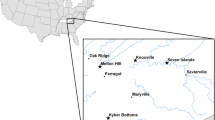
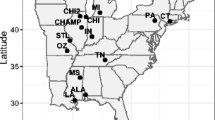
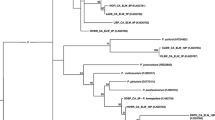
Abstract
Avian blood parasites from the genera Plasmodium, Haemoproteus, and Leucocytozoon (Haemosporida) affect hosts in numerous ways. They influence species interactions, host behavior, reproductive success, and cause pathology and mortality in birds. The Great Lakes region of North America has extensive aquatic and wetland habitat and supports a diverse vector community. Here we describe the community of bird-infecting Haemosporida in southwest Michigan and their host associations by measuring parasite prevalence, diversity, and host breadth across a diverse community of avian hosts. Over 700 songbirds of 55 species were screened for Haemosporida infection across southwest Michigan, including 11 species that were targeted for larger sample sizes. In total, 71 parasite lineages infected over 40% of birds. Of these, 42 were novel, yet richness estimates suggest that approximately half of the actual parasite diversity in the host community was observed despite intensive sampling of multiple host species. Parasite prevalence varied among parasite genera (7–24%) and target host species (0–85%), and parasite diversity was consistently high across most target species. Host breadth varied widely across the most prevalent parasite lineages, and we detected around 60% of host species richness for these parasite lineages. We report many new lineages and novel host-parasite associations, but substantial parasite diversity remains undiscovered in the Midwest.



Similar content being viewed by others
References
Adler PH, Currie DC, Wood DM (2004) The black flies (Simuliidae) of North America. Cornell University Press, Ithaca, USA
Asghar M, Hasselquist D, Hansson B, Zehtindjiev P, Westerdahl H, Bensch S (2015) Hidden costs of infection: chronic malaria accelerates telomere degradation and senescence in wild birds. Science 347(6220):436–438. https://doi.org/10.1126/science.1261121
Astudillo VG, Hernández SM, Kistler WM, Boone SL, Lipp EK, Shrestha S, Yabsley MJ (2013) Spatial, temporal, molecular, and intraspecific differences of haemoparasite infection and relevant selected physiological parameters of wild birds in Georgia, USA. Int J Parasitol Parasites Wildl 2:178–189. https://doi.org/10.1016/j.ijppaw.2013.04.005
Atkinson CT, Dusek RJ, Woods KL, Iko WM (2000) Pathogenicity of avian malaria in experimentally-infected Hawaii Amakihi. J Wildlife Dis 36(2):197–204. https://doi.org/10.7589/0090-3558-36.2.197
Bantock T, Prys-Jones R, Lee PL (2008) New and improved molecular sexing methods for museum bird specimens. Mol Ecol Res 8(3):519–528. https://doi.org/10.1111/j.1471-8286.2007.01999.x
Beadell JS, Gering E, Austin J, Dumbacher JP, Peirce MA, Pratt TK, Atkinson CT, Fleischer RC (2004) Prevalence and differential host-specificity of two avian blood parasite genera in the Australo-Papuan region. Mol Ecol 13(12):3829–3844. https://doi.org/10.1111/j.1365-294X.2004.02363.x
Beadell JS, Ishtiaq F, Covas R, Melo M, Warren BH, Atkinson CT, Bensch S, Graves GR, Jhala YV, Peirce MA (2006) Global phylogeographic limits of Hawaii’s avian malaria. Proc R Soc B Biol Sci 273(1604):2935–2944. https://doi.org/10.1098/rspb.2006.3671
