Abstract
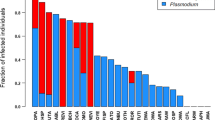
Haemosporidian parasites (genera Plasmodium and Haemoproteus) are common blood parasites of birds transmitted by dipteran insect vectors. We analyzed blood samples from 329 individuals of 43 bird species in eastern Tennessee to better understand the relationship between the local community of birds and their blood parasites, including the distribution of parasites across hosts and the underlying ecological factors and life -history traits that influence parasite prevalence across host species. Using molecular methods, we found 144 individuals of 25 species to be infected with haemosporidian parasites (overall prevalence of 44 %). We distinguished 22 genetic lineages, including 11 in the genus Haemoproteus and 11 in Plasmodium. Fourteen percent of infected individuals harbored more than one parasite lineage. Across species, total prevalence increased with local abundance and decreased with incubation period, but did not vary with nesting or foraging height, average annual survival of host species, migratory or flocking behavior, sexual dimorphism, average species mass, or among sites. The prevalence of Haemoproteus was higher in species that nest 1–5 m above ground than in species that nest below 1 m or above 5 m, and the prevalence of Plasmodium was marginally higher in species with open-cup nests. Infection status did not vary with age, sex, or body condition. Our research reveals substantial variation in prevalence and richness of haemosporidian parasites, some of which is related to specific avian life history traits.
Zusammenfassung
Prävalenz von aviären Hämosporidien in Osttennessee und deren Beziehung zur Biologie der Wirtsvögel
Hämosporidien der Gattungen Plasmodium und Haemoproteus sind bei Vögeln verbreitete Blutparasiten, welche von zweiflügeligen Insekten (Diptera) als Vektoren übertragen werden. Für ein besseres Verständnis der Beziehung zwischen einer lokalen Vogelgemeinschaft und deren Blutparasiten, einschließlich der Verteilung der Parasiten auf die Wirtsorganismen und die zugrunde liegenden ökologischen und biologischen Faktoren, welche die Prävalenz der Parasiten bei den Wirtsarten beeinflussen, analysierten wir Blutproben von 329 Individuen aus 43 Vogelarten in Osttennessee. 144 Individuen aus 25 Arten waren von hämosporidischen Parasiten befallen (Gesamtprävalenz 44 %); mithilfe molekularer Methoden ließen sich 22 genetische Stämme unterscheiden; von diesen gehörten elf zur Gattung Haemoproteus und elf zu Plasmodium. 14 % der infizierten Individuen wiesen mehr als einen Parasitenstamm auf. Bei allen Arten stieg die Gesamtprävalenz mit der lokalen Häufigkeit an und nahm mit der Brutdauer ab, variierte aber nicht in Abhängigkeit von der Nesthöhe oder der Höhe der Futtersuche, der durchschnittlichen jährlichen Überlebensrate der Wirtsart, dem Zug—oder Schwarmverhalten, Geschlechtsdimorphismen, der Durchschnittsmasse der Art oder in Abhängigkeit vom Ort. Die Prävalenz von Haemoproteus war höher bei Arten, die zwischen 1–5 m über dem Erdboden nisteten als bei solchen, welche entweder unter 1 m oder über 5 m nisteten; die Prävalenz von Plasmodium lag bei Arten mit offenen Napfnestern geringfügig höher. Der Infektionsstatus variierte weder mit dem Alter, dem Geschlecht noch mit der Kondition. Unsere Untersuchung belegt eine beträchtliche Variation bezüglich der Prävalenz und Vielfalt hämosporidischer Parasiten, die teilweise von der spezifischen Lebensweise der Vogelarten abhängt.


