Abstract
Purpose
We evaluated the preventive effect of the antioxidant edaravone (EDR) on chemotherapy-induced alopecia (CIA) to improve quality of life in cancer patients.
Methods
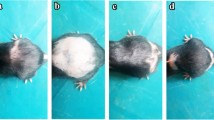
Hair loss was induced by intraperitoneally administering cyclophosphamide (CPA, 75 mg/kg) to rats, and topically applying EDR ointment (100 mg/day) once daily for 16 days (when hair loss starts) or 21 days (just before hair growth). The rats were divided into four groups: control group (without CPA or EDR), EDR 0% group (CPA + EDR 0%), EDR 3% group (CPA + EDR 3%), and EDR 30% group (CPA + EDR 30%). The prevention of CIA was evaluated by the hair coverage score (five levels from 0 to 4). Furthermore, we measured the size of the hair follicle area and the expression levels of insulin-like growth factor (IGF)-1 mRNA in dermal papilla cells.
Results
The EDR 3% and EDR 30% groups exhibited higher hair coverage scores than the EDR 0% group on day 16 and day 21. On day 16, the hair follicle area in the EDR 3% and EDR 30% groups was significantly larger than that in the EDR 0% group. Furthermore, IGF-1 expression levels in the EDR 3% group were significantly higher than those in the EDR 0% group. On day 21, no significant difference was observed in hair follicle area or IGF-1 mRNA levels among the groups.
Conclusion
Our results show that EDR administration lessened hair loss due to CPA in a dose-independent manner above doses of 3%, suggesting potential applications beside chemotherapy.




Similar content being viewed by others
Data Availability
Not applicable.
Code availability
Not applicable.
References
Hesketh PJ, Batchelor D, Golant M, Lyman GH, Rhodes N, Yardley D (2004) Chemotherapy-induced alopecia: psychosocial impact and therapeutic approaches. Support Care Cancer 12:543–549. https://doi.org/10.1007/s00520-003-0562-5
Lemieux J, Maunsell E, Provencher L (2008) Chemotherapy-induced alopecia and effects on quality of life among women with breast cancer: a literature review. Psychooncology 17:317–328. https://doi.org/10.1002/pon.1245
Nangia J, Wang T, Osborne C, Niravath P, Otte K, Papish S, Holmes F, Abraham J, Lacouture M, Courtright J, Paxman R, Rude M, Hilsenbeck S, Osborne CK, Rimawi M (2017) Effect of a scalp cooling device on alopecia in women undergoing chemotherapy for breast cancer: the SCALP randomized clinical trial. J Am Med Assoc 317:596–605. https://doi.org/10.1001/jama.2016.20939
Komen MMC, Smorenburg CH, van den Hurk CJG, Nortier JWR (2013) Factors influencing the effectiveness of scalp cooling in the prevention of chemotherapy-induced alopecia. Oncologist 18:885–891. https://doi.org/10.1634/theoncologist.2012-0332
Singh K, Bhori M, Kasu YA, Bhat G, Marar T (2018) Antioxidants as precision weapons in war against cancer chemotherapy induced toxicity - exploring the armoury of obscurity. Saudi Pharm J 26:177–190. https://doi.org/10.1016/j.jsps.2017.12.013
Sato T, Mizuno K, Ishii F (2008) In vitro metabolism study of edaravone in Wistar and hairless rat skin. Biol Pharm Bull 31:1150–1154. https://doi.org/10.1248/bpb.31.1150
Stenn KS, Paus R (2001) Controls of hair follicle cycling. Physiol Rev 81:449–494. https://doi.org/10.1152/physrev.2001.81.1.449
Onaolapo AY, Adebayo AA, Onaolapo OJ (2018) Oral phenytoin protects against experimental cyclophosphamide-chemotherapy induced hair loss. Pathophysiology 25:31–39. https://doi.org/10.1016/j.pathophys.2017.12.001
