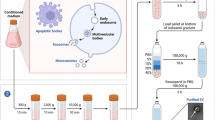
Abstract
Biomaterials that recapitulate the native in vivo microenvironment are promising to facilitate tissue repair and regeneration when used in combination with relevant growth factors (GFs), chemokines, cytokines, and other small molecules and cell sources. However, limitations with the use of exogenous factors and ex vivo cell expansion has prompted cell-/GF-free tissue engineering strategies. Additionally, conventional chemotaxis assays for studying cell migration behavior provide limited information, lack long-term stability, and fail to recapitulate physiologically relevant conditions. In this study, articular cartilage tissue-based biomaterials were developed via a rapid tissue decellularization protocol. The decellularized tissue was further processed into a hydrogel through solubilization and self-assembly. Chemotactic activity of the tissue-derived gel was investigated using sophisticated cellular migration assays. These tissue-derived extracellular matrix (ECM) biomaterials retain biochemical cues of native tissue and stimulate the chemotactic migration of hBMSCs in 2D and 3D cell migration models using a real-time chemotaxis assay. This strategy, in a way, developed a new paradigm in tissue engineering where cartilage tissue repair and regeneration can be approached with decellularized cartilage tissue in the place of an engineered matrix. This strategy can be further expanded for other tissue-based ECMs to develop cell-/GF-free tissue engineering and regenerative medicine strategies for recruiting endogenous cell populations to facilitate tissue repair and regeneration.






Similar content being viewed by others
References
Amini, A. R., C. T. Laurencin, and S. P. Nukavarapu. Bone tissue engineering: recent advances and challenges. Crit. Rev. Biomed. Eng. 40(5):363–408, 2012.
Amini, A., and S. Nukavarapu. Oxygen-tension controlled matrices for enhanced osteogenic cell survival and performance. Ann. Biomed. Eng. 42(6):1261–1270, 2014. https://doi.org/10.1007/s10439-014-0990-z.
Blum, J. C., T. L. Schenck, A. Birt, R. E. Giunta, and P. S. Wiggenhauser. Artificial decellularized extracellular matrix improves the regenerative capacity of adipose tissue derived stem cells on 3D printed polycaprolactone scaffolds. J. Tissue Eng. 12:20417314211022240, 2021. https://doi.org/10.1177/20417314211022242.
Brown, C., X. Pan, and A. Hassid. Nitric oxide and C-type atrial natriuretic peptide stimulate primary aortic smooth muscle cell migration via a cGMP-dependent mechanism. Circ. Res. 84(6):655–667, 1999. https://doi.org/10.1161/01.RES.84.6.655.
Bzymek, R., et al. Real-time two- and three-dimensional imaging of monocyte motility and navigation on planar surfaces and in collagen matrices: roles of Rho. Sci. Rep. 6(1):1, 2016. https://doi.org/10.1038/srep25016.
Caralt, M., et al. Optimization and critical evaluation of decellularization strategies to develop renal extracellular matrix scaffolds as biological templates for organ engineering and transplantation. Am. J. Transpl. 15(1):64–75, 2015. https://doi.org/10.1111/ajt.12999.
Crapo, P. M., T. W. Gilbert, and S. F. Badylak. An overview of tissue and whole organ decellularization processes. Biomaterials. 32(12):3233–3243, 2011. https://doi.org/10.1016/j.biomaterials.2011.01.057.
Dorcemus, D. L., E. O. George, C. N. Dealy, and S. P. Nukavarapu. Harnessing external cues: development and evaluation of an in vitro culture system for osteochondral tissue engineering. Tissue Eng. Part A. 23(15–16):719–737, 2017. https://doi.org/10.1089/ten.tea.2016.0439.
Dorcemus, D. L., H. S. Kim, and S. P. Nukavarapu. Gradient scaffold with spatial growth factor profile for osteochondral interface engineering. Biomed. Mater. Bristol Engl. 2020. https://doi.org/10.1088/1748-605X/abd1ba.
Dormann, D., and C. J. Weijer. Chemotactic cell movement during development. Curr. Opin. Genet. Dev. 13(4):358–364, 2003. https://doi.org/10.1016/S0959-437X(03)00087-X.
