Abstract
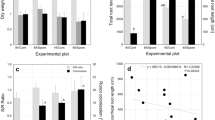
Despite the economic and ecological importance of Hydnum species, in vitro synthesis of ectomycorrhizae of this genus has not been reported due to difficulties in establishing pure cultures. We inoculated pure cultures of 12 monokaryotic and 3 dikaryotic mycelial strains of an undescribed Hydnum albidum-like species on roots of axenic Pinus densiflora seedlings to synthesize ectomycorrhizae and to evaluate their mycorrhiza-forming ability. Six months after inoculation, both monokaryotic and dikaryotic strains formed ectomycorrhizae with Hartig net hyphae at the root cortex. Monokaryotic and dikaryotic strains exhibited similar morpho-anatomical characteristics of ectomycorrhizae, with the exception for clamped septa of emanating and outer mantle hyphae in the latter. Between monokaryotic and descendant dikaryotic strains, there were no significant differences in number of mycorrhizae in pine seedlings, whereas monokaryotic strains showed a greater total number of root tips and lower colonization rates than the descendant dikaryotic strains. These results indicate that both monokaryotic and dikaryotic mycelia of the H. albidum-like species can form mycorrhizae under axenic condition, and that can be applied toward the cultivation of hedgehog mushrooms.





Similar content being viewed by others
References
Agerer R (1991) Characterization of ectomycorrhiza. Methods Microbiol 23:25–73. https://doi.org/10.1016/S0580-9517(08)70172-7
Agerer R (1999) Never change a functional successful principle: the evolution of Boletales s. 1. (Hymenomycetes, Basidiomycota) as seen from below-ground features. Sendteria 6:5–91
Agerer R (2006) Fungal relationships and structural identity of their ectomycorrhizae. Mycol Prog 5:67–107. https://doi.org/10.1007/s11557-006-0505-x
Agerer R, Kraigher H, Javornik B (1996) Identification of ectomycorrhizae of Hydnum rufescens on Norway spruce and the variability of the ITS region of H. rufescens and H. repandum (Basidiomycetes). Nova Hedwigia 63:183–194
Arnolds E (1995) Conservation and management of natural populations of edible fungi. Can J Bot 73:987–998. https://doi.org/10.1139/b95-349
Danell E, Camacho FJ (1997) Successful cultivation of the golden chanterelle. Nature 385:303. https://doi.org/10.1038/385303a0
De Román M, Boa E (2004) Collection, marketing and cultivation of edible fungi in Spain. Micol Apl Int 16:25–33
Debaud JC, Gay G, Prevost A, Lei J, Dexheimer J (1988) Ectomycorrhizal ability of genetically different homokaryotic and dikaryotic mycelia of Hebeloma cylindrosporum. New Phytol 108:323–328. https://doi.org/10.1111/j.1469-8137.1988.tb04169.x
Dennis JJ (1985) Effect of pH and temperature on in vitro growth of ectomycorrhizal fungi. Can For Serv Pacific For Centre, Inf Rep BC-X-273, 1–19
Di Battista C, Selosse MA, Bouchard D, Stenström E, Le Tacon F (1996) Variations in symbiotic efficiency, phenotypic characters and ploidy level among different isolates of the ectomycorrhizal basidiomycete Laccaria bicolor strain S 238. Mycol Res 100:1315–1324. https://doi.org/10.1016/S0953-7562(96)80058-X
Donk MA (1933) Revision der niederländischen Homobasidiomycetae-Aphyllophoraceae II. Meded van de Ned Mycol Ver 22:1–278
Donk MA (1956) The generic names proposed for Hymenomycetes V “Hydnaceae” (Continuation). Taxon 5:69–80. https://doi.org/10.2307/1217317
Douhan GW, Vincenot L, Gryta H, Selosse M-A (2011) Population genetics of ectomycorrhizal fungi: from current knowledge to emerging directions. Fungal Biol 115:569–597. https://doi.org/10.1016/J.FUNBIO.2011.03.005
Endo N, Gisusi S, Fukuda M, Yamada A (2013) In vitro mycorrhization and acclimatization of Amanita caesareoides and its relatives on Pinus densiflora. Mycorrhiza 23:303–315. https://doi.org/10.1007/s00572-012-0471-x
