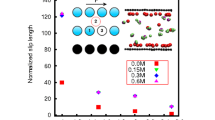
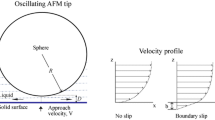
Abstract
Understanding the slip behaviors on the graphene surfaces is crucial in the field of nanofluidics and nanofluids. The reported values of the slip length in the literature from both experimental measurements and simulations are quite scattered. The presence of low concentrations of functional groups may have a greater impact on the flow behavior than expected. Using non-equilibrium molecular dynamics simulations, we specifically investigated the influence of hydroxyl-functionalized graphene surfaces on the boundary slip, particularly the effects related to hydrogen bond dynamics. We observed that hydroxyl groups significantly hindered the sliding motion of neighboring water molecules. Hydrogen bonds can be found between hydroxyl groups and water molecules. During the flow process, these hydrogen bonds continuously form and break, resulting in the energy dissipation. We analyzed the energy balance under different driving forces and proposed a theoretical model to describe the slip length which also considers the influence of hydrogen bond dynamics. The effects of the driving force and the surface functional group concentration were also studied.











Similar content being viewed by others
Data availability
Data sets generated during the current study are available from the corresponding author on reasonable request.
References
Abascal JLF, Vega C (2005) A general purpose model for the condensed phases of water: TIP4P/2005. J Chem Phys 123:234505. https://doi.org/10.1063/1.2121687
Argyris D, Tummala NR, Striolo A, Cole DR (2008) Molecular structure and dynamics in thin water films at the silica and graphite surfaces. J Phys Chem C 112:13587–13599. https://doi.org/10.1021/jp803234a
Berendsen HJC, Grigera JR, Straatsma TP (1987) The missing term in effective pair potentials. J Phys Chem 91:6269–6271. https://doi.org/10.1021/j100308a038
Bocquet L, Barrat J-L (2007) Flow boundary conditions from nano- to micro-scales. Soft Matter 3:685–693. https://doi.org/10.1039/B616490K
Bocquet L, Barrat J-L (2013) On the Green–Kubo relationship for the liquid–solid friction coefficient. J Chem Phys 139:044704. https://doi.org/10.1063/1.4816006
Bocquet L, Charlaix E (2010) Nanofluidics, from bulk to interfaces. Chem Soc Rev 39:1073–1095. https://doi.org/10.1039/B909366B
Chen B, Jiang H, Liu X, Hu X (2017) Observation and analysis of water transport through graphene oxide interlamination. J Phys Chem C 121:1321–1328. https://doi.org/10.1021/acs.jpcc.6b09753
Eijkel JCT, van den Berg A (2005) Nanofluidics: What is it and what can we expect from it? Microfluid Nanofluidics 1:249–267. https://doi.org/10.1007/s10404-004-0012-9
Erickson K, Erni R, Lee Z et al (2010) Determination of the local chemical structure of graphene oxide and reduced graphene oxide. Adv Mater 22:4467–4472. https://doi.org/10.1002/adma.201000732
Eslami H, Heydari N (2014) Hydrogen bonding in water nanoconfined between graphene surfaces: a molecular dynamics simulation study. J Nanoparticle Res 16:1–10. https://doi.org/10.1007/s11051-013-2154-8
Eslami H, Müller-Plathe F (2010) Viscosity of nanoconfined polyamide-6,6 oligomers: atomistic reverse nonequilibrium molecular dynamics simulation. J Phys Chem B 114:387–395. https://doi.org/10.1021/jp908659w
Eslami H, Rahimi M, Müller-Plathe F (2013) Molecular dynamics simulation of a silica nanoparticle in oligomeric poly(methyl methacrylate): a model system for studying the interphase thickness in a polymer–nanocomposite via different properties. Macromolecules 46:8680–8692. https://doi.org/10.1021/ma401443v
Falk K, Sedlmeier F, Joly L et al (2010) Molecular origin of fast water transport in carbon nanotube membranes: Superlubricity versus curvature dependent friction. Nano Lett 10:4067–4073. https://doi.org/10.1021/nl1021046