Elgamoudi, B. A., and V. Korolik. A review of the advantages, disadvantages and limitations of chemotaxis assays for Campylobacter spp. Int. J. Mol. Sci. 23(3):1576, 2022. https://doi.org/10.3390/ijms23031576.
Evans, C. H., et al. Facilitated endogenous repair: making tissue engineering simple, practical, and economical. Tissue Eng. 13(8):1987–1993, 2007. https://doi.org/10.1089/ten.2006.0302.
Fernández-Pérez, J., and M. Ahearne. The impact of decellularization methods on extracellular matrix derived hydrogels. Sci. Rep. 9(1):1, 2019. https://doi.org/10.1038/s41598-019-49575-2.
Francois, E., D. Dorcemus, and S. Nukavarapu. 1-Biomaterials and scaffolds for musculoskeletal tissue engineering. In: Regenerative Engineering of Musculoskeletal Tissues and Interfaces, edited by S. P. Nukavarapu, J. W. Freeman, and C. T. Laurencin. Woodhead Publishing: Sawston, 2015, pp. 3–23.
Freytes, D. O., J. Martin, S. S. Velankar, A. S. Lee, and S. F. Badylak. Preparation and rheological characterization of a gel form of the porcine urinary bladder matrix. Biomaterials. 29(11):1630–1637, 2008. https://doi.org/10.1016/j.biomaterials.2007.12.014.
Gilpin, A., and Y. Yang. Decellularization strategies for regenerative medicine: from processing techniques to applications. BioMed Res. Int. 2017. https://doi.org/10.1155/2017/9831534.
Golebiowska, A. A., and S. P. Nukavarapu. Bio-inspired zonal-structured matrices for bone-cartilage interface engineering. Biofabrication. 14(2):025016, 2022. https://doi.org/10.1088/1758-5090/ac5413.
Herzmann, N., A. Salamon, T. Fiedler, and K. Peters. Analysis of migration rate and chemotaxis of human adipose-derived mesenchymal stem cells in response to LPS and LTA in vitro. Exp. Cell Res. 342(2):95–103, 2016. https://doi.org/10.1016/j.yexcr.2016.03.016.
Igwe, J. C., P. E. Mikael, and S. P. Nukavarapu. Design, fabrication and in vitro evaluation of a novel polymer-hydrogel hybrid scaffold for bone tissue engineering. J. Tissue Eng. Regen. Med. 8(2):131–142, 2014. https://doi.org/10.1002/term.1506.
Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface. 3(10):589–601, 2006. https://doi.org/10.1098/rsif.2006.0124.
Ivanov, A. A., A. V. Kuznetsova, O. P. Popova, T. I. Danilova, and O. O. Yanushevich. Modern approaches to acellular therapy in bone and dental regeneration. Int. J. Mol. Sci. 22(24):13454, 2021. https://doi.org/10.3390/ijms222413454.
Jin, W., et al. Modelling of the SDF-1/CXCR4 regulated in vivo homing of therapeutic mesenchymal stem/stromal cells in mice. PeerJ. 6:e6072, 2018. https://doi.org/10.7717/peerj.6072.
Kamimura, Y., H. Cai, and P. N. Devreotes. 6-TORC2 and chemotaxis in Dictyostelium discoideum. In: The Enzymes, edited by F. Tamanoi, and M. N. Hall. Academic Press: NewYork, 2010, pp. 125–142.
Karkanitsa, M., P. Fathi, T. Ngo, and K. Sadtler. Mobilizing endogenous repair through understanding immune reaction with biomaterials. Front. Bioeng. Biotechnol. 9:730938, 2021. https://doi.org/10.3389/fbioe.2021.730938.
Kim, Y., H. Ko, I. K. Kwon, and K. Shin. Extracellular matrix revisited: roles in tissue engineering. Int. Neurourol. J. 20(Suppl 1):S23-29, 2016. https://doi.org/10.5213/inj.1632600.318.
Kim, H. S., S. G. Kumbar, and S. P. Nukavarapu. Biomaterial-directed cell behavior for tissue engineering. Curr. Opin. Biomed. Eng. 17:100260, 2021. https://doi.org/10.1016/j.cobme.2020.100260.
Kim, H. S., S. G. Kumbar, and S. P. Nukavarapu. Amorphous silica fiber matrix biomaterials: an analysis of material synthesis and characterization for tissue engineering. Bioact. Mater. 19:155–166, 2023. https://doi.org/10.1016/j.bioactmat.2022.04.002.
