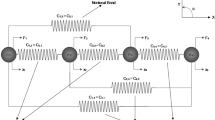
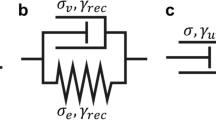
Abstract
Mechanical loading naturally controls cell phenotype, development, motility and various other biological functions; however, prolonged or substantial loading can cause cell damage and eventual death. Loading-induced mechanobiological and mechanostructural responses of different cell types affect their morphology and the internal architecture and the mechanics of the cellular components. Using single, mesenchymal stem cells, we have developed a cell-specific three-dimensional finite-element model; cell models were developed from phase-contrast microscopy images. This allowed us to evaluate the mechanostructural response of the naturally occurring variety of cell morphologies to increase sustained compressive loading. We focus on the morphology of the cytoplasm and the nucleus, as the main mechanically responsive elements, and evaluate formation of tensional strains and area changes in cells undergoing increasing uniaxial compressions. Here, we study mesenchymal stem cells as a model, due to their important role in tissue engineering and regenerative medicine; the method and findings are, however, applicable to any cell type. We observe variability in the cell responses to compression, which correlate directly with the morphology of the cells. Specifically, in cells with or without elongated protrusions (i.e., lamellipodia) tensional strains were, respectively, distributed mostly in the thin extensions or concentrated around the stiff nucleus. Thus, through cell-specific computational modeling of mechanical loading we have identified an underlying cause for stiffening (by actin recruitment) along the length of lamellipodia as well as a role for cell morphology in inducing cell-to-cell variability in mechanostructural response to loading.





Similar content being viewed by others
References
Abuhattum S, Gefen A, Weihs D (2015) Ratio of total traction force to projected cell area is preserved in differentiating adipocytes. Integr Biol 7(10):1212–1217. https://doi.org/10.1039/c5ib00056d
Admacher M, Anfred R (1999) Dimensional and mechanical dynamics of active and stable edges in motile fibroblasts investigated by using atomic force microscopy. Proc Natl Acad Sci USA 96:921–926. https://doi.org/10.1073/pnas.96.3.921
Alvarez-Elizondo MB, Barenholz-Cohen T, Weihs D (2019) Sodium Pyruvate pre-treatment prevents cell death due to localised, damaging mechanical strains in the context of pressure ulcers. Int Wound J. https://doi.org/10.1111/iwj.13173
Amsalem Y, Mardor Y, Feinberg MS, Landa N, Miller L, Daniels D, Ocherashvilli A et al (2007) Iron-oxide labeling and outcome of transplanted mesenchymal stem cells in the infarcted myocardium. Circulation 116(11 SUPPL. 1):38–46. https://doi.org/10.1161/CIRCULATIONAHA.106.680231
Baaijens FPT, Trickey WR, Laursen TA, Guilak F (2005) Large deformation finite element analysis of micropipette aspiration to determine the mechanical properties of the chondrocyte. Ann Biomed Eng 33(4):494–501. https://doi.org/10.1007/s10439-005-2506-3
Boehm B, Westerberg H, Lesnicar-Pucko G, Raja S, Rautschka M, Cotterell J, Swoger J, Sharpe J (2010) The role of spatially controlled cell proliferation in limb. PLoS Biol. https://doi.org/10.1371/journal.pbio.1000420
Breuls RGM, Bouten CVC, Oomens CWJ, Bader DL, Baaijens FPT (2003) Compression induced cell damage in engineered muscle tissue: an in vitro model to study pressure ulcer aetiology. Ann Biomed Eng 31(11):1357–1364. https://doi.org/10.1114/1.1624602
Brooker HR, Gyamfi IA, Wieckowska A, Brooks NJ, Mulvihill DP, Geeves MA (2018) A novel live-cell imaging system reveals a reversible hydrostatic pressure impact on cell-cycle progression. J Cell Sci. https://doi.org/10.1242/jcs.212167
Dailey HL, Ricles LM, Yalcin HC, Ghadiali SN (2009) Image-based finite element modeling of alveolar epithelial cell injury during airway reopening. J Appl Physiol 106:221–232. https://doi.org/10.1152/japplphysiol.90688.2008
Delaine-Smith RM, Reilly GC (2011) The effects of mechanical loading on mesenchymal stem cell differentiation and matrix production. stem cell regulators, vol 87, 1st edn. Elsevier, Amsterdam. https://doi.org/10.1016/B978-0-12-386015-6.00039-1
Dvir L, Nissim R, Alvarez-Elizondo MB, Weihs D (2015) Quantitative measures to reveal coordinated cytoskeleton-nucleus reorganization during in vitro invasion of cancer cells. New J Phys 17(4):043010. https://doi.org/10.1088/1367-2630/17/4/043010