Fang C, Wu X, Yang F, Qiao R (2018) Flow of quasi-two dimensional water in graphene channels. J Chem Phys 148:064702. https://doi.org/10.1063/1.5017491
Figoli A, Hoinkis J, Altinkaya SA, Bundschuh J (2017) Application of nanotechnology in membranes for water treatment. CRC Press, London
González MA, Abascal JLF (2010) The shear viscosity of rigid water models. J Chem Phys 132:096101. https://doi.org/10.1063/1.3330544
Grosjean B, Pean C, Siria A et al (2016) Chemisorption of hydroxide on 2D materials from DFT calculations: graphene versus hexagonal boron nitride. J Phys Chem Lett 7:4695–4700. https://doi.org/10.1021/acs.jpclett.6b02248
Guo S, Meshot ER, Kuykendall T et al (2015) Nanofluidic transport through isolated carbon nanotube channels: advances, controversies, and challenges. Adv Mater 27:5726–5737. https://doi.org/10.1002/adma.201500372
Guo S, Garaj S, Bianco A, Ménard-Moyon C (2022) Controlling covalent chemistry on graphene oxide. Nat Rev Phys 4:247–262. https://doi.org/10.1038/s42254-022-00422-w
Ho TA, Striolo A (2014) Molecular dynamics simulation of the graphene–water interface: comparing water models. Mol Simul 40:1190–1200. https://doi.org/10.1080/08927022.2013.854893
Hummer G, Rasaiah JC, Noworyta JP (2001) Water conduction through the hydrophobic channel of a carbon nanotube. Nature 414:188–190. https://doi.org/10.1038/35102535
Irving JH, Kirkwood JG (2004) The statistical mechanical theory of transport processes. IV. The equations of hydrodynamics. J Chem Phys 18:817–829. https://doi.org/10.1063/1.1747782
Joly L, Tocci G, Merabia S, Michaelides A (2016) Strong coupling between nanofluidic transport and interfacial chemistry: how defect reactivity controls liquid–solid friction through hydrogen bonding. J Phys Chem Lett 7:1381–1386. https://doi.org/10.1021/acs.jpclett.6b00280
Jorgensen WL, Maxwell DS, Tirado-Rives J (1996) Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J Am Chem Soc 118:11225–11236. https://doi.org/10.1021/ja9621760
Kalluri RK, Ho TA, Biener J et al (2013) Partition and structure of aqueous NaCl and CaCl2 electrolytes in carbon-slit electrodes. J Phys Chem C 117:13609–13619. https://doi.org/10.1021/jp4002127
Kannam SK, Todd BD, Hansen JS, Daivis PJ (2013) How fast does water flow in carbon nanotubes? J Chem Phys 138:094701. https://doi.org/10.1063/1.4793396
Kavokine N, Netz RR, Bocquet L (2021) Fluids at the nanoscale: from continuum to subcontinuum transport. Annu Rev Fluid Mech 53:377–410. https://doi.org/10.1146/annurev-fluid-071320-095958
Li J, Zhu Y, Xia J et al (2021) Anomalously low friction of confined monolayer water with a quadrilateral structure. J Chem Phys 154:224508. https://doi.org/10.1063/5.0053361
Luzar A, Chandler D (1996a) Effect of environment on hydrogen bond dynamics in liquid water. Phys Rev Lett 76:928–931. https://doi.org/10.1103/PhysRevLett.76.928
Luzar A, Chandler D (1996b) Hydrogen-bond kinetics in liquid water. Nature 379:55–57. https://doi.org/10.1038/379055a0
Majumder M, Chopra N, Hinds BJ (2011) Mass transport through carbon nanotube membranes in three different regimes: ionic diffusion and gas and liquid flow. ACS Nano 5:3867–3877. https://doi.org/10.1021/nn200222g
Mark P, Nilsson L (2001) Structure and dynamics of the TIP3P, SPC, and SPC/E water models at 298 K. J Phys Chem A 105:9954–9960. https://doi.org/10.1021/jp003020w
Martini A, Roxin A, Snurr RQ et al (2008) Molecular mechanisms of liquid slip. J Fluid Mech 600:257–269. https://doi.org/10.1017/S0022112008000475
Michaelides A (2016) Slippery when narrow. Nature 537:171–172. https://doi.org/10.1038/537171a
Mouhat F, Coudert F-X, Bocquet M-L (2020) Structure and chemistry of graphene oxide in liquid water from first principles. Nat Commun 11:1566. https://doi.org/10.1038/s41467-020-15381-y
Nair RR, Wu HA, Jayaram PN et al (2012) Unimpeded permeation of water through helium-leak-tight graphene-based membranes. Science 335:442–444. https://doi.org/10.1126/science.1211694
