Abstract
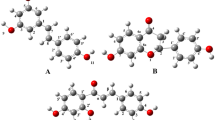
In this work, we present a computational investigation on the reactions between two well-known antioxidants (quercetin and morin) and 2,2-diphenyl-1-picrylhydrazyl (DPPH). A density functional theory (DFT) approach with the B3LYP functional and the 6-31G(d,p) basis set was used for the simulations. The structural and energetic parameters (Gibbs free-energy, ΔG, and Gibbs free-energy of activation, ΔG++) were determined to provide information on the antioxidant activity as well as to evaluate the contributions of each hydroxyl group to the referred property. According to the results obtained, quercetin presented three hydroxyls as being thermodynamically spontaneous in the reaction with DPPH (4\(^{\prime }\)-ArOH, 3\(^{\prime }\)-ArOH, and 3-ArOH, with ΔG = -4.93 kcal/mol, -2.89 kcal/mol, and -1.87 kcal/mol, respectively) against only two in the case of morin (2\(^{\prime }\)-ArOH and 3-ArOH, with ΔG = -7.56 kcal/mol and -4.57 kcal/mol, respectively). Hence, quercetin was found to be a more efficient antioxidant, which is in agreement with different experimental and computational investigations of bond dissociation enthalpies (BDEs). However, the order of contribution of the OH groups of each compound to the antioxidant potential present some differences when compared to what was seen in the previous investigations, especially for morin. These findings are in contrast to what was observed in studies based on the determinations of BDEs. Therefore, experimental investigations on the hydrogen-atom transfer mechanism (HAT) for both compounds are encouraged in order to clarify these observations.







Similar content being viewed by others
References
Shahidi F, Ambigaipalan P (2015) Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects – A review. J Funct Foods 18:820–897
Zhang XC, Chen F, Wang MF (2014) Antioxidant and antiglycation activity of selected dietary polyphenols in a cookie model. J Agric Food Chem 62:1643–1648
Wu S, Yano S, Chen J, Hisanaga A, Sakao K, He J, Hou D-X (2017) Polyphenols from Lonicera caerulea L. berry inhibit LPS-induced inflammation through dual modulation of inflammatory and antioxidant mediators. J Agric Food Chem 65:5133–5141
Aswathy VV, Alper-Hayta S, Yalcin G, Mary YS, Panicker CY, Jojo PJ, Kaynak-Onurdag F, Armaković S, Armaković SJ, Yildiz I, Alsenoy CV (2017) Modification of benzoxazole derivative by bromine-spectroscopic, antibacterial and reactivity study using experimental and theoretical procedures. J Mol Struct 1141:495–511
Belščak-Cvitanović A, Durgo K, Bušić A, Franekić J, Komes D (2014) Phytochemical attributes of four conventionally extracted medicinal plants and cytotoxic evaluation of their extracts on human laryngeal carcinoma (HEp2) cells. J Med Food 17:206–217
Malig TC, Ashkin MR, Burman AL, Barday M, Heyne BJ, Back TG (2017) Comparison of free-radical inhibiting antioxidant properties of carvedilol and its phenolic metabolites. Med Chem Comm 8:606–615
Ren F, Reilly K, Kerry JP, Gaffney M, Hossain M, Rai DK (2017) Higher antioxidant activity, total flavonols, and specific quercetin glucosides in two different onion (Allium cepa L.) varieties grown under organic production: Results from a 6-year field study. J Agric Fodd Chem 65:5122–5132
de Souza GLC, de Oliveira LMF, Vicari RG, Brown A (2016) A DFT investigation on the structural and antioxidant properties of new isolated interglycosidic O-(13) linkage flavonols. J Mol Model 22:100–109
Guajardo-Flores D, Serna-Saldivar SO, Gutiérrez-Uribe JA (2013) Evaluation of the antioxidant and antiproliferative activities of extracted saponins and flavonols from germinated black beans (Phaseolus vulgaris L.) Food Chem 141:1497– 1503
Mendes RA, e Silva BLS, Takeara R, Freitas RG, Brown A, de Souza GLC (2018) Probing the antioxidant potential of phloretin and phlorizin through a computational investigation. J Mol Model 24:101
Mendes RA, Almeida SKC, Soares IN, Barboza CA, Freitas RG, Brown A, de Souza GLC (2018) A computational investigation on the antioxidant potential of myricetin 3,4\(^{\prime }\)-di-O-α-L-rhamnopyranoside. Mol Model 24:133