Elder BD, Athanasiou KA (2009) Hydrostatic pressure in articular cartilage tissue engineering: from chondrocytes to tissue regeneration. Tissue Eng B Rev. https://doi.org/10.1089/ten.teb.2008.0435
Gefen A (2014) Tissue changes in patients following spinal cord injury and implications for wheelchair cushions and tissue loading: a literature review. Ostomy Wound Manag 60(2):34–45
Gefen A (2017) Why is the heel particularly vulnerable to pressure ulcers? Br J Nurs 26(Sup20):S62–S74. https://doi.org/10.12968/bjon.2017.26.Sup20.S62
Gefen A (2018) The future of pressure ulcer prevention is here: detecting and targeting inflammation early. EWMA J 19(2):7–13
Gefen A, Weihs D (2016) Cytoskeleton and plasma-membrane damage resulting from exposure to sustained deformations: a review of the mechanobiology of chronic wounds. Med Eng Phys 38(9):828–833. https://doi.org/10.1016/j.medengphy.2016.05.014
Gladilin E, Micoulet A, Hosseini B, Rohr K, Spatz J, Eils R (2007) 3D finite element analysis of uniaxial cell stretching: from image to insight. Phys Biol 4(2):104–113. https://doi.org/10.1088/1478-3975/4/2/004
Grotte M, Duprat F, Piétri E, Loonis D (2007) Young’s modulus, Poisson’s ratio, and Lame’s coefficients of golden delicious apple. Int J Food Prop 5(2):333–349. https://doi.org/10.1081/JFP-120005789
Hirano Y, Ishiguro N, Sokabe M, Takigawa M, Naruse K (2008) Effects of tensile and compressive strains on response of a chondrocytic cell line embedded in type I collagen gel. J Biotechnol 133(2):245–252. https://doi.org/10.1016/j.jbiotec.2007.07.955
Hochmuth RM, Mohandas N, Blackshear PL Jr (1973) Measurement of the elastic modulus for red cell membrane using a fluid mechanical technique. Biophys J 13(8):747–762. https://doi.org/10.1016/s0006-3495(73)86021-7
Icard-Arcizet D, Cardoso O, Richert A, Hénon S (2008) Cell stiffening in response to external stress is correlated to actin recruitment. Biophys J 94(7):2906–2913. https://doi.org/10.1529/BIOPHYSJ.107.118265
Jean RP, Chen CS, Spector AA (2005) Finite-element analysis of the adhesion-cytoskeleton-nucleus mechanotransduction pathway during endothelial cell rounding: axisymmetric model. J Biomech Eng 127(4):594–600. https://doi.org/10.1115/1.1933997
Katzengold R, Shoham N, Benayahu D, Gefen A (2015) Simulating single cell experiments in mechanical testing of adipocytes. Biomech Model Mechanobiol 14(3):537–547. https://doi.org/10.1007/s10237-014-0620-6
Kim N, Cho SG (2013) Clinical applications of mesenchymal stem cells. Korean J Intern Med. https://doi.org/10.3904/kjim.2013.28.4.387
Kristal-Muscal R, Dvir L, Weihs D (2013) Metastatic cancer cells tenaciously indent impenetrable, soft substrates. New J Phys 15(3):035022. https://doi.org/10.1088/1367-2630/15/3/035022
Laurent VM, Kasas S, Yersin A, Schäffer TE, Catsicas S, Dietler G, Verkhovsky AB, Meister J-J (2005) Gradient of rigidity in the lamellipodia of migrating cells revealed by atomic force microscopy. Biophys J 89(1):667–675. https://doi.org/10.1529/biophysj.104.052316
Linder-Ganz E, Gefen A (2009) Stress analyses coupled with damage laws to determine biomechanical risk factors for deep tissue injury during sitting. J Biomech Eng 131(1):11003. https://doi.org/10.1115/1.3005195
Linder-Ganz E, Engelberg S, Scheinowitz M, Gefen A (2006) Pressure-time cell death threshold for albino rat skeletal muscles as related to pressure sore biomechanics. J Biomech 39(14):2725–2732. https://doi.org/10.1016/j.jbiomech.2005.08.010
Loerakker S (2007) Aetiology of pressure ulcers. Eindhoven University of Technology (TU/E), Eindhoven
Maas SA, Ellis BJ, Ateshian GA, Weiss JA (2012) FEBio: finite elements for biomechanics. J Biomech Eng 134(1):011005. https://doi.org/10.1115/1.4005694
Marom A, Berkovitch Y, Toume S, Alvarez-Elizondo MB, Weihs D (2019) Non-damaging stretching combined with sodium pyruvate supplement accelerate migration of fibroblasts and myoblasts during gap closure. Clin Biomech 62(January):96–103. https://doi.org/10.1016/J.CLINBIOMECH.2019.01.009
Massalha S, Weihs D (2017) Metastatic breast cancer cells adhere strongly on varying stiffness substrates, initially without adjusting their morphology. Biomech Model Mechanobiol 16(3):961–970. https://doi.org/10.1007/s10237-016-0864-4
Milisav I (2011) Cellular stress responses. In: Wislet-Gendebien S (ed) Advances in regenerative medicine. InTech, London, pp 215–232. https://doi.org/10.1016/j.molcel.2010.09.022
