Abstract
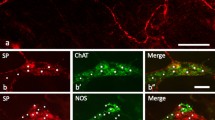
Enteric co-innervation is a peculiar innervation pattern of striated esophageal musculature. Both anatomical and functional data on enteric co-innervation related to various transmitters have been collected in different species, although its function remains enigmatic. However, it is unclear whether catecholaminergic components are involved in such a co-innervation. Thus, we examined to identify catecholaminergic neuronal elements and clarify their relationship to other innervation components in the esophagus, using immunohistochemistry with antibodies against tyrosine hydroxylase (TH), vesicular acetylcholine transporter (VAChT), choline acetyltransferase (ChAT) and protein gene product 9.5 (PGP 9.5), α-bungarotoxin (α-BT) and PCR with primers for amplification of cDNA encoding TH and dopamine-β-hydroxylase (DBH). TH-positive nerve fibers were abundant throughout the myenteric plexus and localized on about 14 % of α-BT-labelled motor endplates differing from VAChT-positive vagal nerve terminals. TH-positive perikarya represented a subpopulation of only about 2.8 % of all PGP 9.5-positive myenteric neurons. Analysis of mRNA showed both TH and DBH transcripts in the mouse esophagus. As ChAT-positive neurons in the compact formation of the nucleus ambiguus were negative for TH, the TH-positive nerve varicosities on motor endplates are presumably of enteric origin, although a sympathetic origin cannot be excluded. In the medulla oblongata, the cholinergic ambiguus neurons were densely supplied with TH-positive varicosities. Thus, catecholamines may modulate vagal motor innervation of esophageal-striated muscles not only at the peripheral level via enteric co-innervation but also at the central level via projections to the nucleus ambiguus. As Parkinson’s disease, with a loss of central dopaminergic neurons, also affects the enteric nervous system and dysphagia is prevalent in patients with this disease, investigation of intrinsic catecholamines in the esophagus may be worthwhile to understand such a symptom.



Similar content being viewed by others
References
Anderson G, Noorian AR, Taylor G, Anitha M, Bernhard D, Srinivasan S, Greene JG (2007) Loss of enteric dopaminergic neurons and associated changes in colon motility in an MPTP mouse model of Parkinson’s disease. Exp Neurol 207:4–12. doi:10.1016/j.expneurol.2007.05.010
Anlauf M, Schäfer MK, Eiden L, Weihe E (2003) Chemical coding of the human gastrointestinal nervous system: cholinergic, VIPergic, and catecholaminergic phenotypes. J Comp Neurol 459:90–111. doi:10.1002/cne.10599
Armstrong DM, Ross CA, Pickel VM, Joh TH, Reis DJ (1982) Distribution of dopamine-, noradrenaline-, and adrenaline-containing cell bodies in the rat medulla oblongata: demonstrated by the immunocytochemical localization of catecholamine biosynthetic enzymes. J Comp Neurol 212:173–187. doi:10.1002/cne.902120207
Baetge G, Gershon MD (1989) Transient catecholaminergic (TC) cells in the vagus nerves and bowel of fetal mice: relationship to the development of enteric neurons. Dev Biol 132:189–211
Baleriola J, Jean Y, Troy C, Hengst U (2015) Detection of axonally localized mRNAs in brain sections using high-resolution in situ hybridization. J Vis Exp 100:e52799. doi:10.3791/52799
