Abstract
The existence of a distinct ganglionated myenteric plexus between the two layers of the striated tunica muscularis of the mammalian esophagus has represented an enigma for quite a while. Although an enteric co-innervation of vagally innervated motor endplates in the esophagus has been suggested repeatedly, it was not possible until recently to demonstrate this dual innervation. Twenty-two years ago, we were able to demonstrate that motor endplates in the rat esophagus receive dual innervation from both vagal nerve fibers originating in the brain stem and from varicose enteric nerve fibers originating in the myenteric plexus. Meanwhile, a considerable amount of data has been gathered on enteric co-innervation and its occurrence in the esophagus of a variety of species including humans, its neurochemistry, spatial relationships on motor endplates, ontogeny and possible functional roles. These data underline the significance of this newly discovered innervation component, although its function in vivo is still largely unknown. The aim of this review, which is an update of our previous paper (Wörl and Neuhuber in Histochem Cell Biol 123(2):117–130. doi:10.1007/s00418-005-0764-7, 2005a), is to summarize the current knowledge about enteric co-innervation of esophageal striated muscle and to provide some hints as to its functional significance.



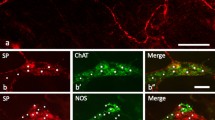
Modified from Breuer et al. (2004), with permission of the publisher



Similar content being viewed by others
Abbreviations
- ACh:
-
Acetylcholine
- AChE:
-
Acetylcholinesterase
- AChR:
-
Acetylcholine receptor
- AMB:
-
Nucleus ambiguus
- α-BT:
-
α-Bungarotoxin
- CGRP:
-
Calcitonin gene-related peptide
- CLR:
-
Calcitonin-receptor-like receptor
- cNTS:
-
Central subnucleus of the nucleus tractus solitarii
- CS:
-
Cryosections
- Co rate:
-
Co-innervation rate
- DiI:
-
1,1′-Dioleyl-3,3,3′,3′-tetramethylindocarbocyanine methane sulfonate
- DBH:
-
Dopamine-β-hydroxylase
- DMX:
-
Dorsal motor nucleus of the vagus nerve
- DRG:
-
Dorsal root ganglion
- E17:
-
Embryonic day 17
- EH:
-
Enzyme histochemistry
- ENF:
-
Enteric nerve fibers
- GAL:
-
Galanin
- Glu:
-
Glutamate
- IGLEs:
-
Intraganglionic laminar endings
- IH:
-
Immunocytochemistry
- L-ENK:
-
Leu-enkephalin
- 3mo:
-
3 Months
- M-ENK:
-
Met-enkephalin
- MEP:
-
Motor endplate
- MF:
-
Muscle fiber
- MG:
-
Myenteric ganglion
- NADPH-d:
-
NADPH-diaphorase
- NKA:
-
Neurokinin A
- NOD:
-
Nodose ganglion
- nNOS:
-
Neuronal nitric oxide synthase
- NPY:
-
Neuropeptide Y
- P4, -7, -14:
-
Postnatal day 4, 7, 14
- PACAP:
-
Pituitary adenylyl cyclase-activating (poly)peptide
- RAMP1:
-
Receptor-activity-modifying protein 1
- SC:
-
Swallowing center
- SP:
-
Substance P
- Tm-i, Tm-o:
-
Tunica muscularis, inner and outer muscle layer
- Tmuc:
-
Tunica mucosa
- Tsubm:
-
Tela submucosa
- TpH-1 and -2:
-
Tryptophan hydroxylase 1 and 2
- TRPV1:
-
Transient receptor potential ion channel of the vanilloid type 1
- TH:
-
Tyrosine hydroxylase
- VAChT:
-
Vesicular acetylcholine transporter
- VIP:
-
Vasoactive intestinal (poly)peptide
- WM:
-
Whole mounts
References
Beyer SD (2005) Nitrerge Co-Innervation am pharyngo-ösophagealen Übergang bei der Ratte. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg
Bieger D, Hopkins DA (1987) Viscerotopic representation of the upper alimentary tract in the medulla oblongata in the rat: the nucleus ambiguus. J Comp Neurol 262:546–562
Boudaka A, Wörl J, Shiina T, Neuhuber WL, Kobayashi H, Shimizu Y, Takewaki T (2007a) Involvement of TRPV1-dependent and -independent components in the regulation of vagally induced contractions in the mouse esophagus. Eur J Pharmacol 556(1–3):157–165. doi:10.1016/j.eiphar.2006.11.005
