Abstract
Main conclusion
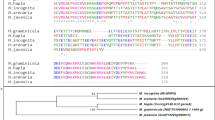
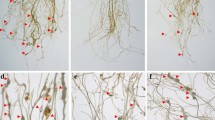
Mi-msp10 and Mi-msp23 effector genes play a significant role during Meloidogyne incognita parasitism on Arabidopsis roots. The role of these genes was confirmed by demonstrating the decrease of the level of susceptibility of Arabidopsis by the silencing of Mi-msp10 and Mi-msp23 genes using HD-RNAi technology.
Abstract
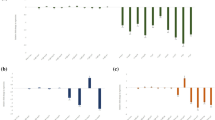
Root-knot nematodes (RKNs) are the most damaging pathogens severely affecting global food production. The sustainable options to minimize menace of nematode populations through economically feasible measures are limited. Thus, the development of innovative and target-specific strategies that aid in their management is imperative. RNAi technology has emerged as a sustainable and target-specific alternative to control phytonematodes. Here, we characterized two novel subventral gland and dorsal gland-specific effectors, Mi-msp10 and Mi-msp23, to determine their potential effectiveness in controlling M. incognita. Comparative developmental profiling using qRT-PCR revealed higher expression of both effectors in the adult nematode female. Furthermore, functional evaluation of Mi-msp10 and Mi-msp23 dsRNA cassettes was performed using host-delivered RNAi (HD-RNAi) in Arabidopsis. The transgenic lines were examined against M. incognita, and the phenotypic effect of HD-RNAi was evident with a 61% and 51% reduction in gall formation in the Mi-msp10 and Mi-msp23 RNAi lines, respectively. A significant drop in the nematode adult females by 59% for Mi-msp10 and 49% for Mi-msp23-RNAi lines was observed. Similarly, production in egg masses decreased significantly by 76% (Mi-msp10) and 60% (Mi-msp23) for the RNAi lines, which eventually decreased the reproductive factor by 92% and 75%, respectively. The gene expression analysis showed a significant decrease in the transcript level by up to 72% (Mi-msp10) and 66% (Mi-msp23) in M. incognita females feeding on RNAi lines, providing further evidence of effective gene silencing. Overall, our findings provide useful information and support further development of RNAi-based strategies to control M. incognita.








Similar content being viewed by others
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- HD-RNAi:
-
Host-delivered RNAi
- PPN:
-
Plant–parasitic nematode
- RKN:
-
Root-knot nematode
References
Abad P, Gouzy J, Aury J-M et al (2008) Genome sequence of the metazoan plant-parasitic nematode Meloidogyne incognita. Nat Biotechnol 26:909–915. https://doi.org/10.1038/nbt.1482
Atkinson HJ, Lilley CJ, Urwin PE (2012) Strategies for transgenic nematode control in developed and developing world crops. Curr Opin Biotechnol 23:251–256. https://doi.org/10.1016/j.copbio.2011.09.004
Back MA, Haydock PPJ, Jenkinson P (2002) Disease complexes involving plant parasitic nematodes and soilborne pathogens. Plant Pathol 51:683–697. https://doi.org/10.1046/j.1365-3059.2002.00785.x
Bakhetia M, Charlton WL, Urwin PE et al (2005) RNA interference and plant parasitic nematodes. Trends Plant Sci 10:362–367. https://doi.org/10.1016/j.tplants.2005.06.007
Banerjee S, Banerjee A, Gill SS et al (2017) RNA interference: a novel source of resistance to combat plant parasitic nematodes. Front Plant Sci 8:834. https://doi.org/10.3389/fpls.2017.00834
