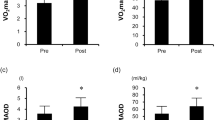
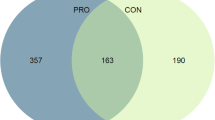
Abstract
Background
Gene expression is an important process underpinning the acute and chronic adaptive response to resistance exercise (RE) training.
Purpose
To investigate the effect of training status on vastus lateralis muscle global transcriptome at rest and following acute RE.
Methods
Muscle biopsies of nine young men (age: 26(2) years; body mass: 69(9) kg; height 172(6) cm) who undertook RE training for 10 weeks were collected pre and 24 h post-RE in the untrained (W1) and trained (W10) states and analysed using microarray. Tests of differential expression were conducted for rested and after RE contrasts in both training states. To control for false discovery rate (FDR), multiple testing correction was performed at a cut-off of FDR < 0.05.
Results
Unaccustomed RE (at W1) upregulated muscle gene transcripts related to stress (e.g., heat shock proteins), damage and inflammation, structural remodelling, protein turnover and increased translational capacity. Trained muscles (at W10) showed changes in the transcriptome signature regarding the regulation of energy metabolism, favouring a more oxidative one, upregulated antioxidant- and immune-related genes/terms, and gene transcripts related to the cytoskeleton and extracellular matrix, muscle contraction, development and growth.
Conclusions
These results highlight that chronic repetition of RE changes muscle transcriptome response towards a more refined response to RE-induced stress.





Similar content being viewed by others
Abbreviations
- FDR:
-
False discovery rate
- GO:
-
Gene ontology
- HSP:
-
Heat shock proteins
- mTOR:
-
Mechanistic target of rapamycin
- p70S6K:
-
p70 Ribosomal S6 kinase
- RE:
-
Resistance exercise
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- W1:
-
Untrained state
- W10:
-
Trained state
References
Adams GR, Hather BM, Baldwin KM, Dudley GA (1993) Skeletal muscle myosin heavy chain composition and resistance training. J Appl Physiol 74:911–915. https://doi.org/10.1152/jappl.1993.74.2.911
Ahtiainen JP et al (2016) Heterogeneity in resistance training-induced muscle strength and mass responses in men and women of different ages. Age 38:10. https://doi.org/10.1007/s11357-015-9870-1
Bellamy LM et al (2014) The acute satellite cell response and skeletal muscle hypertrophy following resistance training. PLoS One 9:e109739. https://doi.org/10.1371/journal.pone.0109739
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Stat Methodol 57:289–300
Bodine SC (2006) mTOR signaling and the molecular adaptation to resistance exercise. Med Sci Sports Exerc 38:1950–1957. https://doi.org/10.1249/01.mss.0000233797.24035.35
Chaillou T, Kirby TJ, McCarthy JJ (2014) Ribosome biogenesis: emerging evidence for a central role in the regulation of skeletal muscle mass. J Cell Physiol 229:1584–1594. https://doi.org/10.1002/jcp.24604
Chen TC (2006) Variability in muscle damage after eccentric exercise and the repeated bout effect Res. Q Exerc Sport 77:362–371. https://doi.org/10.1080/02701367.2006.10599370
Chen TC, Chen HL, Lin MJ, Wu CJ, Nosaka K (2009) Muscle damage responses of the elbow flexors to four maximal eccentric exercise bouts performed every 4 weeks. Eur J Appl Physiol 106:267–275. https://doi.org/10.1007/s00421-009-1016-7
Churchward-Venne TA, Tieland M, Verdijk LB, Leenders M, Dirks ML, de Groot LC, van Loon LJ (2015) There are no nonresponders to resistance-type exercise training in older men and women. J Am Med Dir Assoc 16:400–411. https://doi.org/10.1016/j.jamda.2015.01.071
Damas F, Phillips S, Vechin FC, Ugrinowitsch C (2015) A review of resistance training-induced changes in skeletal muscle protein synthesis and their contribution to hypertrophy. Sports Med 45:801–807. https://doi.org/10.1007/s40279-015-0320-0