Wright JS, Johnson ER, DiLabio GA (2001) Predicting the activity of phenolic antioxidants: Theoretical method, analysis of substituent effects, and application to major families of antioxidants. J Am Chem Soc 123:1173–1183
Leopoldini M, Pitarch IP, Russo N, Toscano M (2004) Structure, conformation, and electronic properties of apigenin, luteolin, and taxifolin antioxidants. A first principle theoretical study. J Phys Chem A 108:92–96
Cai W, Chen Y, Xie L, Zhang H, Hou C (2014) Characterization and density functional theory study of the antioxidant activity of quercetin and its sugar-containing analogues. Eur Food Res Technol 238:121–128
Zheng Y-Z, deng G, Liang Q, Chen D-F, Lai R-C (2017) Antioxidant activity of quercetin and its glucosides from propolis: A theoretical study. Sci Rep 7:7543
Guitard R, Nardello-Rataj V, Aubry J-M (2016) Theoretical and kinetic tools for selecting effective antioxidants: Application to the protection of omega-3 oils with natural and synthetic phenols. Int J Mol Sci 17:1220
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Vosko SH, Wilk L, Nusair M (1980) Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. Can J Phys 58:1200–1211
Stephens PJ, Devlin FJ, Chabalowski CF, Frisch MJ (1994) Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J Phys Chem 98:11623–11627
Zhao Y, Truhlar DG (2006) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Account 120:215–241
Justino GC, Vieira AJSC (2010) Antioxidant mechanisms of quercetin and myricetin in the gas phase and in solution—a comparison and validation of semi-empirical methods. J Mol Model 16:863–876
Mohajeri A, Asemani SS (2009) Theoretical investigation on antioxidant activity of vitamins and phenolic acids for designing a novel antioxidant. J Mol Struct 930:15–20
Sadasivam K, Kumaresan R (2011) Antioxidant behavior of mearnsetin and myricetin flavonoid compounds—a DFT study. Spectrochim Acta A 79:282–293
Nenadis N, Sigalas MP (2008) A DFT study on the radical scavenging activity of maritimetin and related aurones. J Phys Chem A 112:12196–12202
Koleva II, van Beek TA, Linssen JPH, de Groot A, Evstatieva LN (2002) Screening of plant extracts for antioxidant activity: A comparative study on three testing methods. Phytochem Anal 13:9–17
Bran-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. Lebensm Wiss Technol 28:25–30
Trouillas P, Marsal P, Svobovova A, Vostalova J, Gazak R, Hbrac J, Sedmera P, Kren V, Lazzaroni R, Duroux J-L, Walterova D (2008) Mechanism of the antioxidant action of silybin and 2,3-dehydrosilybin flavonolignans: A joint experimental and theoretical study. J Phys Chem A 112:1054–1063
Fezai R, Mezni A, Rzaigui M (2018) Synthesis, structural analysis, Hirshfeld surface, spectroscopic characterization and, in vitro, antioxidant activity of a novel organic cyclohexaphosphate. J Mol Struct 1154:64–71
Yang W, Fortunati E, Bertoglio F, Owczarek JS, Bruni G, Kozanecki M, Kenny JM, Torre L, Visai L, Puglia D (2018) Polyvinyl alcohol/chitosan hydrogels with enhanced antioxidant and antibacterial properties induced by lignin nanoparticles. Carbohydr Polym 181:275–284
Adilah ZAM, Jamilah B, Hanani ZAN (2018) Functional and antioxidant properties of protein-based films incorporated with mango kernel extract for active packaging. Food Hydrocoll 74:207–218
Nantitanon W, Okonogi S (2012) Comparison of antioxidant activity of compounds isolated from guava leaves and a stability study of the most active compound. Drug Discov Ther 6:38–43
De Martino L, Mencherini T, Mancini E, Aquino RP, De Almeida LFR, De Feo V (2012) In vitro phytotoxicity and antioxidant activity of selected flavonoids. Int J Mol Sci 13:5406–5419
Jones RS, Parker MD, Morris ME (2017) Quercetin, morin, luteolin, and phloretin are dietary flavonoid inhibitors of monocarboxylate transporter 6. Mol Pharmaceutics 14:2930–2936
Ricardo KFS, de Oliveira TT, Nagem TJ, Pinto AS, Oliveira MGA, Soares JF (2001) Effect of flavonoids morin; quercetin and nicotinic acid on lipid metabolism of rats experimentally fed with triton. Braz Arch Biol Technol 44:263–267
Rassolov V, Pople JA, Ratner M, Redfern PC, Curtiss LA (2001) 6-31G* basis set for third-row atoms. J Comp Chem 22:976–984
Binkley JS, Pople JA, Hehre WJ (1980) Self-consistent molecular orbital methods. 21. Small split-valence basis sets for first-row elements. J Am Chem Soc 102:939–946
