Abstract
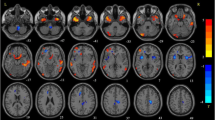
Parkinson’s disease (PD) can be classified into the tremor dominant (TD) subtype and the postural instability gait difficulty (PIGD) subtype, which present with different clinical courses and prognoses. However, the symptom-specific intrinsic neural mechanisms underlying the subtypes of PD still remain elusive. In the current study, we utilized resting-state fMRI (rs-fMRI) combined with the regional homogeneity (ReHo) method to investigate the modulations of neural activity in 13 patients with predominantly PIGD (p-PIGD) and 15 patients with predominantly TD (p-TD) in the resting state. Compared with healthy controls, the p-PIGD and the p-TD groups both displayed ReHo changes in the default mode network (DMN). By contrast, the p-TD group exhibited more ReHo alterations in the cerebellum involved in the cerebello-thalamo-cortical (CTC) loops, whilst the p-PIGD group in extensive cortical and sub-cortical areas, including the frontal, parietal, occipital, temporal, limbic lobes, basal ganglia and thalamus, which are involved in the striatal-thalamo-cortical (STC) loops. Direct comparison between the two groups showed significant ReHo alterations in the primary visual cortex. Our findings underscore the differential involvement of the STC and CTC circuits underlying the two subtypes of PD. Moreover, relatively widespread neural activity abnormality, especially in the motor-related regions as well as the visual network, is apparently a characteristic feature of PIGD symptoms. This study could shed light on the underlying pathophysiology and clinical heterogeneity of PD presentation.



Similar content being viewed by others
References
Alexander GE, Crutcher MD, DeLong MR (1990) Basal ganglia-thalamocortical circuits: parallel substrates for motor, oculomotor, “prefrontal” and “limbic” functions. Prog Brain Res 85:119–146
Alon G, Yungher DA, Shulman LM, Rogers MW (2012) Safety and immediate effect of noninvasive transcranial pulsed current stimulation on gait and balance in Parkinson disease. Neurorehabil Neural Repair 26:1089–1095
Auyeung M, Tsoi TH, Mok V, Cheung CM, Lee CN, Li R, Yeung E (2012) Ten year survival and outcomes in a prospective cohort of new onset Chinese Parkinson’s disease patients. J Neurol Neurosurg Psychiatry 83:607–611
Benninger DH, Thees S, Kollias SS, Bassetti CL, Waldvogel D (2009) Morphological differences in Parkinson’s disease with and without rest tremor. J Neurol 256:256–263
Biswal B, Yetkin FZ, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34:537–541
Bohnen NI, Koeppe RA, Minoshima S, Giordani B, Albin RL, Frey KA, Kuhi DE (2011) Cerebral glucose metabolic features of Parkinson disease and incident dementia: longitudinal study. J Nucl Med 52:848–855
Cardoso EF et al (2010) Abnormal visual activation in Parkinson’s disease patients. Mov Disord 25:1590–1596
Choe IH, Yeo S, Chung KC, Kim SH, Lim S (2013) Decreased and increased cerebral regional homogeneity in early Parkinson’s disease. Brain Res 1527:230–237
Drew T, Prentice S, Schepens B (2004) Cortical and brainstem control of locomotion. Prog Brain Res 143:251–261
Dubois B, Slachevsky A, Litvan I, Pillon B (2000) The FAB: a frontal assessment battery at bedside. Neurology 55:1621–1626
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Garcia-Garcia D et al (2012) Posterior parietooccipital hypometabolism may differentiate mild cognitive impairment from dementia in Parkinson’s disease. Eur J Nucl Med Mol Imaging 39:1767–1777
Giladi N, Shabtai H, Simon ES, Biran S, Tal J, Korczyn AD (2000) Construction of freezing of gait questionnaire for patients with Parkinsonism. Parkinsonism Relat Disord 6:165–170
Grafton ST (2004) Contributions of functional imaging to understanding parkinsonian symptoms. Curr Opin Neurobiol 14:715–719
Helmich RC, de Lange FP, Bloem BR, Toni I (2007) Cerebral compensation during motor imagery in Parkinson’s disease. Neuropsychologia 45:2201–2215
Helmich RC, Hallett M, Deuschl G, Toni I, Bloem BR (2012) Cerebral causes and consequences of parkinsonian resting tremor: a tale of two circuits. Brain 135:3206–3226
Herman T, Rosenberg-Katz K, Jacob Y, Auriel E, Gurevich T, Giladi N, Hausdorff JM (2013) White matter hyperintensities in Parkinson’s disease: do they explain the disparity between the postural instability gait difficulty and tremor dominant subtypes? PLoS One 8:e55193
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Jacobs JV, Horak FB (2007) Cortical control of postural responses. J Neural Transm 114:1339–1348
