Abstract
Key message
Functional stomata can be generated by in vitro ABA applications. This mitigates the desiccation responses of Persian walnut leaves in their early stage of ex vitro exposure. This finding paves the way for discovering a solution for the uncontrollable water loss problem of in vitro-generated walnut plantlets.
Abstract
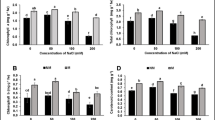
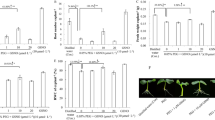
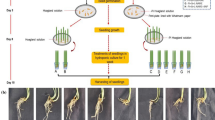
Most in vitro-generated plants are susceptible to wilting when they are transferred to an ex vitro environment, which is characterized by a higher evaporative nature. In this study, two stomatal closing stimuli [abscisic acid (ABA) and polyethylene glycol (PEG)] were added to in vitro culture medium of Persian walnut (Juglans regia L.) to evaluate their leaf ability to control water loss during exposure to an ex vitro environment. Different concentrations of PEG (0, 3 and 5%) and ABA (0, 1 and 10 µM) were tested, and their effects were evaluated in two separate experiments. In the first experiment, the proline concentration decreased in the leaves grown on media containing PEG. In contrast, glycine betaine (GB) increased only in leaves grown on a culture medium with 5% PEG. Leaf osmotic potential (ψs) increased parallel to the increase in PEG concentration. Adding PEG to the culture medium caused a decrease in stomatal aperture, and, as a result, the water conservation capacity of the in vitro-grown leaves was increased during detached leaf ex vitro desiccation. However, PEG-induced osmotic stress caused a negative influence on the morpho-physiologic characteristics and chlorophyll content of in vitro-grown nodal segments. In the second experiment, ABA was tested in the culture medium with the eventual aim of making in vitro leaves lose less water when they were transferred to an ex vitro environment. ABA (especially 10 µM) improved the leaf water conservation capacity during ex vitro desiccation of detached leaves. Small stomata having narrow apertures were increased in number by supplementing the cultures with ABA, although the length of shoots decreased with its use; no serious side effect was observed on morpho-physiologic parameters. Our findings are significant because they can ultimately be useful for preventing the uncontrollable water loss problem of in vitro-generated walnut plantlets.









Similar content being viewed by others
References
Aguilar M, Espadas F, Coello J, Maust B, Trejo C, Robert M, Santamaria J (2000) The role of abscisic acid in controlling leaf water loss, survival and growth of micropropagated Tagetes erecta plants when transferred directly to the field. J Exp Bot 51:1861–1866. https://doi.org/10.1093/jexbot/51.352.1861
Al-Khayri JM, Al-Bahrany AM (2004) Growth, water content and proline accumulation in drought stressed callus of date palm. Biol Plant 48:105–108. https://doi.org/10.1023/B:BIOP.0000024283.74919.4c
Ali-Ahmad M, Hughes HG, Safadi F (1998) Studies on stomatal function, epicuticular wax and stem-root transition region of polyethylene glycol-treated and nontreated in vitro grape plantlets. In vitro Cell Dev Biol Plant 34:1–7. https://doi.org/10.1007/BF02823115
Aliniaeifard S, Malcolm Matamoros P, van Meeteren U (2014) Stomatal malfunctioning under low VPD conditions: induced by alterations in stomatal morphology and leaf anatomy or in the ABA signaling? Physiol Plant 152:688–699. https://doi.org/10.1111/ppl.12216
Aliniaeifard S, van Meeteren U (2013) Can prolonged exposure to low VPD disturb the ABA signalling in stomatal guard cells? J Exp Bot 64:3551–3566. https://doi.org/10.1093/jxb/ert192
Aliniaeifard S, van Meeteren U (2014) Natural variation in stomatal response to closing stimuli among Arabidopsis thaliana accessions after exposure to low VPD as a tool to recognise the mechanism of disturbed stomatal functioning. J Exp Bot 65:6529–6542. https://doi.org/10.1093/jxb/eru370
Asayesh ZM, Vahdati K, Aliniaeifard S (2017a) Investigation of physiological components involved in low water conservation capacity of in vitro walnut plants. Sci Hortic 224:1–7. https://doi.org/10.1016/j.scienta.2017.04.023