Bensch S, Stjernman M, Hasselquist D, Örjan Ö, Hannson B, Westerdahl H, Pinheiro RT (2000) Host specificity in avian blood parasites: a study of Plasmodium and Haemoproteus mitochondrial DNA amplified from birds. Proc R Soc B Biol Sci 267(1452):1583–1589. https://doi.org/10.1098/rspb.2000.1181
Bensch S, Pérez-Tris J, Waldenströum J, Hellgren O (2004) Linkage between nuclear and mitochondrial DNA sequences in avian malaria parasites: multiple cases of cryptic speciation? Evolution 58(7):1617–1621. https://doi.org/10.1111/j.0014-3820.2004.tb01742.x
Bensch S, Hellgren O, Pérez-Tris J (2009) MalAvi: a public database of malaria parasites and related haemosporidians in avian hosts based on mitochondrial cytochrome b lineages. Mol Ecol Res 9(5):1353–1358. https://doi.org/10.1111/j.1755-0998.2009.02692.x
Bernotienė R, Palinauskas V, Iezhova T, Murauskaitė D, Valkiūnas G (2016) Avian haemosporidian parasites (Haemosporida): a comparative analysis of different polymerase chain reaction assays in detection of mixed infections. Exp Parasitol 163:31–37. https://doi.org/10.1016/j.exppara.2016.01.009
Bonter DN, Donovan TM (2009) Characteristics of important stopover locations for migrating birds: remote sensing with radar in the Great Lakes basin. Conserv Biol 23(2):440–448. https://doi.org/10.1111/j.1523-1739.2008.01085.x
Borkent A, Grogan W (2009) Catalog of the New World biting midges north of Mexico (Diptera: Ceratopogonidae). Zootaxa 2273:1–48
Bosholn M, Fecchio A, Silveira P, Braga ÉM, Anciães M (2016) Effects of avian malaria on male behaviour and female visitation in lekking blue-crowned manakins. J Avian Biol 47(4):457–465. https://doi.org/10.1111/jav.00864
Breck JE (2004) Compilation of databases on Michigan Lakes. Michigan Department of Natural Resources. http://michiganlakes.msue.msu.edu/michigan_lakes. Accessed 6 Sept 2017
Bright E (2004) Aquatic insects of Michigan. Museum of Zoology Insect Division, School of Natural Resources and Environment. University of Michigan. http://www.aquaticinsects.org/. Accessed 10 Nov 2016
Cassani JR, Newson H (1980) An annotated list of mosquitoes reported from Michigan. Mosq News 40:356–368
Chagas CRF, Valkiūnas G, Nery CVC, Henrique PC, Gonzalez IHL, Monteiro EF, de Oliveira Guimarães L, Romano CM, Kirchgatter K (2013) Plasmodium (Novyella) nucleophilum from an Egyptian goose in São Paulo zoo, Brazil: microscopic confirmation and molecular characterization. Int J Parasitol Parasites Wildl 2:286–291. https://doi.org/10.1016/j.ijppaw.2013.09.008
Chao A, Colwell RK, Lin C-W, Gotelli NJ (2009) Sufficient sampling for asymptotic minimum species richness estimators. Ecology 90(4):1125–1133. https://doi.org/10.1890/07-2147.1
Chao A, Ma, KH, Hsieh, TC, Chiu, CH (2016) Spade R (Species-richness Prediction and Diversity Estimation in R): an R package in CRAN. https://CRAN.R-project.org/package=SpadeR. Accessed 29 April 2017
Cornelius EA, Davis AK, Altizer SA (2014) How important are hemoparasites to migratory songbirds? Evaluating physiological measures and infection status in three Neotropical migrants during stopover. Physiol Biochem Zool 87(5):719–728. https://doi.org/10.1086/677541
Darsie RF Jr, Ward RA (2005) Identification and geographical distribution of the mosquitoes of North America, north of Mexico. University Press of Florida, Gainesville, Florida
