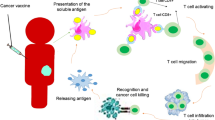
Abstract
Hepatocellular carcinoma (HCC) is one of the major health problems in the world. DCs-based vaccines are a promising immunotherapeutic strategy that aims at the optimal for induction of a specific antitumor immune response and destruction of tumor cells. The present study was conducted to investigate the immunogenic characters of whole tumor lysate-pulsed DCs vaccine and its ability to induce a specific antitumor immune response in HCC mice model. We also evaluate the effectiveness of prophylactic and therapeutic immunization strategies against HCC in mice models. Mice-derived DCs were in vitro loaded with whole tumor lysate prepared from liver tissue of HCC mice and evaluated for expression of surface maturation markers CD83 and CD86. In vivo immunization of mice with whole tumor lysate-pulsed DCs was performed in two strategies; prophylactic (pre-exposure to HCC) and therapeutic (post-exposure to HCC). Effectiveness of both protocols was investigated in terms of histopathological examination of liver sections and measurement of serum levels of immune cytokines interferon-γ (IFN-γ) and interleukin-2 (IL-2). Loading of DCs with whole tumor cell lysate exhibited a significant increase in expression of CD83 and CD86. In vivo administration of prophylactic doses of whole tumor lysate-pulsed DCs in mice before induction of HCC evokes a strong antitumor immune response presented by absence of malignant cells in liver sections and the significant increase in IFN-γ and IL-2. Data herein indicated that prophylactic vaccination with whole tumor lysate-pulsed DCs exhibited an effective antitumor immune response against HCC more than therapeutic protocol.








Similar content being viewed by others
Abbreviations
- CTLs:
-
Cytotoxic T-lymphocytes
- DCs:
-
Dendritic cells
- GM-CSF:
-
Granulocyte macrophage colony-stimulating factor
- HCC:
-
Hepatocellular carcinoma
- IL-4:
-
Interleukin-4
- IL-2:
-
Interleukin-2
- IFN-γ:
-
Interferon-γ
- PBMN:
-
Peripheral blood monocytes
- TAAs:
-
Tumor-associated antigens
References
Greten TF, Duffy AG, Korangy F. Hepatocellular carcinoma from an immunologic perspective. Clin Cancer Res. 2013;19(24):6678–85.
Palucka K, Banchereau J. Dendritic-cell-based therapeutic cancer vaccines. Immunity. 2013;39:38–48.
Han Y, Wu Y, Yang C, et al. Dynamic and specific immune responses against multiple tumor antigens were elicited in patients with hepatocellular carcinoma after cell-based immunotherapy. J Transl Med. 2017;15(1):64.
Miamen AG, Dong H, RoMiamen AG, Dong H, Roberts LR. Immunotherapeutic approaches to hepatocellular carcinoma treatment. Liver Cancer. 2012;1(3–4):226–37.
Rai V, Abdo J, Alsuwaidan AN, Agrawal S, Sharma P, Agrawal DK. Cellular and molecular targets for the immunotherapy of hepatocellular carcinoma. Mol Cell Biochem. 2017. https://doi.org/10.1007/s11010-017-3092-z.
Takahara A, Koido S, Ito M, et al. Gemcitabine enhances Wilms’ tumor gene WT1 expression and sensitizes human pancreatic cancer cells with WT1-specific T-cell-mediated antitumor immune response. Cancer Immunol Immunother. 2011;60:1289–97.
Santos PM, Butterfield LH. Dendritic cell-based cancer vaccines. J Immunol. 2018;200(2):443–9.
Hutten TJ, Thordardottir S, Fredrix H, et al. CLEC12A-mediated antigen uptake and cross-presentation by human dendritic cell subsets efficiently boost tumor-reactive T cell responses. J Immunol. 2016;197(7):2715–25.
Cohn L, Delamarre L. Dendritic cell-targeted vaccines. Front Immunol. 2014;5:255.
Wang W-H, Zhou C-H, Ding J, et al. Immune effect and safety evaluation of vaccine prepared by dendritic cells modified by rAAV-carrying BCSG1 gene. Gene Ther. 2016;23:839–45.
Kajihara M, Takakura K, Ohkusa T, Koido S. The impact of dendritic cell-tumor fusion cells on cancer vaccines-past progress and future strategies. Immunotherapy. 2015;7:1111–22.
Lin C-F, Lin C-M, Lee K-Y, et al. Escape from IFN-γ-dependent immunosurveillance in tumorigenesis. J Biomed Sci. 2017;24:10.
Beatty GL, Gladney WL. Immune escape mechanisms as a guide for cancer immunotherapy. Clin Cancer Res. 2015;21(4):687–92.
Chang YP, Chen CL, Chen SO, et al. Autophagy facilitates an IFN-gamma response and signal transduction. Microbes Infect. 2011;13(11):888–94.
Jiang T, Zhou C, Ren S. Role of IL-2 in cancer immunotherapy. Oncoimmunology. 2016;5(6):e1163462. https://doi.org/10.1080/2162402X.2016.1163462.
Skrombolas D, Frelinger JG. Challenges and developing solutions for increasing the benefits of IL-2 treatment in tumor therapy. Expert Rev Clin Immunol. 2014;10:207–17.
Wang Q, Luan W, Warren L, Kadri H, Kim KW, Goz V, Blank S, Isabel Fiel M, Hiotis SP. Autologous tumor cell lysate-loaded dendritic cell vaccine inhibited tumor progression in an orthotopic murine model for hepatocellular carcinoma. Ann Surg Oncol. 2016;23(Suppl 5):574–82.