Kim, Y. S., M. Majid, A. J. Melchiorri, and A. G. Mikos. Applications of decellularized extracellular matrix in bone and cartilage tissue engineering. Bioeng. Transl. Med. 4(1):83–95, 2019. https://doi.org/10.1002/btm2.10110.
Kowalczewski, C. J., and J. M. Saul. Biomaterials for the delivery of growth factors and other therapeutic agents in tissue engineering approaches to bone regeneration. Front. Pharmacol. 2018. https://doi.org/10.3389/fphar.2018.00513.
Kusindarta, D. L., and H. Wihadmadyatami. The role of extracellular matrix in tissue regeneration. Tissue Regen. 2018. https://doi.org/10.5772/intechopen.75728.
Langer, R., and J. P. Vacanti. Tissue engineering. Science. 260(5110):920–926, 1993.
Larouche, J., S. Sheoran, K. Maruyama, and M. M. Martino. Immune regulation of skin wound healing: mechanisms and novel therapeutic targets. Adv. Wound Care. 7(7):209–231, 2018. https://doi.org/10.1089/wound.2017.0761.
Liu, H., et al. The role of SDF-1-CXCR4/CXCR7 axis in the therapeutic effects of hypoxia-preconditioned mesenchymal stem cells for renal ischemia/reperfusion injury. PLoS ONE. 7(4):e34608, 2012. https://doi.org/10.1371/journal.pone.0034608.
Luo, Z., et al. Comparison of various reagents for preparing a decellularized porcine cartilage scaffold. Am. J. Transl. Res. 11(3):1417–1427, 2019.
Marquez-Curtis, L. A., and A. Janowska-Wieczorek. Enhancing the migration ability of mesenchymal stromal cells by targeting the SDF-1/CXCR4 axis. BioMed Res. Int. 13:561098, 2013. https://doi.org/10.1155/2013/561098.
Mikael, P. E., A. A. Golebiowska, S. G. Kumbar, and S. P. Nukavarapu. Evaluation of autologously derived biomaterials and stem cells for bone tissue engineering. Tissue Eng. Part A. 26(19–20):1052–1063, 2020. https://doi.org/10.1089/ten.tea.2020.0011.
Mikael, P. E., H. S. Kim, and S. P. Nukavarapu. Hybrid extracellular matrix design for cartilage-mediated bone regeneration. J. Biomed. Mater. Res. B. 106(1):300–309, 2018. https://doi.org/10.1002/jbm.b.33842.
Moghadasi, S., et al. A paradigm shift in cell-free approach: the emerging role of MSCs-derived exosomes in regenerative medicine. J. Transl. Med. 19(1):302, 2021. https://doi.org/10.1186/s12967-021-02980-6.
Moroni, F., and T. Mirabella. Decellularized matrices for cardiovascular tissue engineering. Am. J. Stem Cells. 3(1):1–20, 2014.
Muinonen-Martin, A. J., D. M. Veltman, G. Kalna, and R. H. Insall. An improved chamber for direct visualisation of chemotaxis. PLoS ONE. 5(12):e15309, 2010. https://doi.org/10.1371/journal.pone.0015309.
Naderi, H., M. M. Matin, and A. R. Bahrami. Review paper: Critical issues in tissue engineering: biomaterials, cell sources, angiogenesis, and drug delivery systems. J. Biomater. Appl. 26(4):383–417, 2011. https://doi.org/10.1177/0885328211408946.
Nukavarapu, S. P., J. W. Freeman, and C. T. Laurencin (eds.). Related Titles. Regenerative Engineering of Musculoskeletal Tissues and Interfaces. Sawston: Woodhead Publishing, 2015. https://doi.org/10.1016/B978-1-78242-301-0.09001-7.
Oliveira, É. R., et al. Advances in growth factor delivery for bone tissue engineering. Int. J. Mol. Sci. 22(2):903, 2021. https://doi.org/10.3390/ijms22020903.
Park, J. S., et al. Synthetic control of mammalian-cell motility by engineering chemotaxis to an orthogonal bioinert chemical signal. Proc. Natl. Acad. Sci. USA. 111(16):5896–5901, 2014. https://doi.org/10.1073/pnas.1402087111.