Navier C (1823) Mémoire sur les lois du mouvement des fluides. Mém L’académie R Sci L’institut Fr 6:389–440
Plimpton S (1995) Fast parallel algorithms for short-range molecular dynamics. J Comput Phys 117:1–19. https://doi.org/10.1006/jcph.1995.1039
Sala J, Guàrdia E, Martí J (2012) Specific ion effects in aqueous eletrolyte solutions confined within graphene sheets at the nanometric scale. Phys Chem Chem Phys 14:10799–10808. https://doi.org/10.1039/C2CP40537G
Schoch RB, Han J, Renaud P (2008) Transport phenomena in nanofluidics. Rev Mod Phys 80:839–883. https://doi.org/10.1103/RevModPhys.80.839
Secchi E, Marbach S, Niguès A et al (2016) Massive radius-dependent flow slippage in carbon nanotubes. Nature 537:210–213. https://doi.org/10.1038/nature19315
Smith PE, van Gunsteren WF (1993) The viscosity of SPC and SPC/E water at 277 and 300 K. Chem Phys Lett 215:315–318. https://doi.org/10.1016/0009-2614(93)85720-9
Taherian F, Marcon V, van der Vegt NFA, Leroy F (2013) What is the contact angle of water on graphene? Langmuir 29:1457–1465. https://doi.org/10.1021/la304645w
Tocci G, Joly L, Michaelides A (2014) Friction of water on graphene and hexagonal boron nitride from ab initio methods: very different slippage despite very similar interface structures. Nano Lett 14:6872–6877. https://doi.org/10.1021/nl502837d
Vijayaraghavan V, Wong CH (2014) Transport characteristics of water molecules in carbon nanotubes investigated by using molecular dynamics simulation. Comput Mater Sci 89:36–44. https://doi.org/10.1016/j.commatsci.2014.03.025
Wei N, Peng X, Xu Z (2014a) Breakdown of fast water transport in graphene oxides. Phys Rev E 89:012113. https://doi.org/10.1103/PhysRevE.89.012113
Wei N, Peng X, Xu Z (2014b) Understanding water permeation in graphene oxide membranes. ACS Appl Mater Interfaces 6:5877–5883. https://doi.org/10.1021/am500777b
Whitby M, Quirke N (2007) Fluid flow in carbon nanotubes and nanopipes. Nat Nanotechnol 2:87–94. https://doi.org/10.1038/nnano.2006.175
Wu Y, Aluru NR (2013) Graphitic carbon–water nonbonded interaction parameters. J Phys Chem B 117:8802–8813. https://doi.org/10.1021/jp402051t
Wu Y, Tepper HL, Voth GA (2006) Flexible simple point-charge water model with improved liquid-state properties. J Chem Phys 124:024503. https://doi.org/10.1063/1.2136877
Yan J-A, Chou MY (2010) Oxidation functional groups on graphene: Structural and electronic properties. Phys Rev B 82:125403. https://doi.org/10.1103/PhysRevB.82.125403
Yang JZ, Wu X, Li X (2012) A generalized Irving–Kirkwood formula for the calculation of stress in molecular dynamics models. J Chem Phys 137:134104. https://doi.org/10.1063/1.4755946
Ye H, Zhang H, Zheng Y, Zhang Z (2011) Nanoconfinement induced anomalous water diffusion inside carbon nanotubes. Microfluid Nanofluidics 10:1359–1364. https://doi.org/10.1007/s10404-011-0772-y
Ye H, Wang J, Zheng Y et al (2021) Machine learning for reparameterization of four-site water models: TIP4P-BG and TIP4P-BGT. Phys Chem Chem Phys 23:10164–10173. https://doi.org/10.1039/D0CP05831A
Zhang Z, Li S, Mi B et al (2020) Surface slip on rotating graphene membrane enables the temporal selectivity that breaks the permeability-selectivity trade-off. Sci Adv 6:eaba9471. https://doi.org/10.1126/sciadv.aba9471
Zhao YP (2012) Physical mechanics of surfaces and interfaces. Science Press, Beijing
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (12241203, U22B2075) and the Youth Innovation Promotion Association CAS (2020449). The numerical calculations were performed on the supercomputing system in Hefei Advanced Computing Center and the Supercomputing Center of University of Science and Technology of China.
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Zhang, K., Fan, J. et al. Boundary slip moderated by interfacial hydrogen bond dynamics. Microfluid Nanofluid 27, 86 (2023). https://doi.org/10.1007/s10404-023-02695-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-023-02695-8