Hagiwara S, Uchida T, Koga H, Inomata M, Yoshizumi F, Moriyama M, Kitano S, Noguchi T (2011) The α-lipoic acid derivative sodium zinc dihydrolipoylhistidinate reduces chemotherapy-induced alopecia in a rat model: A pilot study. Surg Today 41:693–697. https://doi.org/10.1007/s00595-010-4481-z
Paus R, Bori H, Eichmüller S, Czarnetzki BM (1994) Chemotherapy-induced alopecia in mice. Induction by cyclophosphamide, inhibition by cyclosporine A, and modulation by dexamethasone. Am J Pathol 144:719–734
Schilli MB, Paus R, Menrad A (1998) Reduction of intrafollicular apoptosis in chemotherapy-induced alopecia by topical calcitriol-analogs. J Invest Dermatol 111:598–604. https://doi.org/10.1046/j.1523-1747.1998.00350.x
Yano K, Brown LF, Detmar M (2001) Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest 107:409–417. https://doi.org/10.1172/JCI11317
Müller-Röver S, Handjiski B, van der Veen C, Eichmüller S, Foitzik K, McKay IA, Stenn KS, Paus R (2001) A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J Invest Dermatol 117:3–15. https://doi.org/10.1046/j.0022-202x.2001.01377.x
Hendrix S, Bori H, Peters EMJ, Paus R (2005) A guide to assessing damage response pathways of the hair follicle: lessons from cyclophosphamide-induced alopecia in mice. J Invest Dermatol 125:42–51. https://doi.org/10.1111/j.0022-202X.2005.23787.x
Cook JJ, Haynes KM, Werther GA (1988) Mitogenic effects of growth hormone in cultured human fibroblasts. Evidence for action via local insulin-like growth factor I production. J Clin Invest 81:206–212. https://doi.org/10.1172/JCI113296
Rudman SM, Philpott MP, Thomas GA, Kealey T (1997) The role of IGF-I in human skin and its appendages: morphogen as well as mitogen? J Invest Dermatol 109:770–777. https://doi.org/10.1111/1523-1747.ep12340934
Philpott MP, Sanders DA, Kealey T (1994) Effects of insulin and insulin-like growth factors on cultured human hair follicles: IGF-I at physiologic concentrations is an important regulator of hair follicle growth in vitro. J Invest Dermatol 102:857–861. https://doi.org/10.1111/1523-1747.ep12382494
Weger N, Schlake T (2005) IGF-I signalling controls the hair growth cycle and the differentiation of hair shafts. J Invest Dermatol 125:873–882. https://doi.org/10.1111/j.0022-202X.2005.23946.x
Schlake T, Beibel M, Weger N, Boehm T (2004) Major shifts in genomic activity accompany progression through different stages of the hair cycle. Gene Expr Patterns 4:141–152. https://doi.org/10.1016/j.modgep.2003.09.009
Wikramanayake TC, Amini S, Simon J, Mauro LM, Elgart G, Schachner LA, Jimenez JJ (2012) A novel rat model for chemotherapy-induced alopecia. Clin Exp Dermatol 37:284–289. https://doi.org/10.1111/j.1365-2230.2011.04239.x
Trüeb RM (2009) Chemotherapy-Induced Alopecia. Semin Cutan Med Surg 28:11–14. https://doi.org/10.1016/j.sder.2008.12.001
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Katsuaki Yoneda, Miyu Fujii, Aoi Imaoka, Remi Kobayashi, Ryoya Hayashi, and Takumi Tsuji. The first draft of the manuscript was written by Katsuaki Yoneda and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Approval was obtained from the ethics committee of Setsunan University.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yoneda, K., Fujii, M., Imaoka, A. et al. Preventive effect of edaravone ointment on cyclophosphamide-chemotherapy induced alopecia. Support Care Cancer 29, 6127–6134 (2021). https://doi.org/10.1007/s00520-021-06189-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-021-06189-7