Park, S., H. Jang, B. S. Kim, C. Hwang, G. S. Jeong, and Y. Park. Directional migration of mesenchymal stem cells under an SDF-1α gradient on a microfluidic device. PLoS ONE. 12(9):e0184595, 2017. https://doi.org/10.1371/journal.pone.0184595.
Pati, F., et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat. Commun. 5:3935, 2014. https://doi.org/10.1038/ncomms4935.
Rodrigues, M., N. Kosaric, C. A. Bonham, and G. C. Gurtner. Wound healing: a cellular perspective. Physiol. Rev. 99(1):665–706, 2019. https://doi.org/10.1152/physrev.00067.2017.
Saldin, L. T., M. C. Cramer, S. S. Velankar, L. J. White, and S. F. Badylak. Extracellular matrix hydrogels from decellularized tissues: structure and function. Acta Biomater. 49:1–15, 2017. https://doi.org/10.1016/j.actbio.2016.11.068.
Shupe, T., C. Zimmerman, and B. Petersen. Growth factor retention on decellularized rat liver matrices derived from normal and regenerating liver. FASEB J. 26:274.10-274.10, 2012. https://doi.org/10.1096/fasebj.26.1_supplement.274.10.
Solez, K., et al. The bridge between transplantation and regenerative medicine: beginning a new Banff classification of tissue engineering pathology. Am. J. Transpl. 18(2):321–327, 2018. https://doi.org/10.1111/ajt.14610.
Varanko, A., S. Saha, and A. Chilkoti. Recent trends in protein and peptide-based biomaterials for advanced drug delivery. Adv. Drug Deliv. Rev. 156:133, 2020. https://doi.org/10.1016/j.addr.2020.08.008.
Vargason, A. M., A. C. Anselmo, and S. Mitragotri. The evolution of commercial drug delivery technologies. Nat. Biomed. Eng. 5(9):9, 2021. https://doi.org/10.1038/s41551-021-00698-w.
Vasaturo, A., S. Caserta, I. Russo, V. Preziosi, C. Ciacci, and S. Guido. A Novel chemotaxis assay in 3-D collagen gels by time-lapse microscopy. PLoS ONE. 7(12):e52251, 2012. https://doi.org/10.1371/journal.pone.0052251.
Wang, X., G. Wang, S. Zingales, and B. Zhao. Biomaterials enabled cell-free strategies for endogenous bone regeneration. Tissue Eng. Part B. 24(6):463–481, 2018. https://doi.org/10.1089/ten.teb.2018.0012.
Yang, Z., et al. Fabrication and repair of cartilage defects with a novel acellular cartilage matrix scaffold. Tissue Eng. Part C. 16(5):865–876, 2009. https://doi.org/10.1089/ten.tec.2009.0444.
Yin, H., et al. Functional tissue-engineered microtissue derived from cartilage extracellular matrix for articular cartilage regeneration. Acta Biomater. 77:127–141, 2018. https://doi.org/10.1016/j.actbio.2018.07.031.
Zakrzewski, J. L., M. R. M. van den Brink, and J. A. Hubbell. Overcoming immunological barriers in regenerative medicine. Nat. Biotechnol. 32(8):786–794, 2014. https://doi.org/10.1038/nbt.2960.
Zheng, X.-B., et al. Bone marrow-derived CXCR4-overexpressing MSCs display increased homing to intestine and ameliorate colitis-associated tumorigenesis in mice. Gastroenterol. Rep. 7(2):127–138, 2019. https://doi.org/10.1093/gastro/goy017.
Acknowledgments
The authors acknowledge support from the National Institute of Biomedical Imaging and Bioengineering (NIBIB) of the National Institutes of Health (Grant Nos. #R01EB030060 & #R01EB020640). Dr. Nukavarapu also acknowledges funding from NSF EFMA (Grant No. #1908454). The authors thank Dr. Hargis for critically reading the manuscript.
Conflict of interest
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Emmanuel Opara oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Golebiowska, A.A., Jala, V.R. & Nukavarapu, S.P. Decellularized Tissue-Induced Cellular Recruitment for Tissue Engineering and Regenerative Medicine. Ann Biomed Eng (2023). https://doi.org/10.1007/s10439-023-03182-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10439-023-03182-5