Bellafiore S, Shen Z, Rosso MN et al (2008) Direct identification of the Meloidogyne incognita secretome reveals proteins with host cell reprogramming potential. PLoS Pathog 4:e1000192. https://doi.org/10.1371/journal.ppat.1000192
Berg RH, Fester T, Taylor CG (2009) Development of the root-knot nematode feeding cell. In: Berg RH, Taylor CG (eds) Cell biology of plant nematode parasitism. Springer, Berlin, pp 115–152. https://doi.org/10.1007/978-3-540-85215-5_5
Blok VC, Jones JT, Phillips MS, Trudgill DL (2008) Parasitism genes and host range disparities in biotrophic nematodes: the conundrum of polyphagy versus specialisation. BioEssays 30:249–259. https://doi.org/10.1002/bies.20717
Cabrera J, Díaz-Manzano FE, Fenoll C, Escobar C (2015) Developmental pathways mediated by hormones in nematode feeding sites. Adv Bot Res 73:167–188. https://doi.org/10.1016/bs.abr.2014.12.005
Chaudhary S, Dutta TK, Tyagi N et al (2019) Host-induced silencing of Mi-msp-1 confers resistance to root-knot nematode Meloidogyne incognita in eggplant. Transgenic Res 28:327–340. https://doi.org/10.1007/s11248-019-00126-5
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743. https://doi.org/10.1046/j.1365-313x.1998.00343.x
Danchin EGJ, Arguel M-J, Campan-Fournier A et al (2013) identification of novel target genes for safer and more specific control of root-knot nematodes from a pan-genome mining. PLoS Pathog 9:e1003745. https://doi.org/10.1371/journal.ppat.1003745
Davis EL, Hussey RS, Baum TJ et al (2000) Nematode parasitism genes. Annu Rev Phytopathol 38:365–396. https://doi.org/10.1146/annurev.phyto.38.1.365
Davis EL, Hussey RS, Mitchum MG, Baum TJ (2008) Parasitism proteins in nematode-plant interactions. Curr Opin Plant Biol 11:360–366
Dereeper A, Guignon V, Blanc G et al (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:W465-469. https://doi.org/10.1093/nar/gkn180
Dinh PTY, Brown CR, Elling AA (2014) RNA interference of effector gene Mc16D10L confers resistance against Meloidogyne chitwoodi in Arabidopsis and potato. Phytopathology 104:1098–1106. https://doi.org/10.1094/PHYTO-03-14-0063-R
Dinh PTY, Zhang L, Mojtahedi H et al (2015) Broad Meloidogyne resistance in potato based on RNA interference of effector gene 16D10. J Nematol 47:71–78
Dutta TK, Banakar P, Rao U (2014) The status of RNAi-based transgenic research in plant nematology. Front Microbiol 5:760. https://doi.org/10.3389/fmicb.2014.00760
Elling AA (2013) Major emerging problems with minor Meloidogyne species. Phytopathology 103:1092–1102. https://doi.org/10.1094/PHYTO-01-13-0019-RVW
García LE, Puerta MVS (2012) Characterization of a root-knot nematode population of Meloidogyne arenaria from Tupungato (Mendoza, Argentina). J Nematol 44:291–301
Gleason CA, Liu QL, Williamson VM (2008) Silencing a candidate nematode effector gene corresponding to the tomato resistance gene Mi-1 leads to acquisition of virulence. Mol Plant-Microbe Interact 21:576–585. https://doi.org/10.1094/MPMI-21-5-0576
Grishok A, Tabara H, Mello CC (2000) Genetic requirements for inheritance of RNAi in C. elegans. Science 287:2494–2497. https://doi.org/10.1126/science.287.5462.2494
Grossi-de-Sa M, Petitot A-S, Xavier DA et al (2019) Rice susceptibility to root-knot nematodes is enhanced by the Meloidogyne incognita MSP18 effector gene. Planta 250:1215–1227. https://doi.org/10.1007/s00425-019-03205-3
Hada A, Kumari C, Phani V et al (2020) Host-induced silencing of FMRFamide-like peptide genes, flp-1 and flp-12, in rice impairs reproductive fitness of the root-knot nematode Meloidogyne graminicola. Front Plant Sci 11:894. https://doi.org/10.3389/fpls.2020.00894
