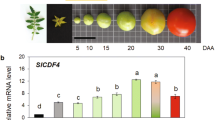
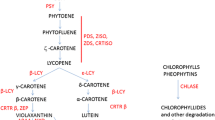
Abstract
Main conclusion
Systemic responses to an arbuscular mycorrhizal fungus reveal opposite phenological patterns in two tomato ripening mutants depending whether ethylene or light reception is involved.
The availability of tomato ripening mutants has revealed many aspects of the genetics behind fleshy fruit ripening, plant hormones and light signal reception. Since previous analyses revealed that arbuscular mycorrhizal symbiosis influences tomato berry ripening, we wanted to test the hypothesis that an interplay might occur between root symbiosis and fruit ripening. With this aim, we screened seven tomato mutants affected in the ripening process for their responsiveness to the arbuscular mycorrhizal fungus Funneliformis mosseae. Following their phenological responses we selected two mutants for a deeper analysis: Green ripe (Gr), deficient in fruit ethylene perception and high-pigment-1 (hp-1), displaying enhanced light signal perception throughout the plant. We investigated the putative interactions between ripening processes, mycorrhizal establishment and systemic effects using biochemical and gene expression tools. Our experiments showed that both mutants, notwithstanding a normal mycorrhizal phenotype at root level, exhibit altered arbuscule functionality. Furthermore, in contrast to wild type, mycorrhization did not lead to a higher phosphate concentration in berries of both mutants. These results suggest that the mutations considered interfere with arbuscular mycorrhiza inducing systemic changes in plant phenology and fruits metabolism. We hypothesize a cross talk mechanism between AM and ripening processes that involves genes related to ethylene and light signaling.






Similar content being viewed by others
Abbreviations
- AM:
-
Arbuscular mycorrhiza
- AMF:
-
Arbuscular mycorrhizal fungi
- ET:
-
Ethylene
- ETRs:
-
Ethyelene receptors
- MYC:
-
Mycorrhizal
- NM:
-
Non-mycorrhizal
References
Balestrini R, Gómez-Ariza J, Lanfranco L, Bonfante P (2007) Laser microdissection reveals that transcripts for five plant and one fungal phosphate transporter genes are contemporaneously present in arbusculated cells. Mol Plant Microbe Interact 20:1055–1062. doi:10.1094/MPMI-20-9-1055
Barry CS, Giovannoni JJ (2006) Ripening in the tomato Green-ripe mutant is inhibited by ectopic expression of a protein that disrupts ethylene signaling. Proc Natl Acad Sci USA 103:7923–7928. doi:10.1073/pnas.0602319103
Barry CS, Giovannoni JJ (2007) Ethylene and fruit ripening. J Plant Growth Regul 26:143–159. doi:10.1007/s00344-007-9002-y
Boldt K, Pörs Y, Haupt B et al (2011) Photochemical processes, carbon assimilation and RNA accumulation of sucrose transporter genes in tomato arbuscular mycorrhiza. J Plant Physiol 168:1256–1263. doi:10.1016/j.jplph.2011.01.026
Bombarely A, Menda N, Tecle IY et al (2011) The sol genomics network (solgenomics.net): growing tomatoes using Perl. Nucleic Acids Res 39:D1149–D1155. doi:10.1093/nar/gkq866
Bonfante P, Genre A (2010) Mechanisms underlying beneficial plant–fungus interactions in mycorrhizal symbiosis. Nat Commun 1:1–11. doi:10.1038/ncomms1046
Ceballos I, Ruiz M, Fernández C et al (2013) The in vitro mass-produced model mycorrhizal fungus, Rhizophagus irregularis, significantly increases yields of the globally important food security crop cassava. PLoS One 8:e70633. doi:10.1371/journal.pone.0070633
Copetta A, Bardi L, Bertolone E, Berta G (2011) Fruit production and quality of tomato plants (Solanum lycopersicum L.) are affected by green compost and arbuscular mycorrhizal fungi. Plant Biosyst Int J Deal Asp Plant Biol 145:106–115. doi:10.1080/11263504.2010.539781
De Mendiburu F (2012) Agricolae: statistical procedures for agricultural research. R Package Version 1:1–6
Etemadi M, Gutjahr C, Couzigou J-M et al (2014) Auxin perception is required for arbuscule development in arbuscular mycorrhizal symbiosis. Plant Physiol 166:281–292. doi:10.1104/pp.114.246595
Fiorilli V, Catoni M, Miozzi L et al (2009) Global and cell-type gene expression profiles in tomato plants colonized by an arbuscular mycorrhizal fungus. New Phytol 184:975–987. doi:10.1111/j.1469-8137.2009.03031.x