Drovetski SV, Aghayan SA, Mata VA, Lopes RJ, Mode NA, Harvey JA, Voelker G (2014) Does the niche breadth or trade-off hypothesis explain the abundance–occupancy relationship in avian Haemosporidia? Mol Ecol 23(13):3322–3329. https://doi.org/10.1111/mec.12744
Dubiec A, Podmokła E, Zagalska-Neubauer M, Drobniak SM, Arct A, Gustafsson L, Cichoń M (2016) Differential prevalence and diversity of haemosporidian parasites in two sympatric closely related non-migratory passerines. Parasitology 143(10):1320–1329. https://doi.org/10.1017/S0031182016000779
Durrant KL, Marra PP, Fallon SM, Colbeck GJ, Gibbs HL, Hobson KA, Norris DR, Bernik B, Lloyd VL, Fleischer RC (2008) Parasite assemblages distinguish populations of a migratory passerine on its breeding grounds. J Zool 274(4):318–326. https://doi.org/10.1111/j.1469-7998.2007.00387.x
Ellis VA, Collins MD, Medeiros MCI, Sari EHR, Coffey ED, Dickerson RC, Lugarini C, Stratford JA, Henry DR, Merrill L, Matthews AE, Hanson AA, Roberts JR, Joyce M, Kunkel MR, Ricklefs RE (2015) Local host specialization, host-switching, and dispersal shape the regional distributions of avian haemosporidian parasites. Proc Natl Acad Sci U S A 112(36):11294–11299. https://doi.org/10.1073/pnas.1515309112
Fast K, Walstrom VW, Outlaw DC (2016) Haemosporidian prevalence and parasitaemia in the Tufted Titmouse (Baelophus bicolor). J Parasitol 102(6):636–642. https://doi.org/10.1645/15-935
Fecchio A, Lima MR, Silveira P, Braga ÉM, Marini MÂ (2011) High prevalence of blood parasites in social birds from a neotropical savanna in Brazil. Emu 111(2):132–138. https://doi.org/10.1071/MU10063
Gonzalez AD, Matta NE, Ellis VA, Miller ET, Ricklefs RE, Gutierrez HR (2014) Mixed species flock, nest height, and elevation partially explain avian haemoparasite prevalence in Colombia. PLoS One 9(6):e100695. https://doi.org/10.1371/journal.pone.0100695
Gonzalez AD, Lotta IA, Garcia LF, Moncada LI, Matta NE (2015) Avian haemosporidians from Neotropical highlands: evidence from morphological and molecular data. Parasitol Int 64(4):48–59. https://doi.org/10.1016/j.parint.2015.01.007
Griffiths R, Double MC, Orr K, Dawson RJ (1998) A DNA test to sex most birds. Mol Ecol 7(8):1071–1075. https://doi.org/10.1046/j.1365-294x.1998.00389.x
Hall RJ, Brown LM, Altizer S (2016) Modeling vector-borne disease risk in migratory animals under climate change. Integr Comp Biol 56(2):353–364. https://doi.org/10.1093/icb/icw049
Hamer GL, Chaves LF, Anderson TK, Kitron UD, Brawn JD, Ruiz MO, Loss SR, Walker ED, Goldberg TL (2011) Fine-scale variation in vector host use and force of infection drive localized patterns of West Nile virus transmission. PLoS One 6(8):e23767. https://doi.org/10.1371/journal.pone.0023767
Harrell FE (2016) Hmisc: Harrel Miscellaneous. https://cran.r-project.org/package=Hmisc. Accessed 5 June 2017
Hellgren O, Waldenstrom J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon, Plasmodium, and Haemoproteus from avian blood. J Parasitol 90(4):797–802. https://doi.org/10.1645/ge-184r1
Hellgren O, Waldenstrom J, Pérez-Tris J, Szollosi E, Hasselquist D, Krizanauskiene A, Ottosson U, Bensch S (2007) Detecting shifts of transmission areas in avian blood parasites—a phylogenetic approach. Mol Ecol 16(11):1281–1290. https://doi.org/10.1111/j.1365-294X.2007.03277.x
Huang X, Dong L, Zhang C, Zhang Y (2015) Genetic diversity, temporal dynamics, and host specificity in blood parasites of passerines in north China. Parasitol Res 114(12):4513–4520. https://doi.org/10.1007/s00436-015-4695-5
