Abstract
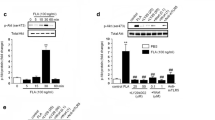
Microglial cells are a major source of pro-inflammatory cytokines during central nervous system (CNS) inflammation. They can develop a pro-inflammatory M1 phenotype and an anti-inflammatory M2 phenotype. Shifting the phenotype from M1 to M2 might be an important mechanism to overcome CNS inflammation and to prevent or reduce neuronal damage. Here, we demonstrate that the anti-inflammatory protein trefoil factor 3 (TFF3) is secreted by astrocytes and that its transcription is significantly reduced after incubation with lipopolysaccharide (LPS). Moreover, we demonstrate that microglial cells cultured in the presence of TFF3 show reduced expression and secretion of pro-inflammatory cytokines after LPS stimulation.



Similar content being viewed by others
References
Arnold P, Mojumder D, Detoledo J, Lucius R, Wilms H (2014) Pathophysiological processes in multiple sclerosis: focus on nuclear factor erythroid-2-related factor 2 and emerging pathways. Clin Pharmacol Adv Appl 6:35–42
Arnold P, Schmidt F, Prox J, Zunke F, Pietrzik C, Lucius R, Becker-Pauly C (2015) Calcium negatively regulates meprin beta activity and attenuates substrate cleavage. FASEB J 29:3549–3557
Blackburn D, Sargsyan S, Monk PN, Shaw PJ (2009) Astrocyte function and role in motor neuron disease: a future therapeutic target? Glia 57:1251–1264
Chaiyarit P, Chayasadom A, Wara-Aswapati N, Hormdee D, Sittisomwong S, Nakaresisoon S, Samson MH, Pitiphat W, Giraud AS (2012) Trefoil factors in saliva and gingival tissues of patients with chronic periodontitis. J Periodontol 83:1129–1138
Chalaris A, Garbers C, Rabe B, Rose-John S, Scheller J (2011) The soluble interleukin 6 receptor: generation and role in inflammation and cancer. Eur J Cell Biol 90:484–494
Chatterton DE, Nguyen DN, Bering SB, Sangild PT (2013) Anti-inflammatory mechanisms of bioactive milk proteins in the intestine of newborns. Int J Biochem Cell Biol 45:1730–1747
Deng YY, Lu J, Ling EA, Kaur C (2010) Microglia-derived macrophage colony stimulating factor promotes generation of proinflammatory cytokines by astrocytes in the periventricular white matter in the hypoxic neonatal brain. Brain Pathol 20:909–925
Devine DA, High AS, Owen PJ, Poulsom R, Bonass WA (2000) Trefoil factor expression in normal and diseased human salivary glands. Hum Pathol 31:509–515
Franco R, Fernandez-Suarez D (2015) Alternatively activated microglia and macrophages in the central nervous system. Prog Neurobiol 131:65–86
Guppy NJ, El-Bahrawy ME, Kocher HM, Fritsch K, Qureshi YA, Poulsom R, Jeffery RE, Wright NA, Otto WR, Alison MR (2012) Trefoil factor family peptides in normal and diseased human pancreas. Pancreas 41:888–896
Heinrichsdorff J, Olefsky JM (2012) Fetuin-A: the missing link in lipid-induced inflammation. Nat Med 18:1182–1183
Hinz M, Schwegler H, Chwieralski CE, Laube G, Linke R, Pohle W, Hoffmann W (2004) Trefoil factor family (TFF) expression in the mouse brain and pituitary: changes in the developing cerebellum. Peptides 25:827–832
Hoffmann W, Jagla W, Wiede A (2001) Molecular medicine of TFF-peptides: from gut to brain. Histol Histopathol 16:319–334
Itoh H, Tomita M, Uchino H, Kobayashi T, Kataoka H, Sekiya R, Nawa Y (1996) cDNA cloning of rat pS2 peptide and expression of trefoil peptides in acetic acid-induced colitis. Biochem J 318:939–944
Jagla W, Wiede A, Hinz M, Dietzmann K, Gülicher D, Gerlach KL, Hoffmann W (1999) Secretion of TFF-peptides by human salivary glands. Cell Tissue Res 298:161–166
Jagla W, Wiede A, Dietzmann K, Rutkowski K, Hoffmann W (2000) Co-localization of TFF3 peptide and oxytocin in the human hypothalamus. FASEB J 14:1126–1131
Kim JH, Min KJ, Seol W, Jou I, Joe EH (2010) Astrocytes in injury states rapidly produce anti-inflammatory factors and attenuate microglial inflammatory responses. J Neurochem 115:1161–1171
Kinoshita K, Taupin DR, Itoh H, Podolsky DK (2000) Distinct pathways of cell migration and antiapoptotic response to epithelial injury: structure-function analysis of human intestinal trefoil factor. Mol Cell Biol 20:4680–4690
Liebner S, Czupalla CJ, Wolburg H (2011) Current concepts of blood–brain barrier development. Int J Dev Biol 55:467–476
Matsuoka Y, Pascall JC, Brown KD (1999) Quantitative analysis reveals differential expression of mucin (MUC2) and intestinal trefoil factor mRNAs along the longitudinal axis of rat intestine. Biochim Biophys Acta 1489:336–344
Muskett FW, May FE, Westley BR, Feeney J (2003) Solution structure of the disulfide-linked dimer of human intestinal trefoil factor (TFF3): the intermolecular orientation and interactions are markedly different from those of other dimeric trefoil proteins. Biochemistry 42:15139–15147
Nakagawa Y, Chiba K (2014) Role of microglial m1/m2 polarization in relapse and remission of psychiatric disorders and diseases. Pharmaceuticals 7:1028–1048