Boudaka A, Wörl J, Shiina T, Saito S, Atoji Y, Kobayashi H, Shimizu Y, Takewaki T (2007b) Key role of mucosal primary afferents in mediating the inhibitory influence of capsaicin on vagally mediated contractions in the mouse esophagus. J Vet Med Sci 69(4):365–372
Boudaka A, Wörl J, Shiina T, Shimizu Y, Takewaki T, Neuhuber WL (2009) Galanin modulates vagally induced contractions in the mouse oesophagus. Neurogastroenterol Motil 21(2):180–188. doi:10.1111/j.1365-2982.2008.01224.x
Breuer C, Neuhuber WL, Wörl J (2004) Development of neuromuscular junctions in the mouse esophagus: morphology suggests a role for enteric co-innervation during maturation of vagal myoneural contacts. J Comp Neurol 475:47–69. doi:10.1002/cne.20156
Cecio A (1976) Further histophysiological observations on the lower esophagus of the rabbit. Cell Tissue Res 168(4):475–488
Chihara D, Romer AI, Bentzinger CF, Rudnicki MA, Krauss RS (2015) PAX7 is required for patterning the esophageal musculature. Skelet Muscle 5:39. doi:10.1186/s13395-015-0068-0
Floyd K (1973) Cholinesterase activity in sheep oesophageal muscle. J Anat 116(3):357–373
Furness JB (2006) The enteric nervous system. Blackwell, Oxford
Furness JB, Callaghan BP, Rivera LR, Cho HJ (2014) The enteric nervous system and gastrointestinal innervation: integrated local and central control. Adv Exp Med Biol 817:39–71. doi:10.1007/978-1-4939-0897-4_3
Goda Y, Davis GW (2003) Mechanisms of synapse assembly and disassembly. Neuron 40(2):243–264
Goetsch E (1910) The structure of the mammalian oesophagus. Am J Anat 10(1):1–40
Gopalakrishnan S, Comai G, Sambasivan R, Francou A, Kelly RG, Tajbakhsh S (2015) A cranial mesoderm origin for esophagus striated muscles. Dev Cell 34(6):694–704. doi:10.1016/j.devcel.2015.07.003
Gruber H (1968) Über die Struktur und Innervation der quergestreiften Muskulatur des Ösophagus der Ratte. Z Zellforsch 91:236–247
Gruber H (1978) Motor innervation of the striated oesophagus muscle. Part 1. Intramural distribution of the right and left vagus nerve in the rat esophagus as revealed by the glycogen depletion technique. J Neurol Sci 36:41–53
Hayakawa T, Kuwahara S, Maeda S, Tanaka K, Seki M (2008) Direct synaptic projections to the myenteric ganglion of the rat subdiaphragmatic esophagus from the dorsal motor nucleus of the vagus. Neurosci Res 61(4):368–374. doi:10.1016/j.neures.2008.04.004
Hempfling C, Seibold R, Shiina T, Heimler W, Neuhuber WL, Wörl J (2009) Enteric co-innervation of esophageal striated muscle fibers: a phylogenetic study. Auton Neurosci 151(2):135–141. doi:10.1016/j.autneu.2009.08.006
Hempfling C, Neuhuber WL, Wörl J (2012) Serotonin-immunoreactive neurons and mast cells in the mouse esophagus suggest involvement of serotonin in both motility control and neuroimmune interactions. Neurogastroenterol Motil 24(1):e67–e78. doi:10.1111/j.1365-2982.2011.01797.x
Horling L, Bunnett NW, Messlinger K, Neuhuber WL, Raab M (2014) Localization of receptors for calcitonin-gene-related peptide to intraganglionic laminar endings of the mouse esophagus: peripheral interaction between vagal and spinal afferents? Histochem Cell Biol 141(3):321–335. doi:10.1007/s00418-013-1162-1
Izumi N, Matsuyama H, Yamamoto Y, Atoji Y, Suzuki Y, Unno T, Takewaki T (2002) Morphological and morphometrical characteristics of the esophageal intrinsic nervous system in the golden hamster. Eur J Morphol 40(3):137–144
Izumi N, Matsuyama H, Ko M, Shimizu Y, Takewaki T (2003) Role of intrinsic nitrergic neurones on vagally mediated striated muscle contractions in the hamster oesophagus. J Physiol 551(Pt 1):287–294. doi:10.1113/jphysiol.2003.044669
Jean A (2001) Brain stem control of swallowing: neuronal network and cellular mechanisms. Physiol Rev 81(2):929–969
Jost WH (2010) Gastrointestinal dysfunction in Parkinson’s disease. J Neurol Sci 289(1–2):69–73. doi:10.1016/j.jns.2009.08.020