Damas F, Nosaka K, Libardi CA, Chen TC, Ugrinowitsch C (2016a) Susceptibility to exercise-induced muscle damage: a cluster analysis with a large sample. Int J Sports Med 37:633–640. https://doi.org/10.1055/s-0042-100281
Damas F et al (2016b) Resistance training-induced changes in integrated myofibrillar protein synthesis are related to hypertrophy only after attenuation of muscle damage. J Physiol 594:5209–5222. https://doi.org/10.1113/JP272472
Damas F, Libardi CA, Ugrinowitsch C (2018a) The development of skeletal muscle hypertrophy through resistance training: the role of muscle damage and muscle protein synthesis. Eur J Appl Physiol 118:485–500. https://doi.org/10.1007/s00421-017-3792-9
Damas F et al (2018b) Early- and later-phases satellite cell responses and myonuclear content with resistance training in young men. PLoS One 13:e0191039. https://doi.org/10.1371/journal.pone.0191039
Dickinson JM et al (2018) Transcriptome response of human skeletal muscle to divergent exercise stimuli. J Appl Physiol 124:1529–1540. https://doi.org/10.1152/japplphysiol.00014.2018
Drummond MJ et al (2009) Expression of growth-related genes in young and older human skeletal muscle following an acute stimulation of protein synthesis. J Appl Physiol 106:1403–1411. https://doi.org/10.1152/japplphysiol.90842.2008
Figueiredo VC, Caldow MK, Massie V, Markworth JF, Cameron-Smith D, Blazevich AJ (2015) Ribosome biogenesis adaptation in resistance training-induced human skeletal muscle hypertrophy. Am J Physiol Endocrinol Metab 309:E72–E83. https://doi.org/10.1152/ajpendo.00050.2015
Gordon PM et al (2012) Resistance exercise training influences skeletal muscle immune activation: a microarray analysis. J Appl Physiol 112:443–453. https://doi.org/10.1152/japplphysiol.00860.2011
Grounds MD, Sorokin L, White J (2005) Strength at the extracellular matrix-muscle interface. Scand J Med Sci Sports 15:381–391. https://doi.org/10.1111/j.1600-0838.2005.00467.x
Gulbin JP, Gaffney PT (2002) Identical twins are discordant for markers of eccentric exercise-induced muscle damage. Int J Sports Med 23:471–476. https://doi.org/10.1055/s-2002-35076
Huber W et al (2015) Orchestrating high-throughput genomic analysis with. Bioconductor Nat Methods 12:115–121. https://doi.org/10.1038/nmeth.3252
Hyldahl RD et al (2015) Extracellular matrix remodeling and its contribution to protective adaptation following lengthening contractions in human muscle. FASEB J 29:2894–2904. https://doi.org/10.1096/fj.14-266668
Lamas L, Aoki MS, Ugrinowitsch C, Campos GE, Regazzini M, Moriscot AS, Tricoli V (2010) Expression of genes related to muscle plasticity after strength and power training regimens. Scand J Med Sci Sports 20:216–225. https://doi.org/10.1111/j.1600-0838.2009.00905.x
Leal ML, Lamas L, Aoki MS, Ugrinowitsch C, Ramos MS, Tricoli V, Moriscot AS (2011) Effect of different resistance-training regimens on the WNT-signaling pathway. Eur J Appl Physiol 111:2535–2545. https://doi.org/10.1007/s00421-011-1874-7
Liu D et al (2010) Skeletal muscle gene expression in response to resistance exercise: sex specific regulation. BMC Genom 11:659. https://doi.org/10.1186/1471-2164-11-659
Luo W, Friedman MS, Shedden K, Hankenson KD, Woolf PJ (2009) GAGE: generally applicable gene set enrichment for pathway analysis. BMC Bioinform 10:161. https://doi.org/10.1186/1471-2105-10-161
Mackey AL et al (2011) Sequenced response of extracellular matrix deadhesion and fibrotic regulators after muscle damage is involved in protection against future injury in human skeletal muscle. FASEB J 25:1943–1959. https://doi.org/10.1096/fj.10-176487
Mahoney DJ et al (2008) Gene expression profiling in human skeletal muscle during recovery from eccentric exercise. Am J Physiol Regul Integr Comp Physiol 294:R1901–R1910. https://doi.org/10.1152/ajpregu.00847.2007