Bieger D, Hopkins DA (1987) Viscerotopic representation of the upper alimentary tract in the medulla oblongata in the rat: the nucleus ambiguus. J Comp Neurol 262:546–562. doi:10.1002/cne.902620408
Blandini F, Balestra B, Levandis G, Cervio M, Greco R, Tassorelli C, Colucci M, Faniglione M, Bazzini E, Nappi G, Clavenzani P, Vigneri S, De Giorgio R, Tonini M (2009) Functional and neurochemical changes of the gastrointestinal tract in a rodent model of Parkinson’s disease. Neurosci Lett 467:203–207. doi:10.1016/j.neulet.2009.10.035
Braak H, de Vos RA, Bohl J, Del Tredici K (2006) Gastric alpha-synuclein immunoreactive inclusions in Meissner’s and Auerbach’s plexuses in cases staged for Parkinson’s disease-related brain pathology. Neurosci Lett 396:67–72. doi:10.1016/j.neulet.2005.11.012
Breuer C, Neuhuber WL, Wörl J (2004) Development of neuromuscular junctions in the mouse esophagus: morphology suggests a role for enteric coinnervation during maturation of vagal myoneural contacts. J Comp Neurol 475:47–69. doi:10.1002/cne.20156
Byrum CE, Guyenet PG (1987) Afferent and efferent connections of the A5 noradrenergic cell group in the rat. J Comp Neurol 261:529–542. doi:10.1002/cne.902610406
Cersosimo MG, Benarroch EE (2012) Pathological correlates of gastrointestinal dysfunction in Parkinson’s disease. Neurobiol Dis 46:559–564. doi:10.1016/j.nbd.2011.10.014
Cochard P, Goldstein M, Black IB (1978) Ontogenetic appearance and disappearance of tyrosine hydroxylase and catecholamines in the rat embryo. Proc Natl Acad Sci USA 75:2986–2990
Colucci M, Cervio M, Faniglione M, De Angelis S, Pajoro M, Levandis G, Tassorelli C, Blandini F, Feletti F, De Giorgio R, Dellabianca A, Tonini S, Tonini M (2012) Intestinal dysmotility and enteric neurochemical changes in a Parkinson’s disease rat model. Auton Neurosci 169:77–86. doi:10.1016/j.autneu.2012.04.005
Costa M, Furness JB, Gabella G (1971) Catecholamine containing nerve cells in the mammalian myenteric plexus. Histochemie 25:103–106
Del Tredici K, Braak H (2012) Lewy pathology and neurodegeneration in premotor Parkinson’s disease. Mov Disord 27:597–607. doi:10.1002/mds.24921
Derrey S, Chastan N, Maltete D, Verin E, Dechelotte P, Lefaucheur R, Proust F, Freger P, Leroi AM, Weber J, Gourcerol G (2015) Impact of deep brain stimulation on pharyngo-esophageal motility: a randomized cross-over study. Neurogastroenterol Motil 27:1214–1222. doi:10.1111/nmo.12607
Fantaguzzi CM, Thacker M, Chiocchetti R, Furness JB (2009) Identification of neuron types in the submucosal ganglia of the mouse ileum. Cell Tissue Res 336:179–189. doi:10.1007/s00441-009-0773-2
Flatmark T (2000) Catecholamine biosynthesis and physiological regulation in neuroendocrine cells. Acta Physiol Scand 168:1–17
Fritschy JM, Grzanna R (1990) Distribution of locus coeruleus axons within the rat brain-stem demonstrated by Phaseolus vulgaris leukoagglutinin anterograde tracing in combination with dopamine-beta-hydroxylase immunofluorescence. J Comp Neurol 293:616–631. doi:10.1002/cne.902930407
Furness JB (2000) Types of neurons in the enteric nervous system. J Auton Nerv Syst 81:87–96
Furness JB, Costa M (1971) Morphology and distribution of intrinsic adrenergic neurones in the proximal colon of the guinea-pig. Z Zellforsch Mikrosk Anat 120:346–363
Furness JB, Costa M, Freeman CG (1979) Absence of tyrosine hydroxylase activity and dopamine beta-hydroxylase immunoreactivity in intrinsic nerves of the guinea-pig ileum. Neuroscience 4:305–310