Jin S, Yang L, Danielson P, Homer C, Fry J, Xian G (2013) A comprehensive change detection method for updating the National Land Cover Database to circa 2011. Remote Sens Environ 132:159–175. https://doi.org/10.1016/j.rse.2013.01.012
Jovani R, Tella JL (2006) Parasite prevalence and sample size: misconceptions and solutions. Trends Parasitol 22(5):214–218. https://doi.org/10.1016/j.pt.2006.02.011
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28(12):1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kelly TR, MacGillivray HL, Sarquis-Adamson Y, Watson MJ, Hobson KA, MacDougall-Shackleton EA (2016) Seasonal migration distance varies with natal dispersal and predicts parasitic infection in song sparrows. Behav Ecol Sociobiol 70(11):1857–1866. https://doi.org/10.1007/s00265-016-2191-2
Kimura M, Dhondt AA, Lovette IJ (2006) Phylogeographic structuring of Plasmodium lineages across the North American range of the house finch (Carpodacus mexicanus). J Parasitol 92(5):1043–1049. https://doi.org/10.1645/GE-639R.1
Knowles SCL, Wood MJ, Sheldon BC (2010) Context-dependent effects of parental effort on malaria infection in a wild bird population, and their role in reproductive trade-offs. Oecologia 164(1):87–97. https://doi.org/10.1007/s00442-010-1706-1
Krizanauskiene A, Pérez-Tris J, Palinauskas V, Hellgren O, Bensch S, Valkiūnas G (2010) Molecular phylogenetic and morphological analysis of haemosporidian parasites (Haemosporida) in a naturally infected European songbird, the blackcap Sylvia atricapilla, with description of Haemoproteus pallidulus sp. nov. Parasitology 137(02):217–227. https://doi.org/10.1017/S0031182009991235
Kulma K, Low M, Bensch S, Qvarnström A (2013) Malaria infections reinforce competitive asymmetry between two Ficedula flycatchers in a recent contact zone. Mol Ecol 22(17):4591–4601. https://doi.org/10.1111/mec.12409
Lafferty KD (2009) The ecology of climate change and infectious diseases. Ecology 90(4):888–900. https://doi.org/10.1890/08-0079.1
Laine A (2009) Role of coevolution in generating biological diversity: spatially divergent selection trajectories. J Exp Bot 60(11):2957–2970. https://doi.org/10.1093/jxb/erp168
Levin I, Zwiers P, Deem S, Geest E, Higashiguchi J, Iezhova T, Jimenez-Uzcategui G, Kim D, Morton J, Perlut N (2013) Multiple lineages of avian malaria parasites (Plasmodium) in the Galapagos Islands and evidence for arrival via migratory birds. Conserv Biol 27(6):1366–1377. https://doi.org/10.1111/cobi.12127
Loiseau C, Harrigan RJ, Cornel AJ, Guers SL, Dodge M, Marzec T, Carlson JS, Seppi B, Sehgal RNM (2012) First evidence and predictions of Plasmodium transmission in Alaskan bird populations. PLoS One 7(9):e44729. https://doi.org/10.1371/journal.pone.0044729
Lutz HL, Hochachka WM, Engel JI, Bell JA, Tkach VV, Bates JM, Hackett SJ, Weckstein JD (2015) Parasite prevalence corresponds to host life history in a diverse assemblage of afrotropical birds and haemosporidian parasites. PLoS One 10(4):e0121254. https://doi.org/10.1371/journal.pone.0121254
Maddison WP, Maddison DR (2017) Mesquite: a modular system for evolutionary analysis. Version 3.2. http://mesquiteproject.org. Accessed 8 July 2017
