Abstract
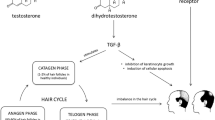
Chemotherapy-induced alopecia (CIA) is probably one of the most shocking and psychologically disturbing side effects for oncological patients. Hair loss occurs because chemotherapy targets all rapidly dividing cells, healthy cells, and cancer cells. Chemotherapy may cause hair loss all over the human body, not just on the scalp. Hair follicles, the structures in the skin filled with tiny blood vessels that make hair, are some of the fastest-growing cells in the body. Hair follicles divide every 23–72 hours. The chemo not only works against cancer cells but it also destroys hair cells. To the best of our knowledge, no guidelines are available about CIA management. The present review may provide evidence that the plant extract is highly effective in hair growth stimulating activity with high anti-hair fall properties, which can be used as a hair-promoting agent for post-chemotherapy alopecia. Investigation on the therapeutic properties of Nardostachys jatamansi as a potential hair growth promoter will provide the scientific basis for developing nutraceuticals to manage chemotherapy-induced alopecia (CIA)––hair falls for cancer patients. This study may be a scientific tool for understanding the complete therapeutic and nutritional properties of Nardostachys jatamansi for developing natural compounds for CIA.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
JAMA Oncol (2017) 3(8):1147. https://doi.org/10.1001/jamaoncol.2017.1026
JAMA Oncol (2021) 7(7):1080. https://doi.org/10.1001/jamaoncol.2021.0896
Dunnill CJ, Al-Tameemi W, Collett A, Haslam IS, Georgopoulos NT (2018) A clinical and biological guide for understanding chemotherapy-induced alopecia and its prevention. Oncologist 23:84–96
Sahu R, Dhongade HJ, Pandey A, Sahu P, Sahu V, Patel D, Kashyap P (2016) Medicinal Properties of Nardostachys jatamansi (A Review). Orient J Chem 32:32(2)
Chatterjee B, Basak U, Datta J, Banerji A, Neuman TP (2005) Studies on the Chemical Constituents of N. jatamansi DC [Valerianaceae]. Cheminform 36:17
Schneider MR, Schmidt-Ullrich R, Paus R (2009) The hair follicle is a dynamic miniorgan. Curr Biol 19:R132–R142
Hendrix S, Handjiski B, Peters EM, Paus RJ (2005) Invest Dermatol 125(1):42–51
Hendrix S, Handjiski B, Peters EM, Paus R (2005) A guide to assessing damage response pathways of the hair follicle: lessons from cyclophosphamide-induced alopecia in mice. J Invest Dermatol 125:42–51
Paus R, Haslam IS, Sharov AA, Botchkarev VA (2013) Pathobiology of chemotherapy-induced hair loss. Lancet Oncol 14:e50–e59
Botchkarev VA (2003) Molecular mechanisms of chemotherapy-induced hair loss. J Investing Dermatol Symp Proc 8(1):72–75. https://doi.org/10.1046/j.1523-1747.2003.12175.x
Mill P, Mo R, Fu H et al (2003) Sonic hedgehog-dependent activation of Gli2 is essential for embryonic hair follicle development. Genes Dev 17:282–294
Loomis CA, Joyner AL (2011) Nerve-derived sonic hedgehog defines a niche for hair follicle stem cells capable of becoming epidermal stem cells. Cell Stem Cell. 8:552–565
Bleiker TO, Nicolaou N, Traulsen J, Hutchinson PE (2005) ‘Atrophic telogen effluvium’ from cytotoxic drugs and a randomized controlled trial to investigate the possible protective effect of pretreatment with a topical vitamin D analogue in humans. Br J Dermatol 153:103–112
Pirmez R, Pineiro-Maceira J, Sodre CT (2013) Exclamation marks and other trichoscopy signs of chemotherapy-induced alopecia. Australas J Dermatol 54:129–132
Kowalska-Olędzka E, Slowinska M, Rakowska A et al (2012) ‘Black dots’ seen under trichoscopy are not specific for alopecia areata. Clin Exp Dermatol 37:615–619