Jurica EJ (1926) Studies on the motility of the denervated mammalian esophagus. Am J Physiol 77:371–384
Kablar B, Tajbakhsh S, Rudnicki MA (2000) Transdifferentiation of esophageal smooth to skeletal muscle is myogenic bHLH factor-dependent. Development 127:1627–1639
Kallmünzer B, Sörensen B, Neuhuber WL, Wörl J (2006) Heterogeneity of neuromuscular junctions in striated muscle of human esophagus demonstrated by triple staining for the vesicular acetylcholine transporter, alpha-bungarotoxin, and acetylcholinesterase. Cell Tissue Res 324(2):181–188. doi:10.1007/s00441-005-0154-4
Kallmünzer B, Sörensen B, Neuhuber WL, Wörl J (2008) Enteric co-innervation of striated muscle fibres in human oesophagus. Neurogastroenterol Motil 20:597–610. doi:10.1111/j.1365-2982.2007.01075.x
Kaufmann P, Lierse W, Stark J, Stelzner F (1968) Die Muskelanordnung in der Speiseröhre. Ergeb Anat Entwicklungsgesch 40:5–34
Krauss RS, Chihara D, Romer AI (2016) Embracing change: striated-for-smooth muscle replacement in esophagus development. Skelet Muscle 6:27. doi:10.1186/s13395-016-0099-1
Kuramoto H, Endo Y (1995) Galanin-immunoreactive nerve terminals innervating the striated muscle fibers of the rat esophagus. Neurosci Lett 188:171–174
Kuramoto H, Kato Y, Sakamoto H, Endo Y (1996) Galanin-containing nerve terminals that are involved in a dual innervation of the striated muscles of the rat esophagus. Brain Res 734:186–192
Kuramoto H, Kawano H, Sakamoto H, Furness JB (1999) Motor innervation by enteric nerve fibers containing both nitric oxide synthase and galanin immunoreactivities in the striated muscle of the rat esophagus. Cell Tissue Res 295:241–245
Kuramoto H, Oomori Y, Murabayashi H, Kadowaki M, Karaki S, Kuwahara A (2004) Localization of neurokinin 1 receptor (NK1R) immunoreactivity in rat esophagus. J Comp Neurol 478(1):11–21. doi:10.1002/cne.20169
Lichtman JW, Colman H (2000) Synapse elimination and indelible memory. Neuron 25:269–278
Meyer GW, Austin RM, Brady CE, Castell DO (1986) Muscle anatomy of the human esophagus. J Clin Gastroenterol 8(2):131–134
Mittal RK (2016) Regulation and dysregulation of esophageal peristalsis by the integrated function of circular and longitudinal muscle layers in health and disease. Am J Physiol Gastrointest Liver Physiol 00182:02016. doi:10.1152/ajpgi.00182.2016
Morikawa S, Komuro T (1998) Distribution of myenteric NO neurons along the guinea-pig esophagus. J Auton Nerv Syst 74:91–99
Nakamori C, Shiina T, Shimizu Y (2012) Postnatal changes in vagal control of esophageal muscle contractions in rats. Life Sci 90(13–14):495–501. doi:10.1016/j.lfs.2012.01.004
Nakamura K, Hasegawa H (2007) Developmental role of tryptophan hydroxylase in the nervous system. Mol Neurobiol 35(1):45–54
Natale G, Pasquali L, Ruggieri S, Paparelli A, Fornai F (2008) Parkinson’s disease and the gut: a well known clinical association in need of an effective cure and explanation. Neurogastroenterol Motil 20(7):741–749. doi:10.1111/j.1365-2982.2008.01162.x
Neuhuber W, Bieger D (2013) Brainstem control of deglutition: Brainstem neural circuits and mediators regulating swallowing. In: Shaker R, Belafsky PC, Postma GN, Easterling C (eds) Principles of deglutition. Springer, New York, pp 89–113. doi:10.1007/978-1-4614-3794-9
Neuhuber WL, Wörl J, Berthoud H-R, Conte B (1994) NADPH-diaphorase-positive nerve fibers associated with motor endplates in the rat esophagus: new evidence for co-innervation of striated muscle by enteric neurons. Cell Tissue Res 276(1):23–30
Neuhuber WL, Kressel M, Stark A, Berthoud HR (1998) Vagal efferent and afferent innervation of the rat esophagus as demonstrated by anterograde DiI and DiA tracing: focus on myenteric ganglia. J Auton Nerv Syst 70(1–2):92–102