Fracetto GGM, Peres LEP, Mehdy MC, Lambais MR (2013) Tomato ethylene mutants exhibit differences in arbuscular mycorrhiza development and levels of plant defense-related transcripts. Symbiosis 60:155–167. doi:10.1007/s13199-013-0251-1
Galpaz N, Wang Q, Menda N et al (2008) Abscisic acid deficiency in the tomato mutant high-pigment 3 leading to increased plastid number and higher fruit lycopene content. Plant J Cell Mol Biol 53:717–730. doi:10.1111/j.1365-313X.2007.03362.x
García-Sánchez M, Palma JM, Ocampo JA et al (2014) Arbuscular mycorrhizal fungi alleviate oxidative stress induced by ADOR and enhance antioxidant responses of tomato plants. J Plant Physiol 171:421–428. doi:10.1016/j.jplph.2013.10.023
Giovannetti M, Avio L, Barale R et al (2012) Nutraceutical value and safety of tomato fruits produced by mycorrhizal plants. Br J Nutr 107:242–251. doi:10.1017/S000711451100290X
Giovannoni JJ (2004) Genetic regulation of fruit development and ripening. Plant Cell 16:S170–S180. doi:10.1105/tpc.019158
Giraudoux P (2015) pgirmess: miscellaneous functions for analysis and display of ecological and spatial data. R Package Version 1:1–63
Gómez-Ariza J, Balestrini R, Novero M, Bonfante P (2009) Cell-specific gene expression of phosphate transporters in mycorrhizal tomato roots. Biol Fertil Soils 45:845–853. doi:10.1007/s00374-009-0399-2
Guether M, Balestrini R, Hannah M et al (2009) Genome-wide reprogramming of regulatory networks, transport, cell wall and membrane biogenesis during arbuscular mycorrhizal symbiosis in Lotus japonicus. New Phytol 182:200–212. doi:10.1111/j.1469-8137.2008.02725.x
Hart M, Ehret DL, Krumbein A et al (2014) Inoculation with arbuscular mycorrhizal fungi improves the nutritional value of tomatoes. Mycorrhiza 25:359–376. doi:10.1007/s00572-014-0617-0
Hewitt EJ (1966) Sand and water culture methods used in the study of plant nutrition. In: Hewitt EJ (ed) Farnham Royal, England: Commonwealth Agricultural Bureaux (1966), p 547; Technical Communication No. 22 (revised 2nd edn) of the Commonwealth Bureau of Horticulture and Plantation Crops, East Malling, Maidstone, Kent. Exp Agric 3:104. doi: 10.1017/S0014479700021852
Kaschuk G, Kuyper TW, Leffelaar PA et al (2009) Are the rates of photosynthesis stimulated by the carbon sink strength of rhizobial and arbuscular mycorrhizal symbioses? Soil Biol Biochem 41:1233–1244
Khatabi B, Schäfer P (2012) Ethylene in mutualistic symbioses. Plant Signal Behav 7:1634–1638. doi:10.4161/psb.22471
Kiers ET, Duhamel M, Beesetty Y et al (2011) Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333:880–882. doi:10.1126/science.1208473
Klee HJ, Giovannoni JJ (2011) Genetics and control of tomato fruit ripening and quality attributes. Annu Rev Genet 45:41–59. doi:10.1146/annurev-genet-110410-132507
Komsta L (2006) Processing data for outliers. R News 6:10–13
Kruskal WH (1952) A Nonparametric test for the several sample problem. Ann Math Stat 23:525–540. doi:10.1214/aoms/1177729332
Kruskal WH, Wallis WA (1952) Use of ranks in one-criterion variance analysis. J Am Stat Assoc 47:583–621. doi:10.1080/01621459.1952.10483441
Lin T, Zhu G, Zhang J et al (2014) Genomic analyses provide insights into the history of tomato breeding. Nat Genet 46:1220–1226. doi:10.1038/ng.3117
Liu Y, Roof S, Ye Z et al (2004) Manipulation of light signal transduction as a means of modifying fruit nutritional quality in tomato. Proc Natl Acad Sci USA 101:9897–9902. doi:10.1073/pnas.0400935101
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. doi:10.1006/meth.2001.1262
López-Ráez JA, Flors V, García JM, Pozo MJ (2010) AM symbiosis alters phenolic acid content in tomato roots. Plant Signal Behav 5:1138–1140. doi:10.4161/psb.5.9.12659
Mustilli AC, Fenzi F, Ciliento R et al (1999) Phenotype of the tomato high pigment-2 mutant is caused by a mutation in the tomato homolog of DEETIOLATED1. Plant Cell 11:145–157
Oldroyd GED (2013) Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nat Rev Microbiol 11:252–263. doi:10.1038/nrmicro2990