Gerbasi V, Lutsenko S, Lewis EJ (2003) A mutation in the ATP7B copper transporter causes reduced dopamine beta-hydroxylase and norepinephrine in mouse adrenal. Neurochem Res 28:867–873
Gershon MD, Chalazonitis A, Rothman TP (1993) From neural crest to bowel: development of the enteric nervous system. J Neurobiol 24:199–214. doi:10.1002/neu.480240207
Greene JG, Noorian AR, Srinivasan S (2009) Delayed gastric emptying and enteric nervous system dysfunction in the rotenone model of Parkinson’s disease. Exp Neurol 218:154–161. doi:10.1016/j.expneurol.2009.04.023
Gruber H, Zenker W (1978) Acetylcholinesterase activity in motor nerve fibres in correlation to muscle fibre types in rat. Brain Res 141:325–334
Gumy LF, Katrukha EA, Kapitein LC, Hoogenraad CC (2014) New insights into mRNA trafficking in axons. Dev Neurobiol 74:233–244. doi:10.1002/dneu.22121
Hawkes CH, Del Tredici K, Braak H (2007) Parkinson’s disease: a dual-hit hypothesis. Neuropathol Appl Neurobiol 33:599–614. doi:10.1111/j.1365-2990.2007.00874.x
Hempfling C, Neuhuber WL, Wörl J (2012) Serotonin-immunoreactive neurons and mast cells in the mouse esophagus suggest involvement of serotonin in both motility control and neuroimmune interactions. Neurogastroenterol Motil 24:e67–e78. doi:10.1111/j.1365-2982.2011.01797.x
Holmqvist S, Chutna O, Bousset L, Aldrin-Kirk P, Li W, Bjorklund T, Wang ZY, Roybon L, Melki R, Li JY (2014) Direct evidence of Parkinson pathology spread from the gastrointestinal tract to the brain in rats. Acta Neuropathol 128:805–820. doi:10.1007/s00401-014-1343-6
Jost WH (2010) Gastrointestinal dysfunction in Parkinson’s disease. J Neurol Sci 289:69–73. doi:10.1016/j.jns.2009.08.020
Kalf JG, de Swart BJ, Bloem BR, Munneke M (2012) Prevalence of oropharyngeal dysphagia in Parkinson’s disease: a meta-analysis. Parkinsonism Relat Disord 18:311–315. doi:10.1016/j.parkreldis.2011.11.006
Kalia M, Fuxe K, Goldstein M (1985) Rat medulla oblongata. II. Dopaminergic, noradrenergic (A1 and A2) and adrenergic neurons, nerve fibers, and presumptive terminal processes. J Comp Neurol 233:308–332. doi:10.1002/cne.902330303
Kallmünzer B, Sörensen B, Neuhuber WL, Wörl J (2006) Heterogeneity of neuromuscular junctions in striated muscle of human esophagus demonstrated by triple staining for the vesicular acetylcholine transporter, alpha-bungarotoxin, and acetylcholinesterase. Cell Tissue Res 324:181–188. doi:10.1007/s00441-005-0154-4
Karnovsky MJ, Roots L (1964) A “direct-coloring” thiocholine method for cholinesterases. J Histochem Cytochem 12:219–221
Khan MB, Lee BR, Kamitani T (2012) A simple and sensitive method for the demonstration of norepinephrine-storing adrenomedullary chromaffin cells. Histochem Cell Biol 138:155–165. doi:10.1007/s00418-012-0942-3
Kuhar MJ, Minneman K, Mull EC (2006) Catecholamines. In: Siegel GJ (ed) Basic neurochemistry, 7th edn. Academic Press/Elsevier, Amsterdam, pp 211–226
Lebouvier T, Chaumette T, Paillusson S, Duyckaerts C, Bruley des Varannes S, Neunlist M, Derkinderen P (2009) The second brain and Parkinson’s disease. Eur J Neurosci 30:735–741. doi:10.1111/j.1460-9568.2009.06873.x
Li ZS, Pham TD, Tamir H, Chen JJ, Gershon MD (2004) Enteric dopaminergic neurons: definition, developmental lineage, and effects of extrinsic denervation. J Neurosci 24:1330–1339. doi:10.1523/JNEUROSCI.3982-03.2004