Asayesh ZM, Vahdati K, Aliniaeifard S, Askari N (2017b) Enhancement of ex vitro acclimation of walnut plantlets through modification of stomatal characteristics in vitro. Sci. Hortic. 220:114–121. https://doi.org/10.1016/j.scienta.2017.03.045
Ashraf M, Foolad M (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Bates L, Waldren R, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Brainerd K, Fuchigami L (1981) Acclimatization of aseptically cultured apple plants to low relative humidity [Water stress, hardiness]. J Am Soc Hortic Sci 33:388–392
Bray EA (2002) Abscisic acid regulation of gene expression during water-deficit stress in the era of the Arabidopsis genome. Plant Cell Environ 25:153–161. https://doi.org/10.1046/j.13653040.2002.00746.x
Brito G, Costa A, Fonseca HMAC, Santos CV (2003) Response of Olea europaea ssp. maderensis in vitro shoots exposed to osmotic stress. Sci Hortic 97:411–417. https://doi.org/10.1046/j.13653040.2002.00746.x
Chandra S, Bandopadhyay R, Kumar V, Chandra R (2010) Acclimatization of tissue cultured plantlets: from laboratory to land. J Biotechnol Lett 32:1199–1205. https://doi.org/10.1007/s10529-010-0290-0
Chen C (2004) Humidity in plant tissue culture vessels. Biosys Eng 88:231–241. https://doi.org/10.1016/j.biosystemseng.2004.02.007
Colon-Guasp W, Nell TA, Kane ME, Barrett JE (1996) Effects of abscisic acid on ex vitro acclimatization of Aronia arbutifolia (L.). Pers J Am Soc Hortic Sci 121:101–104
Conner AJ, Thomas MB (1982) Re-establishing plantlets from tissue culture a review. Comb Proc Int Pl Prop Soc 31:342–357
Cui YY, Hahn EJ, Kozai T, Paek KY (2000) Number of air exchanges, sucrose concentration, photosynthetic photon flux, and differences in photoperiod and dark period temperatures affect growth of Rehmannia glutinosa plantlets in vitro. Plant Cell Tissus Org 62:219–226. https://doi.org/10.1023/A:1006412321864
Dami I, Hughes H (1995) Leaf anatomy and water loss of in vitro PEG-treated ‘Valiant’grape. Plant Cell Tissus Org 42:179–184. https://doi.org/10.1007/BF00034236
Dami I, Hughes H (1997) Effects of PEG-induced water stress on in vitro hardening of ‘Valiant’grape. Plant Cell Tissus Org 47:97–101. https://doi.org/10.1007/BF02318944
Dias M, Pinto G, Santos C (2011) Acclimatization of micropropagated plantlets induces an antioxidative burst: a case study with Ulmus minor Mill. Photosynthetica 49:259–266. https://doi.org/10.1007/s11099-011-0028-9
Driver JA, Kuniyuki AH (1984) In vitro propagation of paradox walnut rootstocks. Hortic Sci Publ J Am Soc Hortic Sci 19:506–509
Estrada-Luna AA, Davies FT, Egilla JN (2001) Physiological changes and growth of micropropagated chile ancho pepper plantlets during acclimatization and post-acclimatization. Plant Cell Tiss Org 66:17–24. https://doi.org/10.1023/A:1010606430060
Farsi M, Fatahi Moghadam M, Zamani Z, Hassani D (2018) Effects of scion cultivar, rootstock age and hormonal treatment on minigrafting of Persian walnut. Int J Hortic Sci Technol 5(2):185–197. https://doi.org/10.22059/ijhst.2018.255460.233
Fuchigami L, Cheng T, Soeldner A (1981) Abaxial transpiration and water loss in aseptically cultured plum. Plant Cell Tissues Org 106:519–522
Grieve C, Grattan S (1983) Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 70:303–307. https://doi.org/10.1007/BF02374789
Grout B, Aston M (1977) Transplanting of cauliflower plants regenerated from meristem culture. I. Water loss and water transfer related to changes in leaf wax and to xylem regeneration. Hortic Res 17:1–7
Kotakis C, Kyzeridou A, Manetas Y (2014) Photosynthetic electron flow during leaf senescence: evidence for a preferential maintenance of photosystem I activity and increased cyclic electron flow. Photosynthetica 52:413–420
Hassankhah A, Vahdati K, Lotfi M, Mirmasoumi M, Preece J, Assareh M (2014) Effects of ventilation and sucrose concentrations on the growth and plantlet anatomy of micropropagated Persian walnut plants. Int J Hortic Sci Technol 1(2):111–120. https://doi.org/10.22059/ijhst.2014.52781