Vagánek A, Rimarčik J, Lukeš V, Klein E (2012) On the energetics of homolytic and heterolytic O–H bond cleavage in flavonols. Comput Theor Chem 991:192–200
Rajaraman D, Sundararajan G, Rajkumar R, Bharanidharan S, Krishnasamy K (2016) Synthesis, crystal structure investigation, DFT studies and DPPH radical scavenging activity of 1-(furan-2-ylmethyl)-2,4,5-triphenyl-1H-imidazole derivatives. J Mol Struct 1108:698–707
Scalmani G, Frisch MJ (2010) Continuous surface charge polarizable continuum models of solvation. I. General formalism. J Chem Phys 132:114110
Cancès E, Mennucci B, Tomasi J (1997) A new integral equation formalism for the polarizable continuum model: Theoretical background and applications to isotropic and anisotropic dielectrics. J Chem Phys 107:3032–3041
Mennucci B, Cancès E, Tomasi J (1997) Evaluation of solvent effects in isotropic and anisotropic dielectrics, and in ionic solutions with a unified integral equation method: Theoretical bases, computational implementation and numerical applications. J Phys Chem B 101:10506–10517
Miliauskas G, Venskutonis PR, van Beek TA (2004) Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem 85:231–237
Kumaran A, Karunakaran RJ (2007) In vitro antioxidant activities of methanol extracts of five Phyllanthus species from India. LWT - Food Sci Technol 40:344–352
Sharma S, Vig AP (2013) Evaluation of in vitro antioxidant properties of methanol and aqueous extracts of Parkinsonia aculeata L. Laves Sci World J 2013:604865
Mahdi-Pour B, Jothy SL, Latha LY, Chen Y, Sasidharan S (2012) Antioxidant activity of methanol extracts of different parts of Lantana camara. Asian Pac J Trop Biomed 2:960– 965
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr. J A, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian, Inc., Wallingford CT, Gaussian 09, Revision D.01
Zhang D, Chu L, Liu Y, Wang A, Ji B, Wu W, Zhou F, Wei Y, Cheng Q, Cai S, Xie L, Jia G (2011) Analysis of the antioxidant capacities of flavonoids under different spectrophotometric assays using cyclic voltammetry and density functional theory. J Agric Food Chem 59:10277–10285
Trouillas P, Marsal P, Siri D, Lazzaroni R, Duroux J-L (2006) A DFT study of the reactivity of OH groups in quercetin and taxifolin antioxidants: The specificity of the 3-OH site. Food Chem 97:679–688
Li M-J, Liu L, Fu Y, Guo Q-X (2007) Accurate bond dissociation enthalpies of popular antioxidants predicted by the ONIOM-G3B3 method. J Mol Struct THEOCHEM 815:1–9
Fiorucci S, Golebiowski J, Cabrol-Bass D, Antonczak S (2007) DFT Study of quercetin activated forms involved in antiradical, antioxidant, and prooxidant biological processes. J Agric Food Chem 55:903–911
Amić D, Stepanić W, Lučić R, Marković Z, Dmitrić Marković J M (2013) PM6 study of free radical scavenging mechanisms of flavonoids: Why does OH bond dissociation enthalpy effectively represent free radical scavenging activity. J Mol Model 19:2593–2603
Marković Z, Milenković D, Dorović J, Marković JMD, Stepanić V, Lucić B, Amić D (2012) PM6 and DFT study of free radical scavenging activity of morin. Food Chem 134:1754–1760
Pérez-González A, Rebollar-Zepeda AM, Léon-Carmona JR, Galano A (2012) Reactivity indexes and O-H bond dissociation energies of a large series of polyphenols: implications for their free radical scavenging activity. J Mex Chem Soc 56:241–249
Islam N (2015) Investigation of comparative shielding of morin against oxidative damage by radicals: A DFT study. Cogent Chem 1:1078272–1–1078272-9
Acknowledgments
First of all, “Fora Temer!”. The Brazilian agency CNPq funded this work (Process number: 306266/2016-4). The authors thank Prof. Alex Brown from Department of Chemistry at University of Alberta for suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper belongs to Topical Collection XIX - Brazilian Symposium of Theoretical Chemistry (SBQT2017)
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maciel, E.N., Almeida, S.K.C., da Silva, S.C. et al. Examining the reaction between antioxidant compounds and 2,2-diphenyl-1-picrylhydrazyl (DPPH) through a computational investigation. J Mol Model 24, 218 (2018). https://doi.org/10.1007/s00894-018-3745-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-018-3745-1