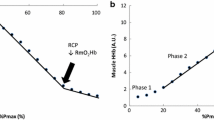
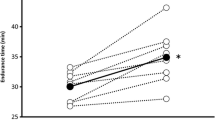
Abstract
Purpose
To non-invasively examine the effect of acute hypoxia and inspiratory threshold loading (ITL) on inspiratory muscles [sternocleidomastoid (SCM), scalene (SA) and parasternal (PS)] oxygenation in healthy adults using near-infrared spectroscopy (NIRS).
Methods
Twenty healthy adults (12 M/8 F) were randomly assigned to perform two ITL tests while breathing a normoxic or hypoxic (FIO2 = 15 %) gas mixture. NIRS devices were placed over the SCM, PS, SA, and a control muscle, tibialis anterior (TA), to monitor oxygenated (O2Hb), deoxygenated (HHb), total hemoglobin (tHb) and tissue saturation index (TSI). With the nose occluded, subjects breathed normally for 4 min through a mouthpiece that was connected to a weighted threshold loading device. ITL began by adding a 100-g weight to the ITL device. Then, every 2 min 50-g was added until task failure. Vital signs, ECG and ventilatory measures were monitored throughout the protocol.
Result
Participants were 31 ± 12 year and had normal spirometry. At task failure, the maximum load and ventilatory parameters did not differ between the hypoxic and normoxic ITL. At hypoxic ITL task failure, SpO2 was significantly lower, and ∆HHb increased more so in SA, SCM and PS than normoxic values. SCM ∆TSI decreased more so during hypoxic compared to normoxic ITL. ∆tHb in the inspiratory muscles (SCM, PS and SA) increased significantly compared to the decrease in TA during both hypoxic and normoxic ITL.
Conclusion
The SCM, an accessory inspiratory muscle was the most vulnerable to deoxygenation during incremental loading and this response was accentuated by acute hypoxia.



Similar content being viewed by others
Abbreviations
- FEV1 :
-
Forced expiratory volume in 1 s
- FEV1/FVC ratio:
-
Ratio of the forced expiratory volume in the first 1 s to the forced vital capacity of the lungs
- FIO2 :
-
Fraction of inspired oxygen
- FVC:
-
Forced vital capacity
- HHb:
-
Deoxygenated hemoglobin
- ITL:
-
Inspiratory threshold loading
- NIRS:
-
Near-infrared spectroscopy
- O2Hb:
-
Oxygenated hemoglobin
- PETCO2 :
-
End tidal carbon dioxide partial pressure
- PmPeak :
-
Peak mouth pressure
- PS:
-
Parasternal
- SA:
-
Scalene
- SCM:
-
Sternocleidomastoid
- SpO2 :
-
Saturation of hemoglobin with oxygen as measured by pulse oximetry
- TA:
-
Tibialis anterior
- tHb:
-
Total hemoglobin
- TSI:
-
Tissue saturation index
- VE :
-
Minute ventilation
- VT :
-
Tidal volume
References
Babcock MA, Johnson BD, Pegelow DF et al (1995) Hypoxic effects on exercise-induced diaphragmatic fatigue in normal healthy humans. J Appl Physiol 78:82–92
Balady GJ, Chaitman B, Driscoll D et al (1998) Recommendations for cardiovascular screening, staffing, and emergency policies at health/fitness facilities. Circulation 97:2283–2293
Borg GA (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14:377–381
Campbell EJM (1955) The role of the scalene and sternomastoid muscles in breathing in normal subjects. An electromyographic study. J Anat 89:378–386
Cannon DT, Grout SL, May CA et al (2007) Recruitment of the serratus anterior as an accessory muscle of ventilation during graded exercise. J Physiol Sci 57:127–131
Cvetko E, Karen P, Eržen I (2012) Myosin heavy chain composition of the human sternocleidomastoid muscle. Ann Anat 194:467–472
De Bisschop C, Beloka S, Groepenhoff H et al (2014) Is there a competition for oxygen availability between respiratory and limb muscles? Respir Physiol Neurobiol 196:8–16