Mantilla JS, González AD, Valkiūnas G, Moncada LI, Matta NE (2013) Description and molecular characterization of Plasmodium (Novyella) unalis sp. nov. from the Great Thrush (Turdus fuscater) in highland of Colombia. Parasitol Res 112(12):4193–4204. https://doi.org/10.1007/s00436-013-3611-0
Martinsen ES, McInerney N, Brightman H, Ferebee K, Walsh T, McShea WJ, Forrester TD, Ware L, Joyner PH, Perkins SL, Latch EK, Yabsley MJ, Schall JJ, Fleischer RC (2016) Hidden in plain sight: Cryptic and endemic malaria parasites in North American white-tailed deer (Odocoileus virginianus). Science Advances 2(2):e1501486. https://doi.or/10.1126/sciadv.1501486
Marzal A, Ricklefs RE, Valkiūnas G, Albayrak T, Arriero E, Bonneaud C, Czirják GA, Ewen J, Hellgren O, Hořáková D, Iezhova TA, Jensen H, Križanauskienė A, Lima MR, de Lope F, Magnussen E, Martin LB, Møller AP, Palinauskas V, Pap PL, Pérez-Tris J, Sehgal RNM, Soler M, Szöllősi E, Westerdahl H, Zetindjiev P, Bensch S (2011) Diversity, loss, and gain of malaria parasites in a globally invasive bird. PLoS One 6(7):e21905. https://doi.org/10.1371/journal.pone.0021905
Matthews AE, Ellis VA, Hanson AA, Roberts JR, Ricklefs RE, Collins MD (2016) Avian haemosporidian prevalence and its relationship to host life histories in eastern Tennessee. J Ornithol 157(2):533–548. https://doi.org/10.1007/s10336-015-1298-y
Medeiros MC, Ricklefs RE, Brawn JD, Hamer GL (2015) Plasmodium prevalence across avian host species is positively associated with exposure to mosquito vectors. Parasitology 142(13):1612–1620. https://doi.org/10.1017/S0031182015001183
Merino S, Moreno J, Vasquez RA, Martinez J, Sanchez-Monsalvez I, Estades CF, Ippi S, Sabat P, Rozzi R, McGehee S (2008) Haematozoa in forest birds from southern Chile: latitudinal gradients in prevalence and parasite lineage richness. Austral Ecol 33(3):329–340. https://doi.org/10.1111/j.1442-9993.2008.01820.x
Middleton AL (1998) Chipping Sparrow (Spizella passerina), version 2.0. Cornell Lab of Ornithology. https://birdsna.org/Species-Account/bna/species/chispa. Accessed 10 June 2017
Oakgrove KS, Harrigan RJ, Loiseau C, Guers S, Seppi B, Sehgal RNM (2014) Distribution, diversity and drivers of blood-borne parasite co-infections in Alaskan bird populations. Int J Parasitol 44(10):717–727. https://doi.org/10.1016/j.ijpara.2014.04.011
Oksanen J, Blanchet FG., Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHH., Szoecs E, Wagner H (2017) vegan: Community Ecology Package. https://cran.r-project.org/package=vegan. Accessed 2 May 2017
Ortego J, Cordero PJ, Aparicio JM, Calabuig G (2008) Consequences of chronic infections with three different avian malaria lineages on reproductive performance of Lesser Kestrels (Falco naumanni). J Ornithol 149(3):337–343. https://doi.org/10.1007/s10336-008-0287-9
Outlaw DC, Ricklefs RE (2009) On the phylogenetic relationships of Haemosporidian parasites from raptorial birds (Falconiformes and Strigiformes). J Parasitol 95(5):1171–1176. https://doi.org/10.1645/ge-1982.1
Outlaw DC, Ricklefs RE (2010) Comparative gene evolution in haemosporidian (Apicomplexa) parasites of birds and mammals. Mol Biol Evol 27:537–542. https://doi.org/10.1093/molbev/msp283
Outlaw DC, Ricklefs RE (2014) Species limits in avian malaria parasites (Haemosporida): how to move forward in the molecular era. Parasitology 141(10):1223–1232. https://doi.org/10.1017/s0031182014000560