Neuhuber WL, Eichhorn U, Wörl J (2001) Enteric co-innervation of striated muscle fibers in the esophagus: just a “hangover”? Anat Rec 262:41–46
Neuhuber WL, Raab M, Berthoud H-R, Wörl J (2006) Innervation of the mammalian esophagus. Adv Anat Embryol Cell Biol 185:1–73
Oppel A (1897) Lehrbuch der vergleichenden mikroskopischen Anatomie der Wirbeltiere, vol 2. Fischer Verlag, Jena
Ottaviani G (1937/1938) Osservazioni istoanatomiche e sperimentali sulla innervazione dell’esofago di alcuni mammiferi. Z Zellforsch Mikrosk Anat 27:393–429
Patapoutian A, Wold BJ, Wagner RA (1995) Evidence for developmentally programmed transdifferentiation in mouse esophageal muscle. Science 270:1818–1820
Peghini PL, Pursnani KG, Gideon MR, Castell JA, Nierman J, Castell DO (1998) Proximal and distal esophageal contractions have similar manometric features. Am J Physiol 274:G325–G330
Powley TL, Mittal RK, Baronowsky EA, Hudson CN, Martin FN, McAdams JL, Mason JK, Phillips RJ (2013) Architecture of vagal motor units controlling striated muscle of esophagus: peripheral elements patterning peristalsis? Auton Neurosci 179(1–2):90–98. doi:10.1016/j.autneu.2013.08.069
Reddy T, Kablar B (2004) Evidence for the involvement of neurotrophins in muscle transdifferentiation and acetylcholine receptor transformation in the esophagus of Myf5(−/−):MyoD(−/−) and NT-3(−/−) embryos. Dev Dyn 231:683–692
Redfern PA (1970) Neuromuscular transmission in new-born rats. J Physiol 209:701–709
Reichel B (1998) Die Coinnervation motorischer Endplatten im Ösophagus der Ratte durch VIP- und NPY-positive enterische Neuronen. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg
Rishniw M, Xin HB, Deng KY, Kotlikoff MI (2003) Skeletal myogenesis in the mouse esophagus does not occur through transdifferentiation. Genesis 36(2):81–82
Rishniw M, Fisher PW, Doran RM, Meadows E, Klein WH, Kotlikoff MI (2007) Smooth muscle persists in the muscularis externa of developing and adult mouse esophagus. J Muscle Res Cell Motil 28(2–3):153–165. doi:10.1007/s10974-007-9112-y
Rodrigo J, Uttenthal LO, Peinado MA, Esteban FJ, Fernández AP, Serrano J, Martínez de Velasco J, Santacana M, Bentura ML, Martínez-Murillo R, Pedrosa JA (1998) Distribution of nitric oxide synthase in the esophagus of the cat and monkey. J Auton Nerv Syst 70:164–179
Samarasinghe DD (1972) Some observations on the innervation of the striated muscle in the mouse esophagus—an electron microscopy study. J Anat 112:173–184
Sang Q, Young HM (1997) Development of nicotinic receptor clusters and innervation accompanying the change in muscle phenotype in the mouse esophagus. J Comp Neurol 386:119–136
Sang Q, Ciampoli D, Greferath U, Sommer L, Young HM (1999) Innervation of the esophagus in mice that lack Mash-1. J Comp Neurol 408:1–10
Seibold R (1997) Vergleichende Anatomie der nitrergen Co-Innervation des Ösophagus. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg
Shiina T, Shimizu Y, Boudaka A, Wörl J, Takewaki T (2006) Tachykinins are involved in local reflex modulation of vagally mediated striated muscle contractions in the rat esophagus via tachykinin NK1 receptors. Neuroscience 139(2):495–503. doi:10.1016/j.neuroscience.2005.12.027
Shiina T, Shima T, Suzuki Y, Wörl J, Shimizu Y (2012) Neural regulation of esophageal striated muscle in the house musk shrew (Suncus murinus). Auton Neurosci 168(1–2):25–31. doi:10.1016/j.autneu.2012.01.003
Shiina T, Naitou K, Nakamori H, Suzuki Y, Horii K, Sano Y, Shimaoka H, Shimizu Y (2016) Serotonin-induced contractile responses of esophageal smooth muscle in the house musk shrew (Suncus murinus). Neurogastroenterol Motil. doi:10.1111/nmo.12863
