Abstract
Main conclusion
The aim of this study is to demonstrate the feasibility of down-regulating endogeneous prefoldin-2 root-knot nematode transcripts by expressing dsRNA with sequence identity to the nematode gene in tobacco roots under the influence of strong Arabidopsis ubiquitin (UBQ1) promoter.
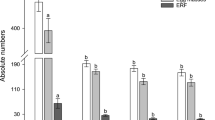
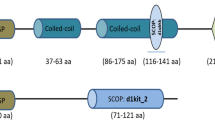
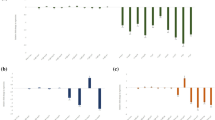
Root-knot nematodes (RKNs) are sedentary endoparasites infecting a wide range of plant species. They parasitise the root system, thereby disrupting water and nutrient uptake and causing major reductions in crop yields. The most reliable means of controlling RKNs is via the use of soil fumigants such as methyl bromide. With the emergence of RNA interference (RNAi) technology, which permits host-mediated nematode gene silencing, a new strategy to control plant pathogens has become available. In the present study, we investigated host-induced RNAi gene silencing of prefoldin-2 in transgenic Nicotiana benthamiana. Reductions in prefoldin-2 mRNA transcript levels were observed when nematodes were soaked in a dsRNA solution in vitro. Furthermore, nematode reproduction was suppressed in RNAi transgenic lines, as evident by reductions in the numbers of root knots (by 34–60 % in independent RNAi lines) and egg masses (by 33–58 %). Endogenous expression of prefoldin-2, analysed via real-time polymerase chain reaction and Western blotting, revealed that the gene was strongly expressed in the pre-parasitic J2 stage. Our observations demonstrate the relevance and potential importance of targeting the prefoldin gene during the nematode life cycle. The work also suggests that further improvements in silencing efficiency in economically important crops can be accomplished using RNAi directed against plant-parasitic nematodes.







Similar content being viewed by others
Abbreviations
- dsRNA:
-
Double-strand RNA
- GUS:
-
β-Glucuronidase
- J2:
-
Juvenile 2
- PFD:
-
Prefoldin
- RKN:
-
Root-knot nematode
- RNAi:
-
RNA interference
- RT-PCR:
-
Reverse transcription-polymerase chain reaction
- UBQ:
-
Ubiquitin
References
Abad P, Favery B, Rosso M, Castagnone-Sereno P (2003) Root-knot nematode parasitism and host response: molecular basis of a sophisticated interaction. Mol Plant Pathol 4:217–224
Bakhetia M, Charlton W, Atkinson HJ, McPherson MJ (2005) RNA interference of dual oxidase in the plant nematode Meloidogyne incognita. Mol Plant Microbe Interact 18:1099–1106
Benfey PN, Chua NH (1990) The cauliflower mosaic virus 35S promoter—combinatorial regulation of transcription in plants. Science 250:959–966
Binet MN, Lepetit M, Weil JH, Tessier LH (1991) Analysis of a sunflower polyubiquitin promoter by transient expression. Plant Sci 79:87–94
Bockenhoff A, Grundler FMW (1994) Studies on the nutrient uptake by the beet cyst nematode Heterodera schachtii by in situ microinjection of fluorescent probes into the feeding structures in Arabidopsis thaliana. Parasitology 109:249–254
Callis J, Vierstra RD (1989) Ubiquitin and ubiquitin genes in higher plants. Oxford Surv Plant Mol Cell Biol 6:1–30
Callis J, Raasch J, Vierstra RD (1990) Ubiquitin extension proteins of Arabidopsis thaliana: structure, localization and expression of their promoters in transgenic tobacco. J Biol Chem 265:12486–12493
Chen Q, Rehman S, Smant G, Jones JT (2005) Functional analysis of pathogenicity proteins of the potato cyst nematode Globodera rostochiensis using RNAi. Mol Plant Microbe Interact 18:621–625
Christensen AH, Sharrok RA, Quail PH (1992) Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18:675–689
Davis EL, Hussey RS, Baum TJ (2004) Getting to the roots of parasitism by nematodes. Trends Parasitol 20:134–141
Elbashir SM, Lendeckel W, Tuschl T (2001) RNA interference is mediated by 21- and 22-nucleotide RNAs. Genes Dev 15:188–200
Fairbairn DJ, Cavallaro AS, Bernard M, Mahalinga-Iyer J, Graham MW, Botella JR (2007) Host-delivered RNAi: an effective strategy to silence genes in plant parasitic nematodes. Planta 226:1525–1533