Hada A, Singh D, Papolu PK et al (2021) Host-mediated RNAi for simultaneous silencing of different functional groups of genes in Meloidogyne incognita using fusion cassettes in Nicotiana tabacum. Plant Cell Rep 40:2287–2302. https://doi.org/10.1007/s00299-021-02767-5
Haegeman A, Mantelin S, Jones JT, Gheysen G (2012) Functional roles of effectors of plant-parasitic nematodes. Gene 492:19–31. https://doi.org/10.1016/j.gene.2011.10.040
Hewezi T (2015) Cellular signaling pathways and posttranslational modifications mediated by nematode effector proteins. Plant Physiol 169:1018–1026. https://doi.org/10.1104/pp.15.00923
Hewezi T, Baum TJ (2013) Manipulation of plant cells by cyst and root-knot nematode effectors. Mol Plant Microbe Interact 26:9–16. https://doi.org/10.1094/MPMI-05-12-0106-FI
Höfgen R, Willmitzer L (1988) Storage of competent cells for Agrobacterium transformation. Nucleic Acids Res 16:9877. https://doi.org/10.1093/nar/16.20.9877
Huang G, Gao B, Maier T et al (2003) A profile of putative parasitism genes expressed in the esophageal gland cells of the root-knot nematode Meloidogyne incognita. Mol Plant Microbe Interact 16:376–381. https://doi.org/10.1094/MPMI.2003.16.5.376
Huang G, Allen R, Davis EL et al (2006) Engineering broad root-knot resistance in transgenic plants by RNAi silencing of a conserved and essential root-knot nematode parasitism gene. Proc Natl Acad Sci USA 103:14302–14306. https://doi.org/10.1073/pnas.0604698103
Hussey R (1989) Disease-inducing secretions of plant-parasitic nematodes. Annu Rev Phytopathol 27:123–141. https://doi.org/10.1146/annurev.phyto.27.1.123
Hussey RS, Mims CW (1991) Ultrastructure of feeding tubes formed in giant-cells induced in plants by the root-knot nematode Meloidogyne incognita. Protoplasma 162:99–107. https://doi.org/10.1007/BF02562553
Jaouannet M, Magliano M, Arguel MJ et al (2013) The root-knot nematode calreticulin Mi-CRT is a key effector in plant defense suppression. Mol Plant Microbe Interact 26:97–105. https://doi.org/10.1094/MPMI-05-12-0130-R
Jaubert S, Milac AL, Petrescu AJ et al (2005) In planta secretion of a calreticulin by migratory and sedentary stages of root-knot nematode. Mol Plant Microbe Interact 18:1277–1284. https://doi.org/10.1094/MPMI-18-1277
Jones MGK, Goto DB (2011) Root-knot nematodes and giant cells. In: Jones J, Gheysen G, Fenoll C (eds) Genomics and molecular genetics of plant-nematode interactions. Springer, Dordrecht, pp 83–100
Joshi I, Kumar A, Singh AK et al (2019) Development of nematode resistance in Arabidopsis by HD-RNAi-mediated silencing of the effector gene Mi-msp2. Sci Rep 9:17404. https://doi.org/10.1038/s41598-019-53485-8
Joshi I, Kumar A, Kohli D et al (2020) Conferring root-knot nematode resistance via host-delivered RNAi-mediated silencing of four Mi-msp genes in Arabidopsis. Plant Sci 298:110592. https://doi.org/10.1016/j.plantsci.2020.110592
Joshi I, Kohli D, Pal A, Chaudhury A, Sirohi A, Jain PK (2022) Host delivered-RNAi of effector genes for imparting resistance against root-knot and cyst nematodes in plants. Physiol Mol Plant Pathol 118:101802. https://doi.org/10.1016/j.pmpp.2022.101802
Kakrana A, Kumar A, Satheesh V et al (2017) Identification, validation and utilization of novel nematode-responsive root-specific promoters in Arabidopsis for inducing host-delivered RNAi mediated root-knot nematode resistance. Front Plant Sci 8:2049. https://doi.org/10.3389/fpls.2017.02049