Endo N, Kawamura F, Kitahara R, Sakuma D, Fukuda M, Yamada A (2014) Synthesis of Japanese Boletus edulis ectomycorrhizae with Japanese red pine. Mycoscience 55:405–416. https://doi.org/10.1016/j.myc.2013.11.008
Feng B, Wang XH, Ratkowsky D et al (2016) Multilocus phylogenetic analyses reveal unexpected abundant diversity and significant disjunct distribution pattern of the Hedgehog Mushrooms (Hydnum L.). Sci Rep 6:1–11. https://doi.org/10.1038/srep25586
Fries N (1984) Spore germination in the higher Basidiomycetes. Proc Plant Sci 93:205–222. https://doi.org/10.1007/BF03053077
Fries N (1987) Ecological and evolutionary aspects of spore germination in the higher basidiomycetes. Trans Br Mycol Soc 88:1–7. https://doi.org/10.1016/S0007-1536(87)80179-1
Gay G, Debaud JC (1987) Genetic study on indole-3-acetic acid production by ectomycorrhizal Hebeloma species: inter- and intraspecific variability in homo- and dikaryotic mycelia. Appl Microbiol Biotechnol 26:141–146. https://doi.org/10.1007/BF00253898
Harrington TJ, Mitchell DT (2002) Characterization of Dryas octopetala ectomycorrhizas from limestone karst vegetation, western Ireland. Can J Bot 80:970–982. https://doi.org/10.1139/b02-082
Kawai M, Yamahara M, Ohta A (2008) Bipolar incompatibility system of an ectomycorrhizal basidiomycete, Rhizopogon rubescens. Mycorrhiza 18:205–210. https://doi.org/10.1007/s00572-008-0167-4
Kikuchi K, Matsushita N, Suzuki K, Hogetsu T (2007) Flavonoids induce germination of basidiospores of the ectomycorrhizal fungus Suillus bovinus. Mycorrhiza 17:563–570. https://doi.org/10.1007/s00572-007-013
Kropp BR, Fortin JA (1986) Formation and regeneration of protoplasts from the ectomycorrhizal basidiomycete Laccaria bicolor. Can J Bot 64:1224–1226. https://doi.org/10.1139/b86-167
Kropp BR, McAfee BJ, Fortin JA (1987) Variable loss of ectomycorrhizal ability in monokaryotic and dikaryotic cultures of Laccaria bicolor. Can J Bot 65:500–504. https://doi.org/10.1139/b87-062
Lamhamedi MS, Fortin JA, Kope HH, Kropp BR (1990) Genetic variation in ectomycorrhiza formation by Pisolithus arhizus on Pinus pinaster and Pinus banksiana. New Phytol 115:689–697. https://doi.org/10.1111/j.1469-8137.1990.tb00501.x
Maas Geestranus R (1971) Hydnaceous fungi of the eastern old world. North-Holland Publishing Company, Netherland, Amsterdam
Massicotte HB, Peterson RL, Ashford AE (1987) Ontogeny of Eucalyptus pilularis – Pisolithus tinctorius ectomycorrhizae. I. Light microscopy and scanning electron microscopy. Can J Bot 65:1927–1939. https://doi.org/10.1139/b87-264
Moncalvo J-M, Nilsson RH, Koster B et al (2006) The cantharelloid clade: dealing with incongruent gene trees and phylogenetic reconstruction methods. Mycologia 98:937–948. https://doi.org/10.1080/15572536.2006.11832623
Niskanen T, Liimatainen K, Nuytinck J et al (2018) Identifying and naming the currently known diversity of the genus Hydnum, with an emphasis on European and north American taxa. Mycologia 110:890–918. https://doi.org/10.1080/00275514.2018.1477004
Pine EM, Hibbett DS, Donoghue MJ (1999) Phylogenetic relationships of cantharelloid and clavarioid homobasidiomycetes based on mitochondrial and nuclear rDNA sequences. Mycologia 91:944–963. https://doi.org/10.2307/3761626
Poitou N, Mamoun M, Ducamp M, Delmas J (1984) Après le bolet granuleux, le lactaire délicieux obtenu en fructification au champ à partir de plants mycorhizés. PHM Rev Hortic 244:65–68
Sawada K, Wan J, Oda K, Nakano S, Aimi T, Shimomura N (2014) Variability in nucleus number in basidiospore isolates of Rhizopogon roseolus and their ability to form ectomycorrhizas with host pine roots. Mycol Prog 13:745–751. https://doi.org/10.1007/s11557-013-0957-8
Sitta N, Floriani M (2008) Nationalization and globalization trends in the wild mushroom commerce of Italy with emphasis on porcini (Boletus edulis and allied species). Econ Bot 62:307–322. https://doi.org/10.1007/s12231-008-9037-4
Smith SE, Read DJ (2008) Mycorrhizal Symbiosis, 3rd edn. Academic Press.