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double stranded RNA in Caenorhabditis elegans. Nature 391:806–811
Fraser AG, Kamath RS, Zipperlen P, Martinez-Campos M, Sohrmann M, Ahringer J (2000) Functional genomic analysis of Caenorhabditis elegans chromosome I by systematic RNA interference. Nature 408:325–330
Garbarino JE, Belknap W (1994) Isolation of ubiquitin-ribosomal protein of gene (ubi3) from potato and expression of its promoter in transgenic plants. Plant Mol Biol 24:119–127
Garbarino JE, Oosumi T, Belknap WR (1995) Isolation of a polyubiquitin promoter and its expression in transgenic potato plants. Plant Physiol 109:1371–1378
Genschik P, Marbach J, Uze M, Feuerman M, Plesse B, Fleck J (1994) Structure and promoter activity of a stress and developmentally regulated polyubiquitin-encoding gene of Nicotiana tabacum. Gene 148:195–202
Gheysen G, Fenoll C (2002) Gene expression in nematode feeding sites. Annu Rev Phytopathol 40:191–2194
Gheysen G, Vanholme B (2007) RNAi from plants to nematodes. Trends Biotechnol 25:89–92
Grefen C, Donald N, Hashimoto K, Kudla J, Schumacher K, Blatt MR (2010) A ubiquitin-10 promoter-based vector set for fluorescent protein tagging facilitates temporal stability and native protein distribution in transient and stable expression studies. Plant J 64:355–365
Hahn BS, Jeon IS, Jung YJ, Kim JB, Park JS, Ha SH, Kim KH, Kim HM, Yang JS, Kim YH (2007) Expression of hemagglutinin-neuraminidase protein of Newcastle disease virus in transgenic tobacco. Plant Biotechnol Rep 1:85–92
Hansen WJ, Cowan NJ, Welch WJ (1999) Prefoldin-nascent chain complexes in the folding of cytoskeletal proteins. J Cell Biol 145:265–277
Hershko A (1988) Ubiquitin-mediated protein degradation. J Biol Chem 263:15237–15240
Horsch RB, Fry JE, Hoffmann N, Eicholz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Huang G, Allen R, Davis EL, Baum TJ, Hussey RS (2006) Engineering broad root-knot resistance in transgenic plants by RNAi silencing of a conserved and essential root-knot nematode parasitism gene. Proc Natl Acad Sci USA 103:14302–14306
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Jukes TH, Cantor CR (1969) Evolution of protein molecules. In: Munro HN (ed) Mammalian protein metabolism. Academic Press, New York, pp 21–132
Jung C, Wyss U (1999) New approaches to control plant parasitic nematodes. App Microbiol Biotechnol 51:439–446
Kamath RS, Fraser AG, Dong Y, Poulin G, Durbin R, Gotta M, Kanapin A, Le Bot N, Moreno S, Sohrmann M, Welchman DP, Zipperlen P, Ahringer J (2003) Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421:231–237
Kamo K, Blowers A, Mcelroy D (2000) Effect of the cauliflower mosaic virus 35S, actin, and ubiquitin promoters on uidA expression from a bar-uidA fusion gene in transgenic gladiolus plants. In Vitro Cell Dev Biol Plant 36:13–20
Kang MJ, Kim YH, Hahn BS (2010) Expressed sequence tag analysis generated from a normalized full-length cDNA library of the root-knot nematode (Meloidogyne incognita). Genes Genom 32:553–562
Klink VP, Hosseini P, Matsye P, Alkharouf N, Matthews BF (2009) A gene expression analysis of syncytia laser microdissected from the roots of the Glycine max (soybean) genotype PI 548402 (Peking) undergoing a resistant reaction after infection by Heterodera glycines (soybean cyst nematode). Plant Mol Biol 71:525–567
Le Bot N, Tsai MC, Andrews RK, Ahringer J (2003) TAC-1, a regulator of microtubule length in the Caenorhabditis elegans embryo. Curr Biol 13:1499–1505
Li X, Wu HX, Southerton SG (2010) Comparative genomics reveals conservative evolution of the xylem transcriptome in vascular plants. BMC Evol Biol 10:190
Lilley CJ, Bakhetia M, Charlton WL, Urwin PE (2007) Recent progress in the development of RNA interference for plant parasitic nematodes. Mol Plant Pathol 8:701–711
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25:402–408
Lundin VF, Srayko M, Hyman AA, Leroux MR (2008) Efficient chaperone-mediated tubulin biogenesis is essential for cell division and cell migration in Caenorhabditis elegans. Dev Biol 313:320–334
Maeda I, Kohara Y, Yamamoto M, Sugimoto A (2001) Large-scale analysis of gene function in Caenorhabditis elegans by high-throughput RNAi. Curr Biol 11:171–176