Rossi A, Caterina Fortuna M, Caro G et al (2018) Monitoring chemotherapy-induced alopecia with trichoscopy [published online ahead of print July 11, 2018]. J Cosmet Dermatol 18:575. https://doi.org/10.1111/jocd.12687
Miteva M, Tosti A (2015) Flame hair. Skin Appendage Disord 1:105–109
Cheng H, Hong B, Zhou L et al (2012) Mitomycin C potentiates TRAIL-induced apoptosis through p53-independent upregulation of death receptors: evidence for the role of c-Jun N-terminal kinase activation. Cell Cycle 11:3312–3323
Haslam I, Xie G, Zhou G et al (2015) Shh signaling regulates the damage response of murine and human hair follicles in chemotherapy-induced alopecia. J Invest Dermatol 135:S7
Adapted alf Paus, Haslam IS, Sharov AA, Botchkarev VA (2013) Pathobiology of chemotherapy-induced hair loss. The Lancet Oncology 14(2):e50–e59
Hughes BT, Sidorova J, Swanger J, Monnat RJ Jr, Clurman BE (2013) Proc Natl Acad Sci U S A 110(22):8954–8959. https://doi.org/10.1073/pnas.1302927110. Epub 2013 May 13
Trueb RM (2009) Chemotherapy-induced alopecia. Semin Cutan Med Surg 28:11–14
Jimenez JJ, Yunis AA (1992) Protection from chemotherapy-induced alopecia by 1,25-dihydroxyvitamin D3. Cancer Res 52:5123–5125
Hidalgo M, Rinaldi D, Medina G et al (1999) A phase I trial of topical topitriol (calcitriol, 1,25-dihydroxyvitamin D3) to prevent chemotherapy-induced alopecia. Anti-Cancer Drugs 10:393–395
Bleiker TO, Nicolaou N, Traulsen J et al (2005) ‘Atrophic telogen effluvium’ from cytotoxic drugs and a randomized controlled trial to investigate the possible protective effect of pretreatment with a topical vitamin D analogue in humans. Br J Dermatol 153:103–112
Lotfi-Jam K, Carey M, Jefford M, Schofield P, Charleson C, Aranda S (2008) Nonpharmacologic strategies for managing common chemotherapy adverse effects: a systematic review. J Clin Oncol 26:5618–5629
Cline BW (1984) Prevention of chemotherapy-induced alopecia: a review of the literature. Cancer Nurs 7:221–228
Hussein AM, Jimenez JJ, McCall CA, Yunis AA (1990) Science. 249(4976):1564–1546. https://doi.org/10.1126/science.2218498. PMID: 2218498
Hussein AM (1995) Int J Dermatol. 34(7):470–3. https://doi.org/10.1111/j.1365-4362.1995.tb00612.x. PMID: 7591409
Hussein AM (1993) South Med J. 86(5):489–96. https://doi.org/10.1097/00007611-199305000-00001.PMID: 8488392
Imanishi H, Tsuruta D, Tateishi C, Sugawara K, Paus R, Tsuji T et al (2010) Laminin-511, inducer of hair growth, is down-regulated and its suppressor in hair growth, laminin-332 up-regulated in chemotherapy-induced alopecia. J Dermatol Sci 58:43–54
You J, Gao F, Tang H, Peng F, Jia L, Huang K, Chow K, Zhao J, Liu H, Lin Y, Chen J (2019) A medicinal and edible formula YH0618 ameliorates the toxicity induced by doxorubicin via regulating the expression of Bax/Bcl-2 and FOXO4. J Cancer 10(16):3665–3677
You JS et al (2016) Effect of YH0618 soup on chemotherapy-induced toxicity in patients with cancer who have completed chemotherapy: study protocol for a randomized controlled trial. Trials 17(1):354
Chan YC et al (2009) Nanonized black soybean enhances immune response in senescence-accelerated mice. Int J Nanomedicine 4:27–35
Yim JH et al (2009) Antinociceptive and anti-inflammatory effects of ethanolic extracts of Glycine max (L.) Merr and Rhynchosia nulubilis seeds. Int J Mol Sci 10(11):4742–4753
Kusano S, Abe H, Tamura H (2001) Isolation of antidiabetic components from white-skinned sweet potato (Ipomoea batatas L.). Biosci Biotechnol Biochem 65(1):109–114