Kohli D, Chidambaranathan P, Prasanth Tej Kumar J, Singh AK, Kumar A et al (2018) Host-mediated RNAi of a Notch-like receptor gene in Meloidogyne incognita induces nematode resistance. Parasitology 145:1896–1906. https://doi.org/10.1017/S003118201800064
Krogh A, Larsson B, von Heijne G, Sonnhammer ELL (2001) Predicting transmembrane protein topology with a hidden markov model: application to complete genomes. J Mol Biol 305:567–580. https://doi.org/10.1006/jmbi.2000.4315
Kumar A, Joshi I, Kohli D, Satheesh V, Abdin MZ, Sirohi A et al (2016) Characterization of root-knot nematode responsive and rootspecific promoter containing PIN domain from Arabidopsis thaliana (L.) Heynh. Indian J Genet Plant Breed 76:75–83. https://doi.org/10.5958/0975-6906.2016.00011.0
Kumar A, Kakrana A, Sirohi A et al (2017) Host-delivered RNAi-mediated root-knot nematode resistance in Arabidopsis by targeting splicing factor and integrase genes. J Gen Plant Pathol 83:91–97. https://doi.org/10.1007/s10327-017-0701-3
Kursel LE, Cope HD, Rog O (2021) Unconventional conservation reveals structure-function relationships in the synaptonemal complex. Elife 10:e72061. https://doi.org/10.7554/eLife.72061
Lisei-de-Sá ME, Rodrigues-Silva PL, Morgante CV, de Melo BP, Lourenço-Tessutti IT, Arraes FBM, Sousa JPA, Galbieri R, Amorim RMS, de Lins CBJ, Macedo LLP, Moreira VJ, Ferreira GF, Ribeiro TP, Fragoso RR, Silva MCM, de Almeida-Engler J, Grossi-de-Sa MF (2021) Pyramiding dsRNAs increases phytonematode tolerance in cotton plants. Planta 254:121. https://doi.org/10.1007/s00425-021-03776-0
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Majumdar R, Rajasekaran K, Cary JW (2017) RNA Interference (RNAi) as a potential tool for control of mycotoxin contamination in crop plants: concepts and considerations. Front Plant Sci 8:200. https://doi.org/10.3389/fpls.2017.00200
McNeilly TN, Frew D, Burgess STG et al (2017) Niche-specific gene expression in a parasitic nematode; increased expression of immunomodulators in Teladorsagia circumcincta larvae derived from host mucosa. Sci Rep 7:7214. https://doi.org/10.1038/s41598-017-07092-0
Mitchum MG, Hussey RS, Baum TJ et al (2013) Nematode effector proteins: an emerging paradigm of parasitism. New Phytol 199:879–894. https://doi.org/10.1111/nph.12323
Naalden D, Haegeman A, de Almeida-Engler J et al (2018) The Meloidogyne graminicola effector Mg16820 is secreted in the apoplast and cytoplasm to suppress plant host defense responses. Mol Plant Pathol 19:2416–2430. https://doi.org/10.1111/mpp.12719
Naito Y, Yamada T, Matsumiya T et al (2005) dsCheck: Highly sensitive off-target search software for double-stranded RNA-mediated RNA interference. Nucleic Acids Res 33:W589-591. https://doi.org/10.1093/nar/gki419
Nguyen C-N, Perfus-Barbeoch L, Quentin M et al (2018) A root-knot nematode small glycine and cysteine-rich secreted effector, MiSGCR1, is involved in plant parasitism. New Phytol 217:687–699. https://doi.org/10.1111/nph.14837
Niu J, Liu P, Liu Q et al (2016) Msp40 effector of root-knot nematode manipulates plant immunity to facilitate parasitism. Sci Rep 6:19443. https://doi.org/10.1038/srep19443
Oostenbrink M (1966) Major characteristics of the relation between nematodes and plants. Medeling/Laboratorium voor phytopathologie, serie nematodologie no. 42. Veenman, https://edepot.wur.nl/284956