Pan X, Zhu B, Zhu H et al (2014) iTRAQ protein profile analysis of tomato Green-ripe mutant reveals new aspects critical for fruit ripening. J Proteome Res 13:1979–1993. doi:10.1021/pr401091n
Regvar M, Vogel-Mikuš K, Ševerkar T (2003) Effect of AMF inoculum from field isolates on the yield of green pepper, parsley, carrot, and tomato. Folia Geobot 38:223–234. doi:10.1007/BF02803154
Rodríguez FI, Esch JJ, Hall AE et al (1999) A copper cofactor for the ethylene receptor ETR1 from Arabidopsis. Science 283:996–998
Ruzicka D, Chamala S, Barrios-Masias FH et al (2013) Inside arbuscular mycorrhizal roots—molecular probes to understand the symbiosis. Plant Genome. doi:10.3835/plantgenome2012.06.0007
Salvioli A, Bonfante P (2013) Systems biology and “omics” tools: a cooperation for next-generation mycorrhizal studies. Plant Sci 203–204:107–114. doi:10.1016/j.plantsci.2013.01.001
Salvioli A, Zouari I, Chalot M, Bonfante P (2012) The arbuscular mycorrhizal status has an impact on the transcriptome profile and amino acid composition of tomato fruit. BMC Plant Biol 12:44. doi:10.1186/1471-2229-12-44
Schwarz D, Welter S, George E et al (2011) Impact of arbuscular mycorrhizal fungi on the allergenic potential of tomato. Mycorrhiza 21:341–349. doi:10.1007/s00572-010-0345-z
Schweiger R, Müller C (2015) Leaf metabolome in arbuscular mycorrhizal symbiosis. Curr Opin Plant Biol 26:120–126. doi:10.1016/j.pbi.2015.06.009
Torres de Los Santos R, Santos R, Vierheilig H, Ocampo JA, García Garrido JM (2011) Altered pattern of arbuscular mycorrhizal formation in tomato ethylene mutants. Plant Signal Behav 6:755–758. doi:10.4161/psb.6.5.15415
Trouvelot A, Kough J, Gianinazzi-Pearson V (1986) Mesure du taux de mycorhization VA d’un système radiculaire. Recherche de méthodes d’estimation ayant une signification fonctionnelle. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Physiological and genetical aspects of mycorrhizae. INRA Press, Paris, pp 217–221
Varma Penmetsa R, Uribe P, Anderson J et al (2008) The Medicago truncatula ortholog of Arabidopsis EIN2, sickle, is a negative regulator of symbiotic and pathogenic microbial associations. Plant J 55:580–595. doi:10.1111/j.1365-313X.2008.03531.x
Volpe V, Giovannetti M, Sun X-G et al (2015) The phosphate transporters LjPT4 and MtPT4 mediate early root responses to phosphate status in non mycorrhizal roots: characterization of AM-induced Pi transporters. Plant, Cell Environ 39:660–671. doi:10.1111/pce.12659
Vrebalov J, Pan IL, Arroyo AJM et al (2009) Fleshy fruit expansion and ripening are regulated by the tomato SHATTERPROOF gene TAGL1. Plant Cell 21:3041–3062. doi:10.1105/tpc.109.066936
Wright D, Scholes J, Read D (1998) Effects of VA mycorrhizal colonization on photosynthesis and biomass production of Trifolium repens L. Plant, Cell Environ 21:209–216
Zouari I, Salvioli A, Chialva M et al (2014) From root to fruit: RNA-Seq analysis shows that arbuscular mycorrhizal symbiosis may affect tomato fruit metabolism. BMC Genom 15:221. doi:10.1186/1471-2164-15-221
Zsögön A, Lambais MR, Benedito VA et al (2008) Reduced arbuscular mycorrhizal colonization in tomato ethylene mutants. Sci Agric 65:259–267. doi:10.1590/S0103-90162008000300006
Acknowledgments
The authors gratefully thank Dr. Agnese Giacomino (Department of Chemistry, University of Turin) for invaluable support during ICP–OES analysis and Dr. Maria Teresa Della Beffa (DBIOS, University of Turin) for maintaining tomato cultures in climatic chambers and greenhouses. We are deeply indebted with Katherine A. Borkovich for the critical reading and language editing of the final manuscript. The PhD fellowship to MC was funded by Compagnia di San Paolo. Research in the Bonfante lab was supported by the Mycoplant Project (Progetto di Ateneo and CSP). Activities in the Giovannoni lab were supported by the US Department of Agriculture. Agricultural Research Service.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chialva, M., Zouari, I., Salvioli, A. et al. Gr and hp-1 tomato mutants unveil unprecedented interactions between arbuscular mycorrhizal symbiosis and fruit ripening. Planta 244, 155–165 (2016). https://doi.org/10.1007/s00425-016-2491-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-016-2491-9