Sugawara R, Yamada A, Kawai M, Sotome K, Maekawa N, Nakagiri A, Endo N (2019) Establishment of monokaryotic and dikaryotic isolates of Hedgehog mushrooms (Hydnum repandum and related species) from basidiospores. Mycoscience 60:201–209. https://doi.org/10.1016/j.myc.2019.02.007
Swenie RA, Baroni TJ, Matheny PB (2018) Six new species and reports of Hydnum (Cantharellales) from eastern North America. MycoKeys 72:35–72. https://doi.org/10.3897/mycokeys.42.27369
Wang Y, Cummings N, Guerin-Laguette A (2012) Cultivation of basidiomycete edible ectomycorrhizal mushrooms: tricholoma, lactarius, and rhizopogon. Springer, Berlin, Heidelberg, pp 281–304
Wong KKY, Montpetit D, Piché Y, Lei J (1990a) Root colonization by four closely related genotypes of the ectomycorrhizal basidiomycete Laccaria bicolor (Maire) Orton – comparative studies using electron microscopy. New Phytol 116:669–679. https://doi.org/10.1111/j.1469-8137.1990.tb00553.x
Wong KKY, Piché Y, Fortin JA (1990b) Differential development of root colonization among four closely related genotypes of ectomycorrhizal Laccaria bicolor. Mycol Res 94:876–884. https://doi.org/10.1016/S0953-7562(09)81300-2
Yamada A (2002) Utility of mycorrhizal mushrooms as food resources in Japan (in Japanese). Journal Fac Agric Shinshu Univ 38:1–17
Yamada A, Katsuya K (1995) Mycorrhizal association of isolates from sporocarps and ectomycorrhizas with Pinus densiflora seedlings. Mycoscience 36:315–323. https://doi.org/10.1007/BF02268607
Yamada A, Ogura T, Ohmasa M (2001) Cultivation of mushrooms of edible ectomycorrhizal fungi associated with Pinus densiflora by in vitro mycorrhizal synthesis. Mycorrhiza 11:67–81. https://doi.org/10.1007/s005720000093
Yamada A, Endo N, Murata H, Ohta A, Fukuda M (2014) Tricholoma matsutake Y1 strain associated with Pinus densiflora shows a gradient of in vitro ectomycorrhizal specificity with Pinaceae and oak hosts. Mycosciecne 55:27–34. https://doi.org/10.1016/j.myc.2013.05.0042013.05.004
Acknowledgements
We thank Ms. Sachiko Ueta, Ms. Ayako Eriguchi, Ms. Mizuki Yokono, Ms. Masako Oka, and Ms. Kiko Hirata for cryopreservation of Hydnum strains. We also thank the staff of the Division of Instrumental Research, Research Center for Supports to Advanced Science, Shinshu University, for their technical support regarding DNA sequencing.
Funding
This study was supported in part by a Grant-in-Aid for JSPS KAKENHI Grant No. JP20J20884 (Ryo Sugawara) and JP19K06088 (Akira Nakagiri) from the Ministry of Education, Culture, Sports, Science and Technology, Japan. This study was also supported in part by a general research grant (Naoki Endo) from the Institute for Fermentation, Osaka (IFO) and a research fund from the president of Tottori University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sugawara, R., Sotome, K., Maekawa, N. et al. Mycorrhizal synthesis, morpho-anatomical characterization of mycorrhizae, and evaluation of mycorrhiza-forming ability of Hydnum albidum–like species using monokaryotic and dikaryotic cultures. Mycorrhiza 31, 349–359 (2021). https://doi.org/10.1007/s00572-021-01024-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-021-01024-7