Mayhew DL, Kim JS, Cross JM, Ferrando AA, Bamman MM (2009) Translational signaling responses preceding resistance training-mediated myofiber hypertrophy in young and old humans. J Appl Physiol 107:1655–1662. https://doi.org/10.1152/japplphysiol.91234.2008
Mitchell CJ et al (2014) Acute post-exercise myofibrillar protein synthesis is not correlated with resistance training-induced muscle hypertrophy in young men. PLoS One 9:e89431. https://doi.org/10.1371/journal.pone.0089431
Mobley CB et al (2018) Biomarkers associated with low, moderate, and high vastus lateralis muscle hypertrophy following 12 weeks of resistance training. PLoS One 13:e0195203. https://doi.org/10.1371/journal.pone.0195203
Ogata H, Goto S, Sato K, Fujibuchi W, Bono H, Kanehisa M (1999) KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res 27:29–34
Paulsen G, Crameri R, Benestad HB, Fjeld JG, Morkrid L, Hallen J, Raastad T (2010) Time course of leukocyte accumulation in human muscle after eccentric exercise. Med Sci Sports Exerc 42:75–85. https://doi.org/10.1249/MSS.0b013e3181ac7adb
Peake J, Nosaka K, Suzuki K (2005) Characterization of inflammatory responses to eccentric exercise in humans. Exerc Immunol Rev 11:64–85
Phillips SM, Tipton KD, Ferrando AA, Wolfe RR (1999) Resistance training reduces the acute exercise-induced increase in muscle protein turnover. Am J Physiol 276:E118–E124
Phillips BE et al (2013) Molecular networks of human muscle adaptation to exercise and age. PLoS Genet 9:e1003389. https://doi.org/10.1371/journal.pgen.1003389
Raue U, Trappe TA, Estrem ST, Qian HR, Helvering LM, Smith RC, Trappe S (2012) Transcriptome signature of resistance exercise adaptations: mixed muscle and fiber type specific profiles in young and old adults. J Appl Physiol 112:1625–1636. https://doi.org/10.1152/japplphysiol.00435.2011
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47. https://doi.org/10.1093/nar/gkv007
Roth SM, Ferrell RE, Peters DG, Metter EJ, Hurley BF, Rogers MA (2002) Influence of age, sex, and strength training on human muscle gene expression determined by microarray. Physiol Genom 10:181–190. https://doi.org/10.1152/physiolgenomics.00028.2002
Salanova M et al (2015) Vibration mechanosignals superimposed to resistive exercise result in baseline skeletal muscle transcriptome profiles following chronic disuse in bed rest. Sci Rep 5:17027. https://doi.org/10.1038/srep17027
Shi W et al (2010a) Estimating the proportion of microarray probes expressed in an RNA sample. Nucleic Acids Res 38:2168–2176. https://doi.org/10.1093/nar/gkp1204
Shi W, Oshlack A, Smyth GK (2010b) Optimizing the noise versus bias trade-off for Illumina whole genome expression BeadChips. Nucleic Acids Res 38:e204. https://doi.org/10.1093/nar/gkq871
Smith ML, Baggerly KA, Bengtsson H, Ritchie ME, Hansen KD (2013) illuminaio: an open source IDAT parsing tool for Illumina microarrays F1000Res 2:264 https://doi.org/10.12688/f1000research.2-264.v1
Stec MJ, Mayhew DL, Bamman MM (2015) The effects of age and resistance loading on skeletal muscle ribosome biogenesis. J Appl Physiol 119:851–857. https://doi.org/10.1152/japplphysiol.00489.2015
Steelman CA, Recknor JC, Nettleton D, Reecy JM (2006) Transcriptional profiling of myostatin-knockout mice implicates Wnt signaling in postnatal skeletal muscle growth and hypertrophy. FASEB J 20:580–582. https://doi.org/10.1096/fj.05-5125fje
Tang JE, Perco JG, Moore DR, Wilkinson SB, Phillips SM (2008) Resistance training alters the response of fed state mixed muscle protein synthesis in young men. Am J Physiol Regul Integr Comp Physiol 294:R172–R178. https://doi.org/10.1152/ajpregu.00636.2007