Mizrahi N, Zhou EHH, Lenormand G, Krishnan R, Weihs D, Butler JP, Weitz DA, Fredberg JJ, Kimmel E (2012) Low intensity ultrasound perturbs cytoskeleton dynamics. Soft Matter 8(8):2438–2443. https://doi.org/10.1039/C2sm07246g
O’Conor CJ, Case N, Guilak F (2013) Mechanical regulation of chondrogenesis. Stem Cell Res Therapy 4(4):61. https://doi.org/10.1186/scrt211
Peeters EAG, Bouten CVC, Oomens CWJ, Baaijens FPT (2003) Monitoring the biomechanical response of individual cells under compression: a new compression device. Med Biol Eng Comput 41(4):498–503. https://doi.org/10.1007/BF02348096
Peeters EAG, Oomens CWJ, Bouten CVC, Bader DL, Baaijens FPT (2005) Mechanical and failure properties of single attached cells under compression. J Biomech 38(8):1685–1693. https://doi.org/10.1016/j.jbiomech.2004.07.018
Pileggi A (2012) Mesenchymal stem cells for the treatment of diabetes. Diabetes 61(6):1355–1356. https://doi.org/10.2337/db12-0355
Responte DJ, Lee JK, Hu JC, Athanasiou KA (2012) Biomechanics-driven chondrogenesis: from embryo to adult. FASEB J. https://doi.org/10.1096/fj.12-207241
Rotsch C, Jacobson K, Condeelis J, Radmacher M (2001) EGF-stimulated lamellipod extension in adenocarcinoma cells. Ultramicroscopy 86:97–106. https://doi.org/10.1016/s0304-3991(00)00102-9
Saeed M, Sharabani-Yosef O, Weihs D, Gefen A (2016) A phase-contrast microscopy-based method for modeling the mechanical behavior of mesenchymal stem cells. Comput Methods Biomech Biomed Eng 19(13):1359–1362. https://doi.org/10.1080/10255842.2016.1141900
Shoham N, Gefen A (2012) Deformations, mechanical strains and stresses across the different hierarchical scales in weight-bearing soft tissues. J Tissue Viability 21(2):39–46. https://doi.org/10.1016/j.jtv.2012.03.001
Slomka N, Gefen A (2010) Confocal microscopy-based three-dimensional cell-specific modeling for large deformation analyses in cellular mechanics. J Biomech 43(9):1806–1816. https://doi.org/10.1016/j.jbiomech.2010.02.011
Slomka N, Gefen A (2011a) Cell-to-cell variability in deformations across compressed myoblasts. J Biomech Eng 133(8):081007. https://doi.org/10.1115/1.4004864
Slomka N, Gefen A (2011b) Finite element modeling of cellular mechanics experiments BT. In: Gefen A (ed) Cellular and biomolecular mechanics and mechanobiology. Springer, Berlin, pp 331–344. https://doi.org/10.1007/8415_2010_31
Slomka N, Or-Tzadikario S, Sassun D, Gefen A (2009) Membrane-stretch-induced cell death in deep tissue injury: computer model studies. Cell Mol Bioeng 2(1):118–132. https://doi.org/10.1007/s12195-009-0046-x
Slomka N, Oomens CWJ, Gefen A (2011) Evaluating the effective shear modulus of the cytoplasm in cultured myoblasts subjected to compression using an inverse finite element method. J Mech Behav Biomed Mater 4(7):1559–1566. https://doi.org/10.1016/j.jmbbm.2011.04.006
Stegemann JP, Hong H, Nerem RM, Jan P (2005) Mechanical, biochemical, and extracellular matrix effects on vascular smooth muscle cell phenotype. J Appl Physiol 98(6):2321–2327. https://doi.org/10.1152/japplphysiol.01114.2004
Toume S, Gefen A, Weihs D (2017) Low-level stretching accelerates cell migration into a gap. Int Wound J 14(4):698–703. https://doi.org/10.1111/iwj.12679
Vermolen FJ, Van Der Meijden RP, Mv Van Es AG, Weihs D (2015) Towards a mathematical formalism for semi-stochastic cell-level computational modeling of tumor initiation. Ann Biomed Eng 43(7):1680–1694. https://doi.org/10.1007/s10439-015-1271-1
Weihs D, Gefen A (2018) Methods and compositions for prevention and treatment of pressure ulcers. PCT/IL2018/051424, issued 2018
Yabuki T, Yamanoha B, Shimizu A (2013) Effect of hydrostatic pressure on the viability of non-adherent HL-60 cells. High Pressure Res 33(2):315–321. https://doi.org/10.1080/08957959.2013.769532
Zhu C, Bao G, Wang N (2000) Cell mechanics: mechanical response, cell adhesion, and molecular deformation. Annu Rev Biomed Eng 2:189–226. https://doi.org/10.1146/annurev.bioeng.2.1.189
Acknowledgements
The work was partially supported by the Israeli Ministry of Science, Technology and Space in the Program for Science, Technology and Innovation for Third Age Populations (Grant No. 3-12804, awarded jointly to Drs. Daphne Weihs and Amit Gefen in 2016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saeed, M., Weihs, D. Finite element analysis reveals an important role for cell morphology in response to mechanical compression. Biomech Model Mechanobiol 19, 1155–1164 (2020). https://doi.org/10.1007/s10237-019-01276-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-019-01276-5