Pagenkopp K, Klicka J, Durrant K, Garvin J, Fleischer R (2008) Geographic variation in malarial parasite lineages in the common yellowthroat (Geothlypis trichas). Conserv Genet 9(6):1577–1588. https://doi.org/10.1007/s10592-007-9497-6
Pérez-Tris J, Hellgren O, Križanauskienė A, Waldenström J, Secondi J, Bonneaud C, Fjeldså J, Hasselquist D, Bensch S (2007) Within-host speciation of malaria parasites. PLoS One 2(2):e235. https://doi.org/10.1371/journal.pone.0000235
Perkins SL (2008) Molecular systematics of the three mitochondrial protein-coding genes of malaria parasites: corroborative and new evidence for the origins of human malaria. Mitochondrial DNA 19(6):471–478. https://doi.org/10.1080/19401730802570926
Pyle P, Howell SN, Yunick RP, DeSante DF (1987) Identification guide to North American passerines. Slate Creek Press, Bolinas, California
Reullier J, Pérez-Tris J, Bensch S, Secondi J (2006) Diversity, distribution and exchange of blood parasites meeting at an avian moving contact zone. Mol Ecol 15(3):753–763. https://doi.org/10.1111/j.1365-294X.2005.02826.x
Ricklefs RE (2010) Host-pathogen coevolution, secondary sympatry and species diversification. Philos Trans R Soc Lond Ser B Biol Sci 365(1543):1139–1147. https://doi.org/10.1098/rstb.2009.0279
Ricklefs RE, Fallon SM (2002) Diversification and host switching in avian malaria parasites. Proc R Soc B Biol Sci 269(1494):885–892. https://doi.org/10.1098/rspb.2001.1940
Ricklefs RE, Swanson BL, Fallon SM, Martínez-Abraín A, Scheuerlein A, Gray J, Latta SC (2005) Community relationships of avian malaria parasites in southern Missouri. Ecol Monogr 75(4):543–559. https://doi.org/10.1890/04-1820
Sargent MS, Carter KS (1999) Managing Michigan wildlife: a landowners guide. Michigan United Conservation Clubs, East Lansing, MI
Sheldon BC, Verhulst S (1996) Ecological immunology: costly parasite defences and trade-offs in evolutionary ecology. Trends Ecol Evol 11:317–321. https://doi.org/10.1016/0169-5347(96)10039-2
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30(9):1312–1313. https://doi.org/10.1093/bioinformatics/btu033
Svensson-Coelho M, Blake JG, Loiselle BA, Penrose AS, Parker PG, Ricklefs RE (2013) Diversity, prevalence, and host specificity of avian Plasmodium and Haemoproteus in a western Amazon assemblage. Ornithol Monogr 76(1):1–47. https://doi.org/10.1525/om.2013.76.1.1
Swanson B, Lyons A, Bouzat J (2014) Distribution, prevalence and host specificity of avian malaria parasites across the breeding range of the migratory lark sparrow (Chondestes grammacus). Genetica 142(3):235–249. https://doi.org/10.1007/s10709-014-9770-9
Szymanski M, Lovette I (2005) High lineage diversity and host sharing of malarial parasites in a local avian assemblage. J Parasitol 91(4):768–774. https://doi.org/10.1645/GE-417R1.1
Theobald DM, Stevens DL Jr, White D, Urquhart NS, Olsen AR, Norman JB (2007) Using GIS to generate spatially balanced random survey designs for natural resource applications. Environ Manag 40(1):134–146. https://doi.org/10.1007/s00267-005-0199-x
Thurber MI, Gamble KC, Krebs B, Goldberg TL (2014) Molecular detection of Plasmodium in free-ranging birds and captive flamingos (Phoenicopterus chilensis) in Chicago. J Zoo Wildlife Med 45(4):749–754. https://doi.org/10.1638/2013-0284.1
van Riper C, van Riper SG, Goff ML, Laird M (1986) The epizootiology and ecological significance of malaria in Hawaiian land birds. Ecol Monogr 56(4):327–344. https://doi.org/10.2307/1942550