De Troyer A, Estenne M (1984) Coordination between rib cage muscles and diaphragm during quiet breathing in humans. J Appl Physiol 57:899–906
Dubois P, Jamart J, Machiels J et al (1994) Prognosis of severely hypoxemic patients receiving long-term oxygen therapy. Chest 105:469–474
Elcadi GH, Forsman M, Crenshaw AG (2011) The relationship between oxygenation and myoelectric activity in the forearm and shoulder muscles of males and females. Eur J Appl Physiol 111:647–658
Farkas GA, McCormick KM, Gosselin LE (2007) Episodic hypoxia exacerbates respiratory muscle dysfunction in DMD(mdx) mice. Muscle Nerve 36:708–710
Gorman RB, McKenzie DK, Gandevia SC (1999) Task failure, breathing discomfort and CO2 accumulation without fatigue during inspiratory resistive loading in humans. Respir Physiol 115:273–286
Houssiere A, Najem B, Ciarka A et al (2005) Chemoreflex and metaboreflex control during static hypoxic exercise. Am J Physiol Heart Circ Physiol 288:H1724–H1729
Hudson AL, Gandevia SC, Butler JE (2007) The effect of lung volume on the co-ordinated recruitment of scalene and sternomastoid muscles in humans. J Physiol 584:261–270
Hussain SN (1996) Regulation of ventilatory muscle blood flow. J Appl Physiol 81(4):1455–1468
Jardim J, Farkas G, Prefaut C et al (1981) The failing inspiratory muscles under normoxic and hypoxic conditions. Am Rev Respir Dis 124:274–279
Johnson MA, Polgar J, Weightman D, Appleton D (1973) Data on the distribution of fibre types in thirty-six human muscles. An autopsy study. J Neurol Sci 18:111–129
Katayama K, Suzuki Y, Hoshikawa M et al (2015) Hypoxia exaggerates inspiratory accessory muscle deoxygenation during hyperpnoea. Resp Physiol Neurobiol 211:1–8
Legrand A, Schneider E, Gevenois P-A, De Troyer A (2003) Respiratory effects of the scalene and sternomastoid muscles in humans. J Appl Physiol 94:1467–1472
Mal H, Armenqaud MHAM (1988) Clinical importance of inspiratory muscle fatigue in acutely ill patients. J Crit Care 3:267–274
Mathur S, Sheel AW, Road JD, Reid WD (2010) Delayed onset muscle soreness after inspiratory threshold loading in healthy adults. Cardiopulm Phys Ther J 21:5–12
Mayock DE, Standaert TA, Woodrum DE (1992) Effect of inspiratory resistive loaded breathing and hypoxemia on diaphragmatic function in the piglet. J Appl Physiol 73:1888–1893
McKenzie DK, Allen GM, Butler JE, Gandevia SC (1997) Task failure with lack of diaphragm fatigue during inspiratory resistive loading in human subjects. J Appl Physiol 82:2011–2019
Miller MR, Hankinson J, Brusasco V et al (2005) Standardisation of spirometry. Eur Respir J 26:319–338
Miura H, McCully K, Nioka S, Chance B (2004) Relationship between muscle architectural features and oxygenation status determined by near infrared device. Eur J Appl Physiol 91:273–278
Mizuno M, Secher NH (1989) Histochemical characteristics of human expiratory and inspiratory intercostal muscles. J Appl Physiol 67:592–598
Moalla W, Merzouk A, Costes F et al (2006) Muscle oxygenation and EMG activity during isometric exercise in children. J Sports Sci 24:1195–1201
Murthy G, Hargens AR, Lehman S, Rempel DM (2001) Ischemia causes muscle fatigue. J Orthop Res 19:436–440
Parthasarathy S, Jubran A, Laghi F, Tobin MJ (2007) Sternomastoid, rib cage, and expiratory muscle activity during weaning failure. J Appl Physiol 103:140–147
Reid W, Yamabayashi C, Sheel AW, Shadgan B, Road JD (2013) Muscle oxygenation not maintained in sternomastoid during incremental inspiratory loading in COPD patients. Cardiopulm Phys Ther J 24(4):36