Paulsen FP, Woon CW, Varoga D, Jansen A, Garreis F, Jäger K, Amm M, Podolsky DK, Steven P, Barker NP, Sel S (2008) Intestinal trefoil factor/TFF3 promotes re-epithelialization of corneal wounds. J Biol Chem 283:13418–13427
Perry VH, Gordon S (1988) Macrophages and microglia in the nervous system. Trends Neurosci 11:273–277
Prat A, Biernacki K, Wosik K, Antel JP (2001) Glial cell influence on the human blood–brain barrier. Glia 36:145–155
Probst JC, Zetzsche T, Weber M, Theilemann P, Skutella T, Landgraf R, Jirikowski GF (1996) Human intestinal trefoil factor is expressed in human hypothalamus and pituitary: evidence for a novel neuropeptide. FASEB J 10:1518–1523
Qin J, Yang X, Mi J, Wang J, Hou J, Shen T, Li Y, Wang B, Li X, Zhu W (2014) Enhanced antidepressant-like effects of the macromolecule trefoil factor 3 by loading into negatively charged liposomes. Int J Nanomedicine 9:5247–5257
Rickert U, Grampp S, Wilms H, Spreu J, Knerlich-Lukoschus F, Held-Feindt J, Lucius R (2014) Glial cell line-derived neurotrophic factor family members reduce microglial activation via inhibiting p38MAPKs-mediated inflammatory responses. J Neurodegener Dis 2014:369468
Rösler S, Haase T, Claassen H, Schulze U, Schicht M, Riemann D, Brandt J, Wohlrab D, Müller-Hilke B, Goldring MB, Sel S, Varoga D, Garreis F, Paulsen FP (2010) Trefoil factor 3 is induced during degenerative and inflammatory joint disease, activates matrix metalloproteinases, and enhances apoptosis of articular cartilage chondrocytes. Arthritis Rheum 62:815–825
Roy S, Bonfield T, Tartakoff AM (2013) Non-apoptotic toxicity of Pseudomonas aeruginosa toward murine cells. PLoS One 8:e54245
Santos Silva E dos, Ulrich M, Döring G, Botzenhart K, Gött P (2000) Trefoil factor family domain peptides in the human respiratory tract. J Pathol 190:133–142
Schulze U, Sel S, Paulsen FP (2010) Trefoil factor family peptide 3 at the ocular surface. A promising therapeutic candidate for patients with dry eye syndrome? Dev Ophthalmol 45:1–11
Schwarz H, Jagla W, Wiede A, Hoffmann W (2001) Ultrastructural co-localization of TFF3-peptide and oxytocin in the neural lobe of the porcine pituitary. Cell Tissue Res 305:411–416
Shi HS, Yin X, Song L, Guo QJ, Luo XH (2012) Neuropeptide trefoil factor 3 improves learning and retention of novel object recognition memory in mice. Behav Brain Res 227:265–269
Suemori S, Lynch-Devaney K, Podolsky DK (1991) Identification and characterization of rat intestinal trefoil factor: tissue- and cell-specific member of the trefoil protein family. Proc Natl Acad Sci U S A 88:11017–11021
Thim L (1997) Trefoil peptides: from structure to function. Cell Mol Life Sci 53:888–903
Vincent VA, Tilders FJ, Van Dam AM (1997) Inhibition of endotoxin-induced nitric oxide synthase production in microglial cells by the presence of astroglial cells: a role for transforming growth factor beta. Glia 19:190–198
Wiede A, Jagla W, Welte T, Köhnlein T, Busk H, Hoffmann W (1999) Localization of TFF3, a new mucus-associated peptide of the human respiratory tract. Am J Respir Crit Care Med 159:1330–1335
Wilms H, Rosenstiel P, Romero-Ramos M, Arlt A, Schäfer H, Seegert D, Kahle PJ, Odoy S, Claasen JH, Holzknecht C, Brandenburg LO, Deuschl G, Schreiber S, Kirik D, Lucius R (2009) Suppression of MAP kinases inhibits microglial activation and attenuates neuronal cell death induced by alpha-synuclein protofibrils. Int J Immunopathol Pharmacol 22:897–909
Wilms H, Sievers J, Rickert U, Rostami-Yazdi M, Mrowietz U, Lucius R (2010) Dimethylfumarate inhibits microglial and astrocytic inflammation by suppressing the synthesis of nitric oxide, IL-1beta, TNF-alpha and IL-6 in an in-vitro model of brain inflammation. J Neuroinflammation 7:30
Wolburg H, Paulus W (2010) Choroid plexus: biology and pathology. Acta Neuropathol 119:75–88
Wu JF, Zhang J, Xue G, Zhang HQ (2014) Expression and localization of trefoil factor family genes in rat submandibular glands. Biotech Histochem 89:424–432
Zhang BH, Yu HG, Sheng ZX, Luo HS, Yu JP (2003) The therapeutic effect of recombinant human trefoil factor 3 on hypoxia-induced necrotizing enterocolitis in immature rat. Regul Pept 116:53–60
Acknowledgments
We acknowledge R. Sprang, R. Worm, M. Grell, M. Kölln, K. Neblung-Masuhr and G. Schäfer for superb technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Philipp Arnold, Uta Rickert and Ann-Kristin Helmers contributed equally to this work.
We thank the medical faculty of the Kiel University for financial support (to P.A.).
Rights and permissions
About this article
Cite this article
Arnold, P., Rickert, U., Helmers, AK. et al. Trefoil factor 3 shows anti-inflammatory effects on activated microglia. Cell Tissue Res 365, 3–11 (2016). https://doi.org/10.1007/s00441-016-2370-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-016-2370-5