Abstract
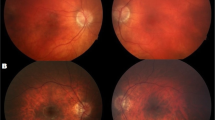
Neurodevelopmental disorder with spastic diplegia and visual defects (NEDSDV) is a rare autosomal dominant genetic disorder caused by genetic alterations in the CTNNB1 gene. CTNNB1 is a gene that encodes β-catenin, an effector protein in the canonical Wnt pathway involved in stem cell differentiation and proliferation, synaptogenesis, and a wide range of essential cellular mechanisms. Mutations in this gene are also found in specific malignancies as well as exudative vitreoretinopathy. To date, only a limited number of cases of this disease have been reported, and though they share some phenotypic manifestations such as intellectual disability, developmental delay, microcephaly, behavioral abnormalities, and dystonia, the variety of phenotypic traits of these patients shows extreme heterogeneity. In this study, two cases of NEDSDV with de novo CTNNB1 mutations: c.1420C>T(p.R474X) and c.1377_1378Del(p.Ala460Serfs*29), found with whole exome sequencing (WES) have been reported and the clinical and paraclinical characteristics of these patients have been described. Due to such a wide range of clinical characteristics, the identification of new patients and novel variants is of great importance in order to establish a more complete phenotypic spectrum, as well as to conclude the genotype-phenotype correlations in these cases.



Similar content being viewed by others
Data availability
The data related to this work is available from the corresponding authors.
References
Kwan V, Unda BK, Singh KK (2016) Wnt signaling networks in autism spectrum disorder and intellectual disability. J Neurodev Disord 8:45
Nusse R (2012) Wnt signaling. Cold Spring Harb Perspect Biol 4(5):a011163
Noelanders R, Vleminckx K (2017) How Wnt signaling builds the brain: bridging development and disease. Neuroscientist 23:314–329
Mo Z, Zeng Z et al (2022) Activation of Wnt/beta-catenin signaling pathway as a promising therapeutic candidate for cerebral ischemia/reperfusion injury. Front Pharmacol 13:914537
Zhuang W, Ye T, Wang W, Song W, Tan T (2023) CTNNB1 in neurodevelopmental disorders. Front Psychiatry 14:1143328
Spagnoli C, Salerno GG et al (2022) Novel CTNNB1 variant leading to neurodevelopmental disorder with spastic diplegia and visual defects plus peripheral neuropathy: a case report. Am J Med Genet A 188:3118–3120
Bulot V, Ramond F, Mauguiere F, Mazzola L (2022) Startle disease: an overlooked symptom of CTNNB1-related neurodevelopmental disorder with spastic diplegia and visual defects. Neurol Genet 8:e200039
Dashti S, Salehpour S et al (2022) Identification of a novel de novo mutation in the CTNNB1 gene in an Iranian patient with intellectual disability. Neurol Sci 43:2859–2863
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Li H, Durbin R (2010) Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26:589–595
McKenna A, Hanna M et al (2010) The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303
Wang K, Li M, Hakonarson H (2010) ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38:e164
Schwarz JM, Cooper DN, Schuelke M, Seelow D (2014) MutationTaster2: mutation prediction for the deep-sequencing age. Nat Methods 11:361–362
Shihab HA, Gough J et al (2013) Predicting the functional, molecular, and phenotypic consequences of amino acid substitutions using hidden Markov models. Hum Mutat 34:57–65
Quang D, Chen Y, Xie X (2015) DANN: a deep learning approach for annotating the pathogenicity of genetic variants. Bioinformatics 31:761–763
Richards S, Aziz N et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–423
Nollet F, Berx G, Molemans F, van Roy F (1996) Genomic organization of the human beta-catenin gene (CTNNB1). Genomics 32:413–424
Bailey A, Norris AL et al (1995) Yeast artificial chromosome cloning of the beta-catenin locus on human chromosome 3p21-22. Chromosome Res 3:201–203
Trent JM, Wiltshire R et al (1995) The gene for the APC-binding protein beta-catenin (CTNNB1) maps to chromosome 3p22, a region frequently altered in human malignancies. Cytogenet Cell Genet 71:343–344
Valenta T, Hausmann G, Basler K (2012) The many faces and functions of beta-catenin. EMBO J 31:2714–2736
Clevers H, Nusse R (2012) Wnt/beta-catenin signaling and disease. Cell 149:1192–1205
Kobayashi M, Honma T et al (2000) Nuclear translocation of beta-catenin in colorectal cancer. Br J Cancer 82:1689–1693
Simcha I, Shtutman M et al (1998) Differential nuclear translocation and transactivation potential of beta-catenin and plakoglobin. J Cell Biol 141:1433–1448
Lee S, Jang SS et al (2022) The extended clinical and genetic spectrum of CTNNB1-related neurodevelopmental disorder. Front Pediatr 10:960450
Kuechler A, Willemsen MH et al (2015) De novo mutations in beta-catenin (CTNNB1) appear to be a frequent cause of intellectual disability: expanding the mutational and clinical spectrum. Hum Genet 134:97–109