Robertson JCH, Foster GH, Johnson JRL (1977) The relationship of respiratory failure to the oxygen consumption of, lactate production by, and distribution of blood flow among respiratory muscles during increasing inspiratory resistance. J Clin Invest 59(1):31–42
Rohrbach M, Perret C, Kayser B, Boutellier U, Spengler CM (2003) Task failure from inspiratory resistive loaded breathing: a role for inspiratory muscle fatigue? Eur J Appl Physiol 90:405–410
Roussos C (1985) Function and fatigue of respiratory muscles. Chest 88:124S–132S
Saboisky JP, Gorman RB, De Troyer A et al (2007) Differential activation among five human inspiratory motoneuron pools during tidal breathing. J Appl Physiol 102:772–780
Sayli O, Akin A, Cotuk H et al (2014) Correlation analysis between surface electromyography and continuous wave near infrared spectroscopy parameters during isometric exercise to volitional fatigue. Turk J Electr Eng Comput Sci 22:780–793
Segizbaeva MO, Donina ZA, Timofeev NN et al (2013) EMG analysis of human inspiratory muscle resistance to fatigue during exercise. Adv Exp Med Biol 788:197–205
Shadgan B, Guenette JA, Sheel AW, Reid WD (2011) Sternocleidomastoid muscle deoxygenation in response to incremental inspiratory threshold loading measured by near infrared spectroscopy. Respir Physiol Neurobiol 178:202–209
Sheel AW, Derchak PA, Morgan BJ et al (2001) Fatiguing inspiratory muscle work causes reflex reduction in resting leg blood flow in humans. J Physiol 537:277–289
Taelman J, Vanderhaegen J, Robijns M et al (2011) Estimation of muscle fatigue using surface electromyography and near-infrared spectroscopy. Adv Exp Med Biol. pp 353–359
Verges S, Bachasson D, Wuyam B (2010) Effect of acute hypoxia on respiratory muscle fatigue in healthy humans. Respir Res 11:109
Vogiatzis I, Georgiadou O, Koskolou M et al (2007) Effects of hypoxia on diaphragmatic fatigue in highly trained athletes. J Physiol 581:299–308
Vogiatzis I, Athanasopoulos D, Habazettl H et al (2009) Intercostal muscle blood flow limitation in athletes during maximal exercise. J Physiol 587:3665–3677
Wahr JA, Tremper KK, Samra S, Delpy DT (1996) Near-infrared spectroscopy: theory and applications. J Cardiothorac Vasc Anesth 10:406–418
Wang L, Yoshikawa T, Hara T et al (2006) Which common NIRS variable reflects muscle estimated lactate threshold most closely? Appl Physiol Nutr Metab 31:612–620
Yamada E, Kusaka T, Arima N et al (2008) Relationship between muscle oxygenation and electromyography activity during sustained isometric contraction. Clin Physiol Funct Imaging 28:216–221
Yamamoto Y, Hoshikawa Y, Miyashita M (1996) Effects of acute exposure to simulated altitude on heart rate variability during exercise. J Appl Physiol 81(3):1223–1229
Zattara-Hartmann MC, Jammes Y (1996) Cardiorespiratory response to progressive leg exercise under acute normobaric hypoxia. Arch Physiol Biochem 104:272–281
Acknowledgments
We thank all subjects for their participation. We are also very appreciative of the valuable assistance given by Carmen Ma during data collection and Dr. Salem Bahubeshi for ECG monitoring. This study was supported by a grant and scholarship funding from Princess Nora bint Abdul Rahman University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Jean-René Lacour.
Rights and permissions
About this article
Cite this article
Basoudan, N., Shadgan, B., Guenette, J.A. et al. Effect of acute hypoxia on inspiratory muscle oxygenation during incremental inspiratory loading in healthy adults. Eur J Appl Physiol 116, 841–850 (2016). https://doi.org/10.1007/s00421-016-3334-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-016-3334-x