Hazarika B (2003) Acclimatization of tissue-cultured plants. Curr Sci 85:1704–1712
Hazarika B (2006) Morpho-physiological disorders in in vitro culture of plants. Sci Hortic 108:105–120. https://doi.org/10.1016/j.scienta.2006.01.038
Hazarika B, Nagaraju V, Parthasarathy V, Bhowmik G (2001) Biochemical basis of acclimatization of micropropagated plantlets—a review. Agric Rev 22:65–67
Hronkova M, Zahradnickova H, Simkova M, Simek P, Heydova A (2003) The role of abscisic acid in acclimation of plants cultivated in vitro to ex vitro conditions. Biol Plant 46:535–541. https://doi.org/10.1023/A:1024811527499
Ivanov AG, Krol M, Maxwell D, Huner NPA (1995) Abscisic acid induced protection against photoinhibition of PSII correlates with enhanced activity of the xanthophyll cycle. FEBS Lett 371:61–64. https://doi.org/10.1016/0014-5793(95)00872-7
Jiang F, Hartung W (2008) Long-distance signalling of abscisic acid (ABA): the factors regulating the intensity of the ABA signal. J Exp Bot 59:37–43. https://doi.org/10.1093/jxb/erm127
Kozai T, Kubota C, Jeong BR (1997) Environmental control for the large-scale production of plants through in vitro techniques. Plant Cell Tissue Org. 51:49–56. https://doi.org/10.1023/A:1005809518371
Lawlor D (1970) Absorption of polyethylene glycols by plants and their effects on plant growth. New Phytol 69:501–513. https://doi.org/10.1111/j.1469-8137.1970.tb02446.x
Maggon R, Singh BD (1995) Promotion of adventitious bud regeneration by ABA in combination with BAP in epicotyl and hypocotyl explants of sweet orange (Citrus sinensis L. Osbeck). Sci Hortic 63:123–128. https://doi.org/10.1016/0304-4238(95)00780-W
Majada JP, Luz Centeno M, Feito I, Fernandez B, Sanchez-Tames R (1998) Stomatal and cuticular traits on carnation tissue culture under different ventilation conditions. Plant Growth Regul 25:113–121. https://doi.org/10.1023/B:GROW.0000009706.70078.1c
Martinez JP, Lutts S, Schanck A, Bajji M, Kinet JM (2004) Is osmotic adjustment required for water stress resistance in the Mediterranean shrub Atriplex halimus L? J Plant Physiol 161:1041–1051. https://doi.org/10.1016/j.jplph.2003.12.009
Michel BE, Kaufmann MR (1973) The osmotic potential of polyethylene glycol 6000. J Plant Physiol 51:914–916. https://doi.org/10.1104/pp.51.5.914
Mohamed M, Harris P, Henderson J (2000) In vitro selection and characterization of a drought tolerant clone of tagetes minuta. Plant Sci 159:213–222. https://doi.org/10.1016/S01689452(00)00339-3
Mondal W, Dey B, Choudhuri M (1985) Proline accumulation as a reliable indicator of monocarpic senescence in rice cultivars. Experientia 41:346–348
Payghamzadeh K, Kazemitabar SK (2011) In vitro propagation of walnut—a review. Afr J Biotechnol 10:290–311. https://doi.org/10.5897/AJB10.324
Pospisilova J (1996) Hardening by abscisic acid of tobacco plantlets grown in vitro. Biol Plant 38:605–609
Pospisilova J, Haisel D, Synkova H, Batkova-Spoustova P (2009a) Improvement of ex vitro transfer of tobacco plantlets by addition of abscisic acid to the last subculture. Biol Plant 53:617–624. https://doi.org/10.1007/s10535-009-0113-0
Pospisilova J, Synkova H, Haisel D, Batkova P (2009b) Effect of abscisic acid on photosynthetic parameters during ex vitro transfer of micropropagated tobacco plantlets. Biol Plant 53:11–20. https://doi.org/10.1007/s10535-009-0003-5
Pospisilova J, Synkova H, Haisel D, Semoradova S (2007) Acclimation of Plantlets to Ex vitro Conditions: Effects of Air Humidity, Irradiance, CO2 Concentration and Abscisic Acid (a Review). Acta Hortic 748:29–38. https://doi.org/10.17660/ActaHortic.2007.748.2
Pospisilova J, Ticha I, Kadlecek P, Haisel D, Plzakova S (1999) Acclimatization of micropropagated plants to ex vitro conditions. Biol Plant 42:481–497
Preece J, Sutter E (1991) Acclimatization of micropropagated plants to the greenhouse and field. In: Debergh PC, Zimmerman RH (eds) Micropropagation. Springer, Dordrecht, pp 71–93. https://doi.org/10.1007/978-94-009-2075-0_5
Safadi F, Hughes H (1990) Comparison of the diffusive resistance of Polyethylene glycol treated and non-treated tissue culture Tobacco plantlets. Hortic Sci 25:1105–1105