Fecek RJ, Storkus WJ. Combination strategies to enhance the potency of monocyte-derived dendritic cell-based cancer vaccines. Immunotherapy. 2016;8(10):1205–18.
Shen J, Wang LF, Zou ZY, Kong WW, Yan J, Meng FY, Chen FJ, Du J, Shao J, Xu QP, Ren HZ, Li RT, Wei J, Qian XP, Liu BR. Phase I clinical study of personalized peptide vaccination combined with radiotherapy for advanced hepatocellular carcinoma. World J Gastroenterol. 2017;23(29):5395–404.
Zeng J, Huang X, Zhou L, et al. Metabolomics identifies biomarker pattern for early diagnosis of hepatocellular carcinoma: from diethylnitrosamine treated rats to patients. Sci Rep. 2015;5:16101. https://doi.org/10.1038/srep16101.
Markov OV, Mironova NL, Sennikov SV, Vlassov VV, Zenkova MA. Prophylactic dendritic cell-based vaccines efficiently inhibit metastases in murine metastatic melanoma. PLoS ONE. 2015;10(9):e0136911. https://doi.org/10.1371/journal.pone.0136911.
Lasky JL 3rd, Panosyan EH, Plant A, et al. Autologous tumor lysate-pulsed dendritic cell immunotherapy for pediatric patients with newly diagnosed or recurrent high-grade gliomas. Anticancer Res. 2013;33(5):2047–56.
Fleury P, Eberhard R. Determination of proteins in human milk by the biuret technic. Ann Biol Clin (Paris). 1955;13(1–2):1–7.
Yewdall AW, Drutman SB, Jinwala F, Bahjat KS, Bhardwaj N. CD8+ T cell priming by dendritic cell vaccines requires antigen transfer to endogenous antigen presenting cells. PLoS ONE. 2010. https://doi.org/10.1371/journal.pone.0011144.
Magalhães A, Ferreira KS, Almeida SR, Nosanchuk JD, Travassos LR, Taborda CP. Prophylactic and therapeutic vaccination using dendritic cells primed with peptide 10 derived from the 43-kilodalton glycoprotein of Paracoccidioides brasiliensis. Clin Vaccine Immunol. 2012;19(1):23–9.
Palucka K, Banchereau J. Dendritic cell-based cancer therapeutic vaccines. Immunity. 2013;39(1):38–48.
El-Ashmawy NE, El-Bahrawy HA, Shamloula MM, El-Feky OA. Biochemical/metabolic changes associated with hepatocellular carcinoma development in mice. Tumour Biol. 2014;35(6):5459–66.
Garg AD, Vandenberk L, Koks C, et al. Dendritic cell vaccines based on immunogenic cell death elicit danger signals and T cell-driven rejection of high-grade glioma. Sci Transl Med. 2016;8(328):328ra27. https://doi.org/10.1126/scitranslmed.aae0105.
Schmidt K, Seeger K, Scheibenbogen C, et al. Histone deacetylase inhibition improves differentiation of dendritic cells from leukemic blasts of patients with TEL/AML1-positive acute lymphoblastic leukemia. J Leukoc Biol. 2009;85(3):563–73.
Mehrotra S, Britten CD, Chin S, et al. Vaccination with poly(IC:LC) and peptide-pulsed autologous dendritic cells in patients with pancreatic cancer. J Hematol Oncol. 2017;10(1):82.
Farashi-Bonab S, Khansari N. Anticancer dendritic cell vaccines producing effective antitumor T Cell responses. Int J Vaccines Vaccin. 2017;4(3):00081.
Waldmann TA. Cytokines in cancer immunotherapy. Cold Spring Harb Perspect Biol. 2017;pii:a028472. https://doi.org/10.1101/cshperspect.a028472.
Ashrafi S, Shapouri R, Mahdavi M. Immunological consequences of immunization with tumor lysate vaccine and propranolol as an adjuvant: a study on cytokine profiles in breast tumor microenvironment. Immunol Lett. 2017;181:63–70.
Rainone V, Martelli C, Ottobrini L, et al. Immunological characterization of whole tumor lysate-loaded dendritic cells for cancer immunotherapy. PLoS ONE. 2016;11(1):e0146622. https://doi.org/10.1371/journal.pone.0146622.
Mendoza L, Bubeník J, Símová J, Jandlová T, Vonka V, Mikysková R. Prophylactic, therapeutic and anti-metastatic effects of BMDC and DC lines in mice carrying HPV 16-associated tumours. Int J Oncol. 2003;23:243–7.
Samadi-Foroushani M, Vahabpour R, Memarnejadian A, et al. Immune responses regulation following antitumor dendritic cell-based prophylactic, concurrent, and therapeutic vaccination. Med Oncol. 2011;28(Suppl 1):S660–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures involving animals were performed in accordance with institutional guidelines and with the approval of the Research Ethics Committee of Faculty of Pharmacy, Tanta University.
Informed consent
No human subjects in this research.
Rights and permissions
About this article
Cite this article
El-Ashmawy, N.E., El-Zamarany, E.A., Khedr, E.G. et al. Immunotherapeutic strategies for treatment of hepatocellular carcinoma with antigen-loaded dendritic cells: in vivo study. Clin Exp Med 18, 535–546 (2018). https://doi.org/10.1007/s10238-018-0521-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-018-0521-6