Kharbanda M, Pilz DT et al (2017) Clinical features associated with CTNNB1 de novo loss of function mutations in ten individuals. Eur J Med Genet 60:130–135
Vissers L, van Nimwegen KJM et al (2017) A clinical utility study of exome sequencing versus conventional genetic testing in pediatric neurology. Genet Med 19:1055–1063
Huelsken J, Vogel R, Erdmann B, Cotsarelis G, Birchmeier W (2001) Beta-catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell 105:533–545
Yan D, Sun Y et al (2022) Genetic and clinical characteristics of 24 mainland Chinese patients with CTNNB1 loss-of-function variants. Mol Genet Genomic Med 10:e2067
Haseeb M, Pirzada RH, Ain QU, Choi S (2019) Wnt signaling in the regulation of immune cell and cancer therapeutics. Cells 8(11):1380. https://doi.org/10.3390/cells8111380
Namazzi G, Hildenwall H et al (2019) Prevalence and associated factors of neurodevelopmental disability among infants in eastern Uganda: a population based study. BMC Pediatr 19:379
Mamidala MP, Polinedi A et al (2013) Prenatal, perinatal and neonatal risk factors of autism spectrum disorder: a comprehensive epidemiological assessment from India. Res Dev Disabil 34:3004–3013
Sachdeva S, Amir A et al (2010) Global developmental delay and its determinants among urban infants and toddlers: a cross sectional study. Indian J Pediatr 77:975–980
Liu J, Xiao Q et al (2022) Wnt/beta-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct Target Ther 7:3
Nusse R, Varmus HE (1982) Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell 31:99–109
Niehrs C (2012) The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol 13:767–779
Clevers H (2006) Wnt/beta-catenin signaling in development and disease. Cell 127:469–480
Akoumianakis I, Polkinghorne M, Antoniades C (2022) Non-canonical WNT signalling in cardiovascular disease: mechanisms and therapeutic implications. Nat Rev Cardiol 19:783–797
Chavali M, Klingener M et al (2018) Non-canonical Wnt signaling regulates neural stem cell quiescence during homeostasis and after demyelination. Nat Commun 9:36
Davidson KC, Jamshidi P et al (2007) Wnt3a regulates survival, expansion, and maintenance of neural progenitors derived from human embryonic stem cells. Mol Cell Neurosci 36:408–415
Dravid G, Ye Z et al (2005) Defining the role of Wnt/beta-catenin signaling in the survival, proliferation, and self-renewal of human embryonic stem cells. Stem Cells 23:1489–1501
Inestrosa NC, Varela-Nallar L (2014) Wnt signaling in the nervous system and in Alzheimer’s disease. J Mol Cell Biol 6:64–74
Huelsken J, Vogel R et al (2000) Requirement for beta-catenin in anterior-posterior axis formation in mice. J Cell Biol 148:567–578
Lickert H, Kutsch S et al (2002) Formation of multiple hearts in mice following deletion of beta-catenin in the embryonic endoderm. Dev Cell 3:171–181
Thomas KR, Capecchi MR (1990) Targeted disruption of the murine int-1 proto-oncogene resulting in severe abnormalities in midbrain and cerebellar development. Nature 346:847–850
Wickham RJ, Alexander JM et al (2019) Learning impairments and molecular changes in the brain caused by beta-catenin loss. Hum Mol Genet 28:2965–2975
Brigidi GS, Bamji SX (2011) Cadherin-catenin adhesion complexes at the synapse. Curr Opin Neurobiol 21:208–214
Kayumi S, Perez-Jurado LA et al (2022) Genomic and phenotypic characterization of 404 individuals with neurodevelopmental disorders caused by CTNNB1 variants. Genet Med 24:2351–2366
Acknowledgements
The authors acknowledge the family who participated in this study.
Author information
Authors and Affiliations
Contributions
AM, STF, M-RG, PK, PL, and MM completed recruitment and performed clinical confirmation. M-RG, FH-G, and MM performed molecular experiments like DNA extraction and analyzed WES results. Bioinformatic analyses were done by AM, M-RG, and FH-G. AM and STF wrote the manuscript. M-RG, FH-G, HS, RM, and MM contributed to the revisions of the manuscript. The final revision has been confirmed and essential ideas for the revision of the manuscript have been provided by all authors.
Corresponding authors
Ethics declarations
Ethical approval and consent to participate
This study was approved by the Research Ethics Committee of the Faculty of Medicine, Shahid Beheshti University of Medical Sciences, and was conducted in accordance with the tenets of the Declaration of Helsinki. Informed consent was obtained from parents.
Consent for publication
All the contributing authors agreed for the publication in this journal.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moeinafshar, A., Tehrani Fateh, S., Sadeghi, H. et al. Alopecia areata-like pattern of baldness: the most recent update and the expansion of novel phenotype and genotype in the CTNNB1 gene. Neurol Sci 44, 4041–4048 (2023). https://doi.org/10.1007/s10072-023-06922-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-06922-6