Thalacker-Mercer AE, Dell’Italia LJ, Cui X, Cross JM, Bamman MM (2010) Differential genomic responses in old vs. young humans despite similar levels of modest muscle damage after resistance loading. Physiol Genomics 40:141–149. https://doi.org/10.1152/physiolgenomics.00151.2009
Thalacker-Mercer A, Stec M, Cui X, Cross J, Windham S, Bamman M (2013) Cluster analysis reveals differential transcript profiles associated with resistance training-induced human skeletal muscle hypertrophy. Physiol Genomics 45:499–507. https://doi.org/10.1152/physiolgenomics.00167.2012
Thompson HS, Maynard EB, Morales ER, Scordilis SP (2003) Exercise-induced HSP27, HSP70 and MAPK responses in human skeletal muscle. Acta Physiol Scand 178:61–72. https://doi.org/10.1046/j.1365-201X.2003.01112.x
Welle S, Tawil R, Thornton CA (2008) Sex-related differences in gene expression in human skeletal muscle. PLoS One 3:e1385. https://doi.org/10.1371/journal.pone.0001385
Wilkinson SB, Phillips SM, Atherton PJ, Patel R, Yarasheski KE, Tarnopolsky MA, Rennie MJ (2008) Differential effects of resistance and endurance exercise in the fed state on signalling molecule phosphorylation and protein synthesis in human muscle. J Physiol 586:3701–3717. https://doi.org/10.1113/jphysiol.2008.153916
Witard OC, Jackman SR, Breen L, Smith K, Selby A, Tipton KD (2014) Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise Am. J Clin Nutr 99:86–95. https://doi.org/10.3945/ajcn.112.055517
Wu D, Smyth GK (2012) Camera: a competitive gene set test accounting for inter-gene correlation. Nucleic Acids Res 40:e133. https://doi.org/10.1093/nar/gks461
Zierath JR, Wallberg-Henriksson H (2015) Looking ahead perspective: where will the future of exercise biology take us? Cell Metab 22:25–30. https://doi.org/10.1016/j.cmet.2015.06.015
Acknowledgements
We acknowledge all the volunteers that participated in this study. This study was supported by the São Paulo Research Foundation (FAPESP) Grants (#2013/21218-4 and #2017/04299-1 to CAL), CAPES-PROEX and Natural Science and Engineering Research Council (NSERC) of Canada (RGPIN-2015-04613 to SMP). SMP also acknowledges support from the Canada Research Chairs program. FD was supported by FAPESP Grants (#2012/24499-1, #2014/19594-0, #2016/24259-1 and #2018/13064-0). CU, HR and VT are supported by the National Council for Scientific and Technological Development (CNPq) Grant (CU: #303085/2015-0 and #448387/2014-0; HR: #307023/2014-1; VT: #310823/2013-7).
Author information
Authors and Affiliations
Contributions
FD had original idea of the study and the final study design was developed by FD, CU, CAL, MEL, FCV, VT, HR and SMP. Participants were recruited and trained at the School of Physical Education and Sport, University of São Paulo, by FD, MEL, FCV and PRJ. AJH and CM conducted the RNA preparation to the microarray procedure. HM performed the biostatistical procedures and analyses. All authors participated in the interpretation of the data. FD, HM and PRJ designed the figures and supplementary tables. FD wrote the first version of the manuscript. All authors contributed to the manuscript, reviewed it, and approved the content of the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflicts of interest financial or otherwise to declare.
Ethical standard
All procedures performed herein were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Communicated by Phillip D. Chilibeck.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Damas, F., Ugrinowitsch, C., Libardi, C.A. et al. Resistance training in young men induces muscle transcriptome-wide changes associated with muscle structure and metabolism refining the response to exercise-induced stress. Eur J Appl Physiol 118, 2607–2616 (2018). https://doi.org/10.1007/s00421-018-3984-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-018-3984-y