Shima T, Shiina T, Naitou K, Nakamori H, Shimizu Y (2014) Functional roles of capsaicin-sensitive intrinsic neural circuit in the regulation of esophageal peristalsis in rats: in vivo studies using a novel method. Am J Physiol Gastrointest Liver Physiol 306(9):G811–G818. doi:10.1152/ajpgi.00250.2013
Sommer D (2013) Exzitatorische enterische Co-Innervation von quergestreifter Muskulatur im Mäuseösophagus. Dissertation, Friedrich-Alexander-University Erlangen-Nürnberg
Sörensen B (2001) Vergleichende Anatomie der nitrergen Co-Innervation von quergestreifter Ösophagusmuskulatur bei verschiedenen Spezies einschließlich des Menschen. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg
Storr M, Geisler F, Neuhuber WL, Schusdziarra V, Allescher HD (2001) Characterization of vagal input to the rat esophageal muscle. Auton Neurosci 91(1–2):1–9
Stratton CJ, Bayguinov Y, Sanders KM, Ward SM (2000) Ultrastructural analysis of the transdifferentiation of smooth muscle to skeletal muscle in the murine esophagus. Cell Tissue Res 301:283–298
Su PH, Wang TC, Wong ZR, Huang BM, Yang HY (2011) The expression of nestin delineates skeletal muscle differentiation in the developing rat esophagus. J Anat 218(3):311–323. doi:10.1111/j.1469-7580.2010.01331.x
Toyama T, Yokoyama I, Nishi K (1975) Effects of hexamethonium and other ganglionic blocking agents on electrical activity of the esophagus induced by vagal stimulation in the dog. Eur J Pharmacol 31(1):63–71
van der Keylen P (2016) Katecholaminerge Innervation der quergestreiften Muskulatur im Mäuse- und Rattenösophagus. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg
van der Keylen P, Garreis F, Steigleder R, Sommer D, Neuhuber WL, Wörl J (2016) Involvement of catecholaminergic neurons in motor innervation of striated muscle in the mouse esophagus. Histochem Cell Biol 145(5):573–585. doi:10.1007/s00418-015-1403-6
Vietze S (2000) Postnatale Entwicklung der nitrergen Co-Innervation von motorischen Endplatten im Ösophagus der Ratte. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg
Weisbrodt NW (1976) Neuromuscular organization of esophageal and pharyngeal motility. Arch Intern Med 136(5):524–531
Whitmore I (1983) The ultrastructure of oesophageal striated muscle in the guinea-pig and marmoset. Cell Tissue Res 234:365–376
Wörl J, Neuhuber WL (2000) Spatial and temporal organization of TrkB expression in the developing musculature of the mouse esophagus. Histochem Cell Biol 114(3):229–238
Wörl J, Neuhuber WL (2005a) Enteric co-innervation of motor endplates in the esophagus: state of the art ten years after. Histochem Cell Biol 123(2):117–130. doi:10.1007/s00418-005-0764-7
Wörl J, Neuhuber WL (2005b) Ultrastructural analysis of the smooth-to-striated transition zone in the developing mouse esophagus: emphasis on apoptosis of smooth and origin and differentiation of striated muscle cells. Dev Dyn 233(3):964–982. doi:10.1002/dvdy.20436
Wörl J, Mayer B, Neuhuber WL (1994) Nitrergic innervation of the rat esophagus: focus on motor endplates. J Auton Nerv Syst 49(3):227–233
Wörl J, Mayer B, Neuhuber WL (1997) Spatial relationships of enteric nerve fibers to vagal motor terminals and the sarcolemma in motor endplates of the rat esophagus. A confocal laser scanning and electron-microscopic study. Cell Tissue Res 287:113–118
Wörl J, Fischer J, Neuhuber WL (1998) Nonvagal origin of galanin-containing nerve terminals innervating striated muscle fibers of the rat esophagus. Cell Tissue Res 292:453–461
Wörl J, Dütsch F, Neuhuber WL (2002) Development of neuromuscular junctions in the mouse esophagus: focus on establishment and reduction of enteric co-innervation. Anat Embryol 205:141–152. doi:10.1007/s00429-002-0239-8
Wörl J, Breuer C, Neuhuber WL (2009) Deletion of Pax7 changes the tunica muscularis of the mouse esophagus from an entirely striated into a mixed phenotype. Dev Dyn 238:864