Similar content being viewed by others
References
Astudillo VG, Hernández SM, Kistler WM et al (2013) Spatial, temporal, molecular, and intraspecific differences of haemoparasite infection and relevant selected physiological parameters of wild birds in Georgia, USA. Int J Parasitol Parasites Wildl 2:178–189. doi:10.1016/j.ijppaw.2013.04.005
Bates M (1949) The natural history of mosquitoes. Macmillan, New York
Beadell JS, Fleischer RC (2005) A restriction enzyme-based assay to distinguish between avian hemosporidians. J Parasitol 91:683–685. doi:10.1645/GE-3412RN
Bensch S, Waldenström J, Jonzén N et al (2007) Temporal dynamics and diversity of avian malaria parasites in a single host species. J Anim Ecol 76:112–122. doi:10.2307/4125100
Bivand RS, Pebesma E, Gómez-Rubio V (2013) Applied spatial data analysis with R. Springer, New York
Borgia G, Collis K (1989) Female choice for parasite-free male satin bowerbirds and the evolution of bright male plumage. Behav Ecol Sociobiol 25:445–453. doi:10.1007/BF00300191
Brown CR, Brown MB (1992) Ectoparasitism as a cause of natal dispersal in cliff swallows. Ecology 73:1718–1723. doi:10.2307/1940023
Brown CR, Komar N, Quick SB et al (2001) Arbovirus infection increases with group size. Proc R Soc Lond Ser B Biol Sci 268:1833–1840. doi:10.1098/rspb.2001.1749
Cerný O, Votýpka J, Svobodová M (2011) Spatial feeding preferences of ornithophilic mosquitoes, blackflies and biting midges. Med Vet Entomol 25:104–108. doi:10.1111/j.1365-2915.2010.00875.x
Dawson RD, Bortolotti GR (2000) Effects of hematozoan parasites on condition and return rates of American kestrels. Auk 117:373–380. doi:10.2307/4089719
Desante DF, Burton KM, Saracco JF, Walker BL (1995) Productivity indices and survival rate estimates from MAPS, a continent-wide programme of constant-effort mist-netting in North America. J Appl Stat 22:935–948. doi:10.1080/02664769524720
Dobson A (2004) Population dynamics of pathogens with multiple host species. Am Nat 164:S64–S78. doi:10.1086/424681
Doucet SM, Montgomerie R (2003) Structural plumage colour and parasites in satin bowerbirds Ptilonorhynchus violaceus: implications for sexual selection. J Avian Biol 34:237–242. doi:10.1034/j.1600-048X.2003.03113.x
Dunning JB Jr (2008) CRC handbook of avian body masses, 2nd edn. CRC Press, Boca Raton
Eaton MD (2005) Human vision fails to distinguish widespread sexual dichromatism among sexually “monochromatic” birds. Proc Natl Acad Sci USA 102:10942–10946
Fallon SM, Ricklefs RE (2008) Parasitemia in PCR-detected Plasmodium and Haemoproteus infections in birds. J Avian Biol 39:514–522. doi:10.1111/j.2008.0908-8857.04308.x
Fallon SM, Ricklefs RE, Swanson BL, Bermingham E (2003) Detecting avian malaria: an improved polymerase chain reaction diagnostic. J Parasitol 89:1044–1047. doi:10.1645/GE-3157
Fallon SM, Ricklefs RE, Latta SC, Bermingham E (2004) Temporal stability of insular avian malarial parasite communities. Proc R Soc B Biol Sci 271:493–500. doi:10.1098/rspb.2003.2621
Fallon SM, Fleischer RC, Graves GR (2006) Malarial parasites as geographical markers in migratory birds? Biol Lett 2:213–216. doi:10.1098/rsbl.2005.0429
Fecchio A, Lima MR, Silveira P et al (2011) High prevalence of blood parasites in social birds from a neotropical savanna in Brazil. Emu 111:132–138. doi:10.1071/MU10063
Fecchio A, Lima MR, Svensson-Coelho M et al (2013) Structure and organization of an avian haemosporidian assemblage in a Neotropical savanna in Brazil. Parasitology 140:181–192. doi:10.1017/S0031182012001412
Ferrell ST, Snowden K, Marlar AB et al (2007) Fatal hemoprotozoal infections in multiple avian species in a zoological park. J Zoo Wildl Med 38:309–316. doi:10.1638/1042-7260(2007)038[0309:FHIIMA]2.0.CO;2
Fitze PS, Tschirren B, Richner H (2004) Life history and fitness consequences of ectoparasites. J Anim Ecol 73:216–226. doi:10.1111/j.0021-8790.2004.00799.x
Garamszegi LM, Møller AP (2012) The interspecific relationship between prevalence of blood parasites and sexual traits in birds when considering recent methodological advancements. Behav Ecol Sociobiol 66:107–119. doi:10.1007/s00265-011-1259-2