Martin-Benito J, Boskovic J, Gomez-Puertas P, Carrascosa JL, Simons CT, Lewis SA, Bartolini F, Cowan NJ, Valpuesta JM (2002) Structure of eukaryotic prefoldin and of its complexes with unfolded actin and the cytosolic chaperonin CCT. EMBO J 21:6377–6386
McElroy D, Zhang W, Cao J, Wu R (1990) Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2:163–171
Norris SR, Meyer SE, Callis J (1993) The intron of Arabidopsis thaliana polyubiquitin genes is conserved in location and is a quantitative determinant of chimeric gene expression. Plant Mo1. Biol 21:895–906
Ogawa S, Matsubayashi Y, Nishida E (2004) An evolutionary conserved gene required for proper microtubule architecture in Caenorhabditis elegans. Gen Cells 9:83–93
Ozkaynak E, Finley D, Solomon MJ, Varshavsky A (1987) The yeast ubiquitin genes: a family of natural gene fusions. EMBO J 6:1429–1439
Rosso MN, Dubrana MP, Cimbolini N, Jaubert S, Abad P (2005) Application of RNA interference to root-knot nematode genes encoding esophageal gland proteins. Mol Plant Mic Inter 18:615–620
Rost B (2001) Review: protein secondary structure prediction continues to rise. J Struct Biol 134:204–218
Saitou N, Nei M (1987) The neighbor-joining method–a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Schlesinger MJ, Bond U (1987) Ubiquitin genes. Oxf Surv Eukaryot Genes 4:77–91
Siegert R, Leroux MR, Scheufler C, Hartl FU, Moarefi I (2000) Structure of the molecular chaperone prefoldin: unique interaction of multiple coiled coil tentacles with unfolded proteins. Cell 103:621–632
Simmer F, Moorman C, Van Der Linden AM, Kuijk E, Van Den Berghe PV, Kamath R, Fraser AG, Ahringer J, Plasterk RH (2003) Genome-wide RNAi of Caenorhabditis elegans using the hypersensitive rrf-3 strain reveals novel gene functions. PLoS Biol 1:77–84
Sindhu AS, Maier TR, Mitchum MG, Hussey RS, Davis EL, Baum TJ (2009) Effective and specific in planta RNAi in cyst nematodes: expression interference of four parasitism genes reduces parasitic success. J Exp Bot 60:315–324
Smalle J, Vierstra RD (2004) The ubiquitin 26S proteosome proteolytic pathway. Annu Rev Plant Biol 55:555–590
Srayko Kaya A, Stamford J, Hyman AA (2005) Identification and characterization of factors required for microtubule growth and nucleation in the early Caenorhabditis elegans embryo. Dev Cell 9:223–236
Steeves RM, Todd TC, Essig JS, Trick HN (2006) Transgenic soybeans expressing siRNAs specific to a major sperm protein gene suppress Heterodera glycines reproduction. Func Plant Biol 33:991–999
Urwin PE, Lilley CJ, Atkinson HJ (2002) Ingestion of double stranded RNA by preparasitic juvenile cyst nematodes leads to RNA interference. Mol Plant Mic Inter 15:747–752
Vainberg IE, Lewis SA, Rommelaere H, Ampe C, Vandekerckhove J, Klein HL, Cowan NJ (1998) Prefoldin, a chaperone that delivers unfolded proteins to cytosolic chaperonin. Cell 93:863–873
Williamson VM, Gleason CA (2003) Plant–nematode interactions. Curr Opi Plant Biol 6:327–333
Yadav BC, Veluthambi K, Subramaniam K (2006) Host-generated double stranded RNA induces RNAi in plant-parasitic nematodes and protects the host from infection. Mol Biochem Parasitol 148:219–222
Zako Iizuka R, Okochi M, Nomura T, Ueno T, Tadakuma H, Yohda M, Funatsu T (2005) Facilitated release of substrate protein from prefoldin by chaperonin. FEBS Lett 579:3718–3724
Acknowledgments
This project was supported by a grant from the National Academy of Agricultural Science (PJ008541), RDA, Korea. This study was supported by the 2014 Postdoctoral Fellowship Program (to H. Y. C. and I. C. C.) of the National Academy of Agricultural Science, RDA, Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
H. Ajjappala and H. Y. Chung contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ajjappala, H., Chung, H.Y., Sim, JS. et al. Disruption of prefoldin-2 protein synthesis in root-knot nematodes via host-mediated gene silencing efficiently reduces nematode numbers and thus protects plants. Planta 241, 773–787 (2015). https://doi.org/10.1007/s00425-014-2211-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-014-2211-2