Opperman CH, Bird DM, Williamson VM et al (2008) Sequence and genetic map of Meloidogyne hapla: a compact nematode genome for plant parasitism. Proc Natl Acad Sci USA 105:14802–14807. https://doi.org/10.1073/pnas.0805946105
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. https://doi.org/10.1038/nmeth.1701
Prentis PJ, Pavasovic A, Norton RS (2018) Sea anemones: quiet achievers in thefield of peptide toxins. Toxins (basel) 10:36. https://doi.org/10.3390/toxins10010036
Putker M, Madl T, Vos HR et al (2013) Redox-dependent control of FOXO/DAF-16 by transportin-1. Mol Cell 49:730–742. https://doi.org/10.1016/j.molcel.2012.12.014
Quevillon E, Silventoinen V, Pillai S et al (2005) InterProScan: protein domains identifier. Nucleic Acids Res 33:W116-120. https://doi.org/10.1093/nar/gki442
Rehman S, Gupta VK, Goyal AK (2016) Identification and functional analysis of secreted effectors from phytoparasitic nematodes. BMC Microbiol 16:48. https://doi.org/10.1186/s12866-016-0632-8
Roderick H, Urwin PE, Atkinson HJ (2018) Rational design of biosafe crop resistance to a range of nematodes using RNA interference. Plant Biotechnol J 16:520–529. https://doi.org/10.1111/pbi.12792
Rutter WB, Hewezi T, Abubucker S et al (2014) Mining novel effector proteins from the esophageal gland cells of Meloidogyne incognita. Mol Plant-Microbe Interact 27:965–974. https://doi.org/10.1094/MPMI-03-14-0076-R
Shi Q, Mao Z, Zhang X et al (2018) A Meloidogyne incognita effector MiISE5 suppresses programmed cell death to promote parasitism in host plant. Sci Rep 8:7256. https://doi.org/10.1038/s41598-018-24999-4
Siddique S, Grundler FM (2018) Parasitic nematodes manipulate plant development to establish feeding sites. Curr Opin Microbiol 46:102–108. https://doi.org/10.1016/j.mib.2018.09.004
Song H, Lin B, Huang Q et al (2021) The Meloidogyne javanica effector Mj2G02 interferes with jasmonic acid signalling to suppress cell death and promote parasitism in Arabidopsis. Mol Plant Pathol 22:1288–1301. https://doi.org/10.1111/mpp.13111
Süel KE, Chook YM (2009) Kap104p imports the PY-NLS-containing transcription factor Tfg2p into the nucleus. J Biol Chem 284:15416–15424. https://doi.org/10.1074/jbc.M809384200
Truong NM, Chen Y, Mejias J et al (2021) The Meloidogyne incognita nuclear effector MiEFF1 interacts with Arabidopsis cytosolic glyceraldehyde-3-phosphate dehydrogenases to promote parasitism. Front Plant Sci 12:641480. https://doi.org/10.3389/fpls.2021.641480
Vieira P, Gleason C (2019) Plant-parasitic nematode effectors—insights into their diversity and new tools for their identification. Curr Opin Plant Biol 50:37–43. https://doi.org/10.1016/j.pbi.2019.02.007
Vieira P, Shao J, Vijayapalani P et al (2020) A new esophageal gland transcriptome reveals signatures of large scale de novo effector birth in the root lesion nematode Pratylenchus penetrans. BMC Genomics 21:738. https://doi.org/10.1186/s12864-020-07146-0
Wang J, Replogle A, Hussey R et al (2011) Identification of potential host plant mimics of CLAVATA3/ESR (CLE)-like peptides from the plant-parasitic nematode Heterodera schachtii. Mol Plant Pathol 12:177–186. https://doi.org/10.1111/j.1364-3703.2010.00660.x
Wang X-R, Moreno YA, Wu H-R et al (2012) Proteomic profiles of soluble proteins from the esophageal gland in female Meloidogyne incognita. Int J Parasitol 42:1177–1183. https://doi.org/10.1016/j.ijpara.2012.10.008