Santamaria J, Davies W, Atkinson C (1993) Stomata of micropropagated Delphinium plants respond to ABA, CO2, light and water potential, but fail to close fully. J Exp Bot 44:99–107. https://doi.org/10.1093/jxb/44.1.99
Shackel K, Novello V, Sutter E (1990) Stomatal function and cuticular conductance in whole tissue-cultured apple shoots. J Am Soc Hortic Sci 115:468–472
Sharp RE, LeNoble ME, Else MA, Thorne ET, Gherardi F (2000) Endogenous ABA maintains shoot growth in tomato independently of effects on plant water balance: evidence for an interaction with ethylene. J Exp Bot 51:1575–1584. https://doi.org/10.1093/jexbot/51.350.1575
Short K, Warburton J, Roberts A (1987) In vitro hardening of cultured Cauliflower and Chrysanthemum plantlets to humidity. Acta Hortic 212(50):329–340. https://doi.org/10.17660/ActaHortic.1987.212.50
Sivritepe N, Erturk U, Yerlikaya C, Turkan I, Bor M, Ozdemir F (2008) Response of the cherry rootstock to water stress induced in vitro. Biol Plant 52:573–576. https://doi.org/10.1007/s10535-008-0114-4
Slavik B (1974) Methods of studying plant water relations. Chapman and Hall, London
Smart CM (1994) Gene expression during leaf senescence. New Phytol 126:419–448
Sutter E (1988) Stomatal and cuticular water loss from apple, cherry, and sweetgum plants after removal from in vitro culture. J Am Soc Hortic Sci 113:234–238
Vahdati K, Asayesh ZM, Aliniaeifard S, Leslie C (2017) Improvement of ex vitro desiccation through elevation of CO2 concentration in the atmosphere of culture vessels during in vitro growth. Hortic Sci 52:1006–1012. https://doi.org/10.21273/HORTSCI11922-17
Wang CY, Cheng SH, Kao CH (1982) Senescence of rice leaves: Vii. proline accumulation in senescing excised leaves. Plant Physiol 69:1348–1349
Woltering EJ (1989) Effect of the gaseous composition on development of gerbera plantlets grown in-vitro. Acta Hortic 261:377–383
Zaid A, Hughes H (1995a) A comparison of stomatal function and frequency of in vitro polyethylene glycol treated and greenhouse grown plants of date palm, Phoenix dactylifera L. Trop Agric 72:130–134
Zaid A, Hughes H (1995b) In vitro acclimatization of date palm (Phoenix dactylifera L.) plantlets: a quantitative comparison of epicuticular leaf wax as a function of polyethylene glycol treatment. Plant Cell Rep 15:111–114
Zhang J, Jia W, Yang J, Ismail AM (2006) Role of ABA in integrating plant responses to drought and salt stresses. Field Crops Res. 97:111–119. https://doi.org/10.1016/j.fcr.2005
Zhang L, Becker D (2015) Connecting proline metabolism and signaling pathways in plant senescence. Front Plant Sci. https://doi.org/10.3389/fpls.2015.00552
Ziv M, Schwartz A, Fleminger D (1987) Malfunctioning stomata in vitreous leaves of carnation (Dianthus caryophyllus) plants propagated in vitro; implications for hardening. Plant Sci 52:127–134. https://doi.org/10.1016/0168-9452(87)90114-2
Acknowledgements
The authors appreciate Iran National Science Foundation (INSF), Center of Excellence of Walnut Improvement and Technology of Iran, and University of Tehran for their support.
Author information
Authors and Affiliations
Contributions
Sasan Aliniaeifard: Made substantial contributions to the conception and design of the experiments, the analysis and interpretation of data and finally the writing of the paper. Zeinab Maleki Asayesh: Performed the experiments, prepared the data and helped in writing the paper. John Driver: Gave technical advice and edited the paper scientifically. Kourosh Vahdati: Made substantial contributions to the conception and design of the experiments, revised the paper and finally approved of the manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by S.A. Merkle.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aliniaeifard, S., Asayesh, Z.M., Driver, J. et al. Stomatal features and desiccation responses of Persian walnut leaf as caused by in vitro stimuli aimed at stomatal closure. Trees 34, 1219–1232 (2020). https://doi.org/10.1007/s00468-020-01992-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-020-01992-x