RS, Delgado S (2014) Impact of next generation sequencing techniques in food microbiology. Curr Genom 15:293–309. doi:10.2174/1389202915666140616233211
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nielsen MW, Strube ML, Isbrand A, Al-Medrasi WD, Boye M, Jensen TK, Klitgaard K (2016) Potential bacterial core species associated with digital dermatitis in cattle herds identified by molecular profiling of interdigital skin samples. Vet Microbiol 186:139–149. doi:10.1016/j.vetmic.2016.03.003
Parola P, Paddock CD, Socolovschi C, Labruna MB, Mediannikov O, Kernif T, Abdad MY, Stenos J, Bitam I, Fournier PE, Raoult D (2013) Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev 26:657–702. doi:10.1128/CMR.00032-13
Patton TG, Dietrich G, Brandt K, Dolan MC, Piesman J, Gilmore RD Jr (2012) Saliva, salivary gland, and hemolymph collection from ixodes scapularis ticks. J Vis Exp 60:e3894. doi:10.3791/3894
Podschun R, Ullmann U (1998) Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev 11:589–603
Qiu Y, Nakao R, Ohnuma A, Kawamori F, Sugimoto C (2014) Microbial population analysis of the salivary glands of ticks; a possible strategy for the surveillance of bacterial pathogens. PLoS ONE 9:e103961. doi:10.1371/journal.pone.0103961
Reif KE, Palmer GH, Ueti MW, Scoles GA, Margolis JJ, Monack DM, Noh SM (2011) Dermacentor andersoni transmission of Francisella tularensis subsp. novicida reflects bacterial colonization, dissemination, and replication coordinated with tick feeding. Infect Immun 79:4941–4946. doi:10.1128/IAI.05676-11
Roy C, Roque JL, François PM, Ferrieres A, Raboisson D (2012) Investigation of the aetiology of udder-thigh dermatitis in French dairy cattle. Vet J 193:274–276. doi:10.1016/j.tvjl.2011.09.023
Socolovschi C, Mediannikov O, Raoult D, Parola P (2009) The relationship between spotted fever group rickettsiae and ixodid ticks. Vet Res 40:34. doi:10.1051/vetres/2009017
Wielinga PR, Gaasenbeek C, Fonville M, de Boer A, de Vries A, Dimmers W, Akkerhuis Op Jagers G, Schouls LM, Borgsteede F, van der Giessen JW (2006) Longitudinal analysis of tick densities and Borrelia, Anaplasma, and Ehrlichia infections of Ixodes ricinus ticks in different habitat areas in The Netherlands. Appl Environ Microbiol 72:7594–7601. doi:10.1128/AEM.01851-06
Wikel SK (1999) Tick modulation of host immunity: an important factor in pathogen transmission. Int J Parasitol 29:851–859. doi:10.1016/S0020-7519(99)00042-9
Woodward WD, Besser TE, Ward AC, Corbeil LB (1987) In vitro growth inhibition of mastitis pathogens by bovine teat skin normal flora. Can J Vet Res 51:27–31
Xu XL, Cheng TY, Yang H, Yan F (2015) Identification of intestinal bacterial flora in Rhipicephalus microplus ticks by conventional methods and PCR-DGGE analysis. Exp Appl Acarol 66:257–268. doi:10.1007/s10493-015-9896-1
Yano T, Moe KK, Yamazaki K, Ooka T, Hayashi T, Misawa N (2010) Identification of candidate pathogens of papillomatous digital dermatitis in dairy cattle from quantitative 16S rRNA clonal analysis. Vet Microbiol 143:352–362. doi:10.1016/j.vetmic.2009.12.009
Zhang XC, Yang ZN, Lu B, Ma XF, Zhang CX, Xu HJ (2014) The composition and transmission of microbiome in hard tick, Ixodes persulcatus, during blood meal. Ticks Tick-Borne Dis 5:864–870. doi:10.1016/j.ttbdis.2014.07.009
Zinicola M, Higgins H, Lima S, Machado V, Guard C, Bicalho R (2015a) Shotgun metagenomic sequencing reveals functional genes and microbiome associated with bovine digital dermatitis. PLoS ONE 10:e0133674. doi:10.1371/journal.pone.0133674
Zinicola M, Lima F, Lima S, Machado V, Gomez M, Döpfer D, Guard C, Bicalho R (2015b) Altered microbiomes in bovine digital dermatitis lesions, and the gut as a pathogen reservoir. PLoS ONE 10:e0120504. doi:10.1371/journal.pone.0120504
Acknowledgments
We thank Huan Tan and Yu Zhao for their help in sampling. Liangliang Xiang and Błażej Poźniak contributed equally to this study. This research was financially supported by a grant from the National Natural Science Foundation of China (No. 31372431).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Xiang, L., Poźniak, B. & Cheng, TY. Bacteriological analysis of saliva from partially or fully engorged female adult Rhipicephalus microplus by next-generation sequencing. Antonie van Leeuwenhoek 110, 105–113 (2017). https://doi.org/10.1007/s10482-016-0780-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-016-0780-8