Whitehead AG, Hemming JR (1965) A comparison of some quantitative methods of extracting small vermiform nematodes from soil. Ann Appl Biol 55:25–38. https://doi.org/10.1111/j.1744-7348.1965.tb07864.x
Xie J, Li S, Mo C et al (2016) A novel Meloidogyne incognita effector Misp12 suppresses plant defense response at latter stages of nematode parasitism. Front Plant Sci 7:964. https://doi.org/10.3389/fpls.2016.00964
Xue B, Hamamouch N, Li C et al (2013) The 8D05 parasitism gene of Meloidogyne incognita is required for successful infection of host roots. Phytopathology 103:175–181. https://doi.org/10.1094/PHYTO-07-12-0173-R
Yadav BC, Veluthambi K, Subramaniam K (2006) Host-generated double stranded RNA induces RNAi in plant-parasitic nematodes and protects the host from infection. Mol Biochem Parasitol 148:219–222. https://doi.org/10.1016/j.molbiopara.2006.03.013
Yamamura Y, Shim WB (2008) The coiled-coil protein-binding motif in Fusarium verticillioides Fsr1 is essential for maize stalk rot virulence. Microbiology 154:1637–1645. https://doi.org/10.1099/mic.0.2008/016782-0
Yang Y, Jittayasothorn Y, Chronis D et al (2013) Molecular characteristics and efficacy of 16D10 siRNAs in inhibiting root-knot nematode infection in transgenic grape hairy roots. PLoS One 8:e69463. https://doi.org/10.1371/journal.pone.0069463
Zhang L, Davies LJ, Elling AA (2015) A Meloidogyne incognita effector is imported into the nucleus and exhibits transcriptional activation activity in planta. Mol Plant Pathol 16:48–60. https://doi.org/10.1111/mpp.12160
Zhao J, Mejias J, Quentin M et al (2020) The root-knot nematode effector MiPDI1 targets a stress-associated protein (SAP) to establish disease in Solanaceae and Arabidopsis. New Phytol 228:1417–1430. https://doi.org/10.1111/nph.16745
Zhuo K, Chen J, Lin B et al (2017) A novel Meloidogyne enterolobii effector MeTCTP promotes parasitism by suppressing programmed cell death in host plants. Mol Plant Pathol 18:45–54. https://doi.org/10.1111/mpp.12374
Acknowledgements
The authors would like to express their gratitude to Dr. K. Subramaniam of IIT Kanpur for providing the RNAi vector construct and the staff of the National Phytotron Facility (NPF), IARI, New Delhi, India, for providing space for Arabidopsis plant experiments.
Funding
The present study was supported by a grant from the ICAR-NASF (formerly known as NFBSFARA) (code: NFBSFARA/RNA-3022) project during the years 2012–2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that we have no significant competing financial, professional, or personal interests that might have influenced the performance or presentation of the work described in this manuscript.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Suppl. Fig. S1
pBC-6 vector shows cloning of M. incognita genes in sense and antisense orientation driven by 35S promoter. a Mi-msp10 and b Mi-msp23 (JPG 97 KB)

Suppl. Fig. S2
Confirmation of RNAi lines using PCR. a Mi-msp10 and b Mi-msp23. E1, E2, E3 and E4 are independent RNAi lines; M, marker (JPG 206 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, A., Joshi, I., Changwal, C. et al. Host-delivered RNAi-mediated silencing of the root-knot nematode (Meloidogyne incognita) effector genes, Mi-msp10 and Mi-msp23, confers resistance in Arabidopsis and impairs reproductive ability of the root-knot nematode. Planta 256, 74 (2022). https://doi.org/10.1007/s00425-022-03977-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-022-03977-1