Valkiūnas G (2005) Avian malaria parasites and other Haemosporidia. CRC Press, Boca Raton, Florida
Valkiūnas G, Zehtindjiev P, Dimitrov D, Križanauskienė A, Iezhova TA, Bensch S (2008) Polymerase chain reaction-based identification of Plasmodium (Huffia) elongatum, with remarks on species identity of haemosporidian lineages deposited in Genbank. Parasitol Res 102(6):1185–1193. https://doi.org/10.1007/s00436-008-0892-9
Valkiūnas G, Sehgal RN, Iezhova TA, Hull AC (2010) Identification of Leucocytozoon toddi group (Haemosporida: Leucocytozoidae), with remarks on the species taxonomy of leucocytozoids. J Parasitol 96(1):170–177. https://doi.org/10.1645/GE-2109.1
Valkiūnas G, Palinauskas V, Ilgūnas M, Bukauskaitė D, Dimitrov D, Bernotienė R, Zehtindjiev P, Ilieva M, Iezhova TA (2014) Molecular characterization of five widespread avian haemosporidian parasites (Haemosporida), with perspectives on the PCR-based detection of haemosporidians in wildlife. Parasitol Res 113(6):2251–2263. https://doi.org/10.1007/s00436-014-3880-2
Vanderhoff N, Pyle P, Patten MA, Sallabanks R, James FC (1999) American Robin (Turdus migratorius), version 2.0. Cornell Lab of Ornithology. https://birdsna.org/Species-Account/bna/species/amerob. Accessed 10 June 2017
Walstrom VW, Outlaw DC (2016) Distribution and prevalence of Haemosporidian parasites in the Northern Cardinal (Cardinalis cardinalis). J Parasitol 103(1):63–68. https://doi.org/10.1645/14-693
Walther EL, Valkiūnas G, Gonzalez AD, Matta NE, Ricklefs RE, Cornel A, Sehgal RNM (2014) Description, molecular characterization, and patterns of distribution of a widespread New World avian malaria parasite (Haemosporida: Plasmodiidae), Plasmodium (Novyella) homopolare sp. nov. Parasitol Res 113(9):3319–3332. https://doi.org/10.1007/s00436-014-3995-5
Walther EL, Carlson JS, Cornel A, Morris BK, Sehgal RN (2016) First molecular study of prevalence and diversity of avian haemosporidia in a Central California songbird community. J Ornithol 157(2):549–564. https://doi.org/10.1007/s10336-015-1301-7
Wiersch S, Maier W, Kampen H (2005) Plasmodium (Haemamoeba) cathemerium gene sequences for phylogenetic analysis of malaria parasites. Parasitol Res 96(2):90–94. https://doi.org/10.1007/s00436-005-1324-8
Xian G, Homer C, Dewitz J, Fry J, Hossain N, Wickham J (2011) Change of impervious surface area between 2001 and 2006 in the conterminous United States. Photogramm Eng Remote Sens 77:758–762
Zélé F, Vézilier J, L’Ambert G, Nicot A, Gandon S, Rivero A, Duron O (2014) Dynamics of prevalence and diversity of avian malaria infections in wild Culex pipiens mosquitoes: the effects of Wolbachia, filarial nematodes and insecticide resistance. Parasite Vector 7(1):437. https://doi.org/10.1186/1756-3305-7-437
Funding
Funding was provided by Western Michigan University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
All applicable federal, state, and institutional guidelines for the care and use of animals were followed in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 5.81 kb)
Rights and permissions
About this article
Cite this article
Smith, J.D., Gill, S.A., Baker, K.M. et al. Prevalence and diversity of avian Haemosporida infecting songbirds in southwest Michigan. Parasitol Res 117, 471–489 (2018). https://doi.org/10.1007/s00436-017-5724-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5724-3