Garvin MC, Greiner EC (2003) Ecology of Culicoides (Diptera: Ceratopogonidae) in southcentral Florida and experimental Culicoides vectors of the avian hematozoan Haemoproteus danilewskyi Kruse. J Wildl Dis 39:170–178. doi:10.7589/0090-3558-39.1.170
Garvin MC, Remsen JV, Bishop MA, Bennett GF (1993) Hematozoa from passeriform birds in Louisiana. J Parasitol 79:318–321. doi:10.2307/3283564
Gibson G, Torr SJ (1999) Visual and olfactory responses of haematophagous Diptera to host stimuli. Med Vet Entomol 13:2–23. doi:10.1046/j.1365-2915.1999.00163.x
González AD, Matta NE, Ellis VA et al (2014) Mixed species flock, nest height, and elevation partially explain avian haemoparasite prevalence in Colombia. PLoS One 9:e100695. doi:10.1371/journal.pone.0100695
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387. doi:10.1126/science.7123238
Hellgren O, Pérez-Tris J, Bensch S (2009) A jack-of-all-trades and still a master of some: prevalence and host range in avian malaria and related blood parasites. Ecology 90:2840–2849. doi:10.1890/08-1059.1
Hijmans RJ, van Etten J, Cheng J et al (2014) raster: geographic data analysis and modeling. R package version 2.1-16. http://CRAN.R-project.org/package=raster
Hochachka WM, Dhondt AA (2000) Density-dependent decline of host abundance resulting from a new infectious disease. Proc Natl Acad Sci USA 97:5303–5306. doi:10.1073/pnas.080551197
Hudson PJ, Dobson AP, Lafferty KD (2006) Is a healthy ecosystem one that is rich in parasites? Trends Ecol Evol 21:381–385. doi:10.1016/j.tree.2006.04.007
Isaaks EH, Srivastava RM (1989) Applied geostatistics. Oxford University Press, New York
Isaksson C, Sepil I, Baramidze V, Sheldon BC (2013) Explaining variance of avian malaria infection in the wild: the importance of host density, habitat, individual life-history and oxidative stress. BMC Ecol 13:1–11. doi:10.1186/1472-6785-13-15
Knowles SCL, Palinauskas V, Sheldon BC (2010) Chronic malaria infections increase family inequalities and reduce parental fitness: experimental evidence from a wild bird population. J Evol Biol 23:557–569. doi:10.1111/j.1420-9101.2009.01920.x
Kuris AM, Hechinger RF, Shaw JC et al (2008) Ecosystem energetic implications of parasite and free-living biomass in three estuaries. Nature 454:515–518. doi:10.1038/nature06970
Lachish S, Knowles SCL, Alves R et al (2011) Fitness effects of endemic malaria infections in a wild bird population: the importance of ecological structure. J Anim Ecol 80:1196–1206. doi:10.1111/j.1365-2656.2011.01836.x
Lafferty KD, Dobson AP, Kuris AM (2006) Parasites dominate food web links. Proc Natl Acad Sci USA 103:11211–11216. doi:10.1073/pnas.0604755103
Lafferty KD, Allesina S, Arim M et al (2008) Parasites in food webs: the ultimate missing links. Ecol Lett 11:533–546. doi:10.1111/j.1461-0248.2008.01174.x
Lassen SB, Nielsen SA, Kristensen M (2012) Identity and diversity of blood meal hosts of biting midges (Diptera: Ceratopogonidae: Culicoides Latreille) in Denmark. Parasit Vectors 5:143. doi:10.1186/1756-3305-5-143
Loiseau C, Iezhova T, Valkiūnas G et al (2010) Spatial variation of haemosporidian parasite infection in African rainforest bird species. J Parasitol 96:21–29. doi:10.1645/GE-2123.1
Longmire JL, Maltbie M, Baker RJ (1997) Use of “‘lysis buffer’” in DNA isolation and its implication for museum collections. Occas Pap Mus Tex Tech Univ 163:1–4
Lutz HL, Hochachka WM, Engel JI et al (2015) Parasite prevalence corresponds to host life history in a diverse assemblage of afrotropical birds and haemosporidian parasites. PLoS One 10:e0121254. doi:10.1371/journal.pone.0121254
Martínez-de la Puente J, Merino S, Tomás G et al (2010) The blood parasite Haemoproteus reduces survival in a wild bird: a medication experiment. Biol Lett 6:663–665. doi:10.1098/rsbl.2010.0046
Marzal A, De Lope F, Navarro C, Møller AP (2005) Malarial parasites decrease reproductive success: an experimental study in a passerine bird. Oecologia 142:541–545. doi:10.1007/s00442-004-1757-2