Li Z, Caron MG, Blakely RD, Margolis KG, Gershon MD (2010) Dependence of serotonergic and other nonadrenergic enteric neurons on norepinephrine transporter expression. J Neurosci 30:16730–16740. doi:10.1523/JNEUROSCI.2276-10.2010
Lloyd RV, Sisson JC, Shapiro B, Verhofstad AA (1986) Immunohistochemical localization of epinephrine, norepinephrine, catecholamine-synthesizing enzymes, and chromogranin in neuroendocrine cells and tumors. Am J Pathol 125:45–54
Massari VJ, Dickerson LW, Gray AL, Lauenstein JM, Blinder KJ, Newsome JT, Rodak DJ, Fleming TJ, Gatti PJ, Gillis RA (1998) Neural control of left ventricular contractility in the dog heart: synaptic interactions of negative inotropic vagal preganglionic neurons in the nucleus ambiguus with tyrosine hydroxylase immunoreactive terminals. Brain Res 802:205–220
Nagatsu T, Levitt M, Udenfriend S (1964) Tyrosine hydroxylase. The initial step in norepinephrine biosynthesis. J Biol Chem 239:2910–2917
Noorian AR, Taylor GM, Annerino DM, Greene JG (2011) Neurochemical phenotypes of myenteric neurons in the rhesus monkey. J Comp Neurol 519:3387–3401. doi:10.1002/cne.22679
Ny L, Alm P, Ekstrom P, Hannibal J, Larsson B, Andersson KE (1994) Nitric oxide synthase-containing, peptide-containing, and acetylcholinesterase-positive nerves in the cat lower oesophagus. Histochem J 26:721–733
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Pfeiffer RF (2003) Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol 2:107–116
Pfeiffer RF (2011) Gastrointestinal dysfunction in Parkinson’s disease. Parkinsonism Relat Disord 17:10–15. doi:10.1016/j.parkreldis.2010.08.003
Phillips JK, Dubey R, Sesiashvilvi E, Takeda M, Christie DL, Lipski J (2001) Differential expression of the noradrenaline transporter in adrenergic chromaffin cells, ganglion cells and nerve fibres of the rat adrenal medulla. J Chem Neuroanat 21:95–104
Phillips RJ, Walter GC, Wilder SL, Baronowsky EA, Powley TL (2008) Alpha-synuclein-immunopositive myenteric neurons and vagal preganglionic terminals: autonomic pathway implicated in Parkinson’s disease? Neuroscience 153:733–750. doi:10.1016/j.neuroscience.2008.02.074
Phillips RJ, Hudson CN, Powley TL (2013) Sympathetic axonopathies and hyperinnervation in the small intestine smooth muscle of aged Fischer 344 rats. Auton Neurosci 179:108–121. doi:10.1016/j.autneu.2013.09.002
Qu ZD, Thacker M, Castelucci P, Bagyanszki M, Epstein ML, Furness JB (2008) Immunohistochemical analysis of neuron types in the mouse small intestine. Cell Tissue Res 334:147–161. doi:10.1007/s00441-008-0684-7
Raab M, Neuhuber WL (2004) Intraganglionic laminar endings and their relationships with neuronal and glial structures of myenteric ganglia in the esophagus of rat and mouse. Histochem Cell Biol 122:445–459. doi:10.1007/s00418-004-0703-z
Sang Q, Young HM (1998) The origin and development of the vagal and spinal innervation of the external muscle of the mouse esophagus. Brain Res 809:253–268
Schemann M, Schaaf C, Mader M (1995) Neurochemical coding of enteric neurons in the guinea pig stomach. J Comp Neurol 353:161–178. doi:10.1002/cne.903530202
Shiina T, Shimizu Y, Boudaka A, Wörl J, Neuhuber WL, Takewaki T (2007) Local neural regulation of the motility of the striated muscle portion in the mammalian esophagus. Curr Top Pharmacol 11:11–17