Macduff C, Mackenzie T, Hutcheon A et al (2003) The effectiveness of scalp cooling in preventing alopecia for patients receiving epirubicin and docetaxel. Eur J Cancer Care 12:154–161
Mols F, Van Den Hurk CJ, Vingerhoets AJJM et al (2009) Scalp cooling to prevent chemotherapy-induced hair loss: practical and clinical considerations. Support Care Cancer 17:181–189
Kennedy M, Packard R, Grant M et al (1983) The effects of using chemocap on occurrence of chemotherapy-induced alopecia. Oncol Nurs Forum 10:19–24
Lovejoy NC (1979) Preventing hair loss during adriamycin therapy. Cancer Nurs 2:117–121
Rodriguez R, Machiavelli M, Leone B et al (1994) Minoxidil (mx) as a prophylaxis of doxorubicin-induced alopecia. Ann Oncol 5:769–770
Granai CO, Frederickson H, Gajewski W et al (1991) The use of minoxidil to attempt to prevent alopecia during chemotherapy for gynecologic malignancies. Eur J Gynaecol Oncol 12:129–132
Villani C, Inghirami P, Pietrangeli D (1986) Prevention by hypothermic cap of antiblastic induced-alopecia. Eur J Gynaecol Oncol 7:15–17
Edelstyn GA, MacDonald M, MacRae KD (1977) Doxorubicin-induced hair loss and possible modification by scalp cooling. Lancet 2:253–254
Peck HJ, Mitchell H, Stewart AL (2000) Evaluating the efficacy of scalp cooling using the penguin cold cap system to reduce alopecia in patients undergoing chemotherapy for breast cancer. Eur J Oncol Nurs 4:246–248
Jadhav VM, Throat RM, Kadam VJ, Kamble SS (2009) Herbal Anxiolyte, Nardostachs jatamansi. J Pharm Res 2(8):1208–1211
Yeap SK, Rahman MBA, Alitheen NB, Ho WY et al (2011) Evaluation of immunomodulatory effect, selection of the correct targets for Immunostimulation study. Am J Immunol 7(2):17–23
Inui S, Nakajima T, Itami S (2010) Coudability hairs: a revisited sign of alopecia areata assessed by trichoscopy. Clin Exp Dermatol 35(4):361–365
Peralta L, Morais P (2012) Photoletter to the editor: the friar tuck sign in trichotillomania. J Dermatol Case Rep 6(2):63–64
Wu MY, Li J. (2021) CMAJ. 193(4):E126. https://doi.org/10.1503/cmaj.200894
Grevelman EG, Breed WP (2005) Prevention of chemotherapy-induced hair loss by scalp cooling. Ann Oncol 16:352–358
Al-Tameemi W, Dunnill C, Hussain O et al (2014) Use of in vitro human keratinocyte models to study the effect of cooling on chemotherapy drug-induced cytotoxicity. Toxicol In Vitro 28:1366–1376
McGarvey EL, Baum LD, Pinkerton RC, Rogers LM (2001) Psychological sequelae and alopecia among women with cancer. Cancer Pract 9:283–289
Botchkarev V, Sharov AA, Syska W, Maurer M, Gilchrest BA (2002) Involvement of p53 and Fas in hair follicle apoptosis in human skin/SCID model for chemotherapy-induced hair loss. J Invest Dermatol 119:286
Food and H.H.S. Drug Administration (2016) Medical devices; general and plastic surgery devices; classification of the scalp cooling system to reduce the likelihood of chemotherapy-induced alopecia. Final order. Fed Regist 81(29):7452–7454
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Azhagu Saravana Babu, P., Vajiha Aafrin, B., Goyali, S., Geethika, M., Sugumari, V., Sukumar, M. (2023). Therapeutic Properties of Nardostachys jatamansi and Its Applications in Post-Chemotherapy-Induced Hair Loss in Cancer Patients. In: Arunachalam, K., Yang, X., Puthanpura Sasidharan, S. (eds) Bioprospecting of Tropical Medicinal Plants. Springer, Cham. https://doi.org/10.1007/978-3-031-28780-0_24
Download citation
DOI: https://doi.org/10.1007/978-3-031-28780-0_24
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-28779-4
Online ISBN: 978-3-031-28780-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)