Marzal A, Bensch S, Reviriego M et al (2008) Effects of malaria double infection in birds: one plus one is not two. J Evol Biol 21:979–987. doi:10.1111/j.1420-9101.2008.01545.x
Marzal A, Ricklefs RE, Valkiūnas G et al (2011) Diversity, loss, and gain of malaria parasites in a globally invasive bird. PLoS One 6:1–8. doi:10.1371/journal.pone.0021905
Medeiros MJ, Eiben JA, Haines WP et al (2013) The importance of insect monitoring to conservation actions in Hawaii. Hawaii Entomol Soc 45:149–166
Medeiros MCI, Ellis VA, Ricklefs RE (2014) Specialized avian Haemosporida trade reduced host breadth for increased prevalence. J Evol Biol 27:2520–2528. doi:10.1111/jeb.12514
Michel N, DeSante DF, Kaschube DR et al (2011) The Monitoring Avian Productivity and Survivorship (MAPS) Program annual reports, 1989–2006. NBII/MAPS Avian Demographics Query Interface. http://www.birdpop.org/nbii2006/NBIIHome.asp
Møller AP (2008) Flight distance and blood parasites in birds. Behav Ecol 19:1305–1313. doi:10.1093/beheco/arn074
Norris K, Anwar M, Read AF (1994) Reproductive effort influences the prevalence of haematozoan parasites in great tits. J Anim Ecol 63:601–610. doi:10.2307/5226
Norte AC, Araújo PM, Sampaio HL et al (2009) Haematozoa infections in a Great Tit Parus major population in Central Portugal: relationships with breeding effort and health. Ibis 151:677–688. doi:10.1111/j.1474-919X.2009.00960.x
Olival KJ, Stiner EO, Perkins SL (2007) Detection of Hepatocystis sp. in southeast Asian flying foxes (Pteropodidae) using microscopic and molecular methods. J Parasitol 93:1538–1540. doi:10.1645/GE-1208.1
Pérez-Tris J, Bensch S (2005) Diagnosing genetically diverse avian malarial infections using mixed-sequence analysis and TA-cloning. Parasitology 131:15–23. doi:10.1017/S003118200500733X
Pérez-Tris J, Hasselquist D, Hellgren O et al (2005) What are malaria parasites? Trends Parasitol 21:209–211. doi:10.1016/j.pt.2005.03.007
Perkins SL, Schall JJ (2002) A molecular phylogeny of malarial parasites recovered from cytochrome b gene sequences. J Parasitol 88:972–978. doi:10.1645/0022-3395(2002)088[0972:AMPOMP]2.0.CO;2
Pyle P (1997) Identification guide to North American birds: a compendium of information on identifying, ageing, and sexing “near-passerines” and passerines in the hand. Slate Creek Press, Bolinas
Read AF (1987) Comparative evidence supports the Hamilton and Zuk hypothesis on parasites and sexual selection. Nature 328:68–70. doi:10.1038/328068a0
R Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Ribeiro SF, Sebaio F, Branquinho FCS et al (2005) Avian malaria in Brazilian passerine birds: parasitism detected by nested PCR using DNA from stained blood smears. Parasitology 130:261–267. doi:10.1017/S0031182004006596
Richner H, Christe P, Oppliger A (1995) Paternal investment affects prevalence of malaria. Proc Natl Acad Sci USA 92:1192–1194. doi:10.1073/pnas.92.4.1192
Ricklefs RE (1992) Embryonic development period and the prevalence of avian blood parasites. Proc Natl Acad Sci USA 89:4722–4725. doi:10.1073/pnas.89.10.4722
Ricklefs RE, Swanson BL, Fallon SM et al (2005) Community relationships of avian malaria parasites in southern Missouri. Ecol Monogr 75:543–559. doi:10.2307/4539117
Ricklefs RE, Outlaw DC, Svensson-Coelho M et al (2014) Species formation by host shifting in avian malaria parasites. Proc Natl Acad Sci USA 111:14816–14821. doi:10.1073/pnas.1416356111
Rodewald P (ed) (2015) The Birds of North America Online. Cornell Laboratory of Ornithology, Ithaca, NY. http://bna.birds.cornell.edu/BNA/
SAS Institute Inc. (2011) Base SAS® 9.3 procedures guide. SAS Institute Inc, Cary
Sauer JR, Hines JE, Fallon JE et al (2011) The North American breeding bird survey, results and analysis 1966–2010. Version 12.07.2011. USGS Patuxent Wildlife Research Center, Laurel
Scheuerlein A, Ricklefs RE (2004) Prevalence of blood parasites in European passeriform birds. Proc R Soc B Biol Sci 271:1363–1370. doi:10.1098/rspb.2004.2726