Shults CW (2006) Lewy bodies. Proc Natl Acad Sci USA 103:1661–1668. doi:10.1073/pnas.0509567103
Tan LL, Bornstein JC, Anderson CR (2010) The neurochemistry and innervation patterns of extrinsic sensory and sympathetic nerves in the myenteric plexus of the C57Bl6 mouse jejunum. Neuroscience 166:564–579. doi:10.1016/j.neuroscience.2009.12.034
Ter Horst GJ, Toes GJ, Van Willigen JD (1991) Locus coeruleus projections to the dorsal motor vagus nucleus in the rat. Neuroscience 45:153–160
Toti L, Travagli RA (2014) Gastric dysregulation induced by microinjection of 6-OHDA in the substantia nigra pars compacta of rats is determined by alterations in the brain-gut axis. Am J Physiol Gastrointest Liver Physiol 307:G1013–G1023. doi:10.1152/ajpgi.00258.2014
Uddman R, Grunditz T, Luts A, Desai H, Fernstrom G, Sundler F (1995) Distribution and origin of the peripheral innervation of rat cervical esophagus. Dysphagia 10:203–212
Wakabayashi K, Takahashi H, Takeda S, Ohama E, Ikuta F (1988) Parkinson’s disease: the presence of Lewy bodies in Auerbach’s and Meissner’s plexuses. Acta Neuropathol 76:217–221
Wakabayashi K, Takahashi H, Ohama E, Ikuta F (1989) Tyrosine hydroxylase-immunoreactive intrinsic neurons in the Auerbach’s and Meissner’s plexuses of humans. Neurosci Lett 96:259–263
Wattchow DA, Furness JB, Costa M (1988) Distribution and coexistence of peptides in nerve fibers of the external muscle of the human gastrointestinal tract. Gastroenterology 95:32–41
Wörl J, Neuhuber WL (2005) Enteric co-innervation of motor endplates in the esophagus: state of the art ten years after. Histochem Cell Biol 123:117–130. doi:10.1007/s00418-005-0764-7
Wörl J, Mayer B, Neuhuber WL (1994) Nitrergic innervation of the rat esophagus: focus on motor endplates. J Auton Nerv Syst 49:227–233
Wörl J, Dutsch F, Neuhuber WL (2002) Development of neuromuscular junctions in the mouse esophagus: focus on establishment and reduction of enteric co-innervation. Anat Embryol (Berl) 205:141–152. doi:10.1007/s00429-002-0239-8
Zhao WJ, Sun QJ, Lung MS, Birch D, Guo RC, Pilowsky PM (2011) Substance P, tyrosine hydroxylase and serotonin terminals in the rat caudal nucleus ambiguus. Respir Physiol Neurobiol 178:337–340. doi:10.1016/j.resp.2011.06.002
Zhu HC, Zhao J, Luo CY, Li QQ (2012) Gastrointestinal dysfunction in a Parkinson’s disease rat model and the changes of dopaminergic, nitric oxidergic, and cholinergic neurotransmitters in myenteric plexus. J Mol Neurosci 47:15–25. doi:10.1007/s12031-011-9560-0
Acknowledgments
The authors are indebted to Karin Löschner, Stephanie Link, Andrea Hilpert, Anita Hecht and Hedwig Symowski for their expert technical assistance. Special thanks go to an anonymous reviewer for his valuable comments and suggestions to improve the quality of the manuscript. The present work was performed in partial fulfillment of the requirements of the Friedrich-Alexander University of Erlangen-Nürnberg for obtaining the degree “Dr. rer. biol. hum.” This study was supported by DFG NE 534/3-1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van der Keylen, P., Garreis, F., Steigleder, R. et al. Involvement of catecholaminergic neurons in motor innervation of striated muscle in the mouse esophagus. Histochem Cell Biol 145, 573–585 (2016). https://doi.org/10.1007/s00418-015-1403-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-015-1403-6