Jankovic J et al (1990) Variable expression of Parkinson’s disease: a base-line analysis of the DATATOP cohort. The Parkinson Study Group. Neurology 40:1529–1534
Kegelmeyer DA, Kloos AD, Thomas KM, Kostyk SK (2007) Reliability and validity of the Tinetti Mobility Test for individuals with Parkinson disease. Phys Ther 87:1369–1378
Kelly VE et al (2015) Association of cognitive domains with postural instability/gait disturbance in Parkinson’s disease. Parkinsonism Relat Disord 21:692–697
Lewis SJ, Barker RA (2009) A pathophysiological model of freezing of gait in Parkinson’s disease. Parkinsonism Relat Disord 15:333–338
Long XY et al (2008) Default mode network as revealed with multiple methods for resting-state functional MRI analysis. J Neurosci Methods 171:349–355
Middleton FA, Strick PL (2001) Cerebellar projections to the prefrontal cortex of the primate. J Neurosci 21:700–712
Mito Y, Yoshida K, Yabe I, Makino K, Tashiro K, Kikuchi S, Sasaki H (2006) Brain SPECT analysis by 3D-SSP and phenotype of Parkinson’s disease. J Neurol Sci 241:67–72
Mure H et al (2011) Parkinson’s disease tremor-related metabolic network: characterization, progression, and treatment effects. Neuroimage 54:1244–1253
Playford ED, Jenkins IH, Passingham RE, Nutt J, Frackowiak RS, Brooks DJ (1992) Impaired mesial frontal and putamen activation in Parkinson’s disease: a positron emission tomography study. Ann Neurol 32:151–161
Power JD, Barnes KA, Snyder AZ, Schlaggar BL, Petersen SE (2012) Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 59:2142–2154
Rosenberg-Katz K, Herman T, Jacob Y, Giladi N, Hendler T, Hausdorff JM (2013) Gray matter atrophy distinguishes between Parkinson disease motor subtypes. Neurology 80:1476–1484
Sage MD, Almeida QJ (2010) A positive influence of vision on motor symptoms during sensory attention focused exercise for Parkinson’s disease. Mov Disord 25:64–69
Sambataro F, Murty VP, Callicott JH, Tan HY, Das S, Weinberger DR, Mattay VS (2010) Age-related alterations in default mode network: impact on working memory performance. Neurobiol Aging 31:839–852
Schepens B, Stapley P, Drew T (2008) Neurons in the pontomedullary reticular formation signal posture and movement both as an integrated behavior and independently. J Neurophysiol 100:2235–2253
Shumway-Cook A, Brauer S, Woollacott M (2000) Predicting the probability for falls in community-dwelling older adults using the timed up & go test. Phys Ther 80:896–903
Tessitore A et al (2012a) Resting-state brain connectivity in patients with Parkinson’s disease and freezing of gait. Parkinsonism Relat Disord 18:781–787
Tessitore A et al (2012b) Default-mode network connectivity in cognitively unimpaired patients with Parkinson disease. Neurology 79:2226–2232
Timmermann L, Gross J, Dirks M, Volkmann J, Freund HJ, Schnitzler A (2003) The cerebral oscillatory network of parkinsonian resting tremor. Brain 126:199–212
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic review of levodopa dose equivalency reporting in parkinson’s disease. Mov Disord 25:2649–2685
Uc EY, Rizzo M, Anderson SW, Qian S, Rodnitzky RL, Dawson JD (2005) Visual dysfunction in Parkinson disease without dementia. Neurology 65:1907–1913
Zhang J, Wei L, Hu X, Xie B, Zhang Y, Wu GR, Wang J (2015) Akinetic-rigid and tremor-dominant Parkinson’s disease patients show different patterns of intrinsic brain activity. Parkinsonism Relat Disord 21:23–30
Zuo XN et al (2013) Toward reliable characterization of functional homogeneity in the human brain: preprocessing, scan duration, imaging resolution and computational space. Neuroimage 65:374–386
Acknowledgments
This work was supported by the Natural science foundation of Jiangsu Province (No. BK20141494), the Jiangsu Provincial Personnel Department “the Great of Six Talented Man Peak” Project (No. 2014-WSN-013), the University Natural Science Research Project in Jiangsu Province (No. 13KJB32009), the Opening Project of Jiangsu Key Laboratory of Neurodegeneration (No. SJ11KF01), the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and the Science and Technology Project of Jiangsu Bureau of Traditional Chinese Medicine (No. YB2015163).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
S. Jiang, M. Wang and L. Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Jiang, S., Wang, M., Zhang, L. et al. Regional homogeneity alterations differentiate between tremor dominant and postural instability gait difficulty subtypes of Parkinson’s disease. J Neural Transm 123, 219–229 (2016). https://doi.org/10.1007/s00702-015-1490-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-015-1490-5