Schrader MS, Walters EL, James FC, Greiner EC (2003) Seasonal prevalence of a haematozoan parasite of red-bellied woodpeckers (Melanerpes carolinus) and its association with host condition and overwinter survival. Auk 120:130–137. doi:10.1642/0004-8038(2003)120[0130:SPOAHP]2.0.CO;2
Schulte-Hostedde AI, Zinner B, Millar JS, Hickling GJ (2005) Restitution of mass-size residuals: validating body condition indices. Ecology 86:155–163. doi:10.1890/04-0232
Svensson LME, Ricklefs RE (2009) Low diversity and high intra-island variation in prevalence of avian Haemoproteus parasites on Barbados, Lesser Antilles. Parasitology 136:1121–1131. doi:10.1017/S0031182009990497
Svensson-Coelho M, Blake JG, Loiselle BA et al (2013) Diversity, prevalence, and host specificity of avian Plasmodium and Haemoproteus in a Western Amazon assemblage. Ornithol Monogr 76:1–47
Tamura K, Peterson D, Peterson N et al (2011) MEGA5: Molecular Evolutionary Genetics Analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739. doi:10.1093/molbev/msr121
Tella JL, Blanco G, Forero MG et al (1999) Habitat, world geographic range, and embryonic development of hosts explain the prevalence of avian hematozoa at small spatial and phylogenetic scales. Proc Natl Acad Sci USA 96:1785–1789. doi:10.1073/pnas.96.4.1785
Thomas F, Schmidt-Rhaesa A, Martin G et al (2002) Do hairworms (Nematomorpha) manipulate the water seeking behaviour of their terrestrial hosts? J Evol Biol 15:356–361. doi:10.1046/j.1420-9101.2002.00410.x
Torchin ME, Lafferty KD, Dobson AP et al (2003) Introduced species and their missing parasites. Nature 421:628–630. doi:10.1038/nature01346
Valkiūnas G (2005) Avian malaria parasites and other haemosporidia. CRC Press, Boca Raton
Valkiūnas G, Bensch S, Iezhova TA et al (2006) Nested cytochrome b polymerase chain reaction diagnostics underestimate mixed infections of avian blood Haemosporidian parasites: microscopy is still essential. J Parasitol 92:418–422. doi:10.1645/GE-3547RN.1
Valkiūnas G, Iezhova TA, Križanauskienė A et al (2008) A comparative analysis of microscopy and PCR-based detection methods for blood parasites. J Parasitol 94:1395–1401. doi:10.1645/GE-1570.1
van Riper C, van Riper SG, Goff ML, Laird M (1986) The epizootiology and ecological significance of malaria in Hawaiian land birds. Ecol Monogr 56:327–344. doi:10.2307/1942550
Waldenström J, Bensch S, Kiboi S et al (2002) Cross-species infection of blood parasites between resident and migratory songbirds in Africa. Mol Ecol 11:1545–1554
Wilson K, Bjørnstad ON, Dobson AP et al (2002) Heterogeneities in macroparasite infections: patterns and processes. In: Hudson PJ, Rizzoli A, Grenfell BT et al (eds) The ecology of wildlife diseases. Oxford University Press, Oxford, pp 6–44
Zhang Y, Wu Y, Zhang Q et al (2014) Prevalence patterns of avian Plasmodium and Haemoproteus parasites and the influence of host relative abundance in southern China. PLoS One 9:e99501. doi:10.1371/journal.pone.0099501
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1023. doi:10.1016/S0020-7519(96)00086-0
Acknowledgments
Bill Smith, Justine Cucchiara, Stephen Lyn Bales, Knox County Parks and Recreation, and Jeff Debree provided access to banding sites. Anna Harris provided useful suggestions on the project design, and Jonathan Fitz Gerald provided assistance with laboratory techniques. Sean Gunter and Sarah Walters generously provided housing accommodations. This work was supported by grants from the National Science Foundation Malaria Research Coordination Network, the Whitney R. Harris World Ecology Center, and the Tennessee Ornithological Society. The experiments in this study comply with the current laws of the United States. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. C. Klasing.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Matthews, A.E., Ellis, V.A., Hanson, A.A. et al. Avian haemosporidian prevalence and its relationship to host life histories in eastern Tennessee. J Ornithol 157, 533–548 (2016). https://doi.org/10.1007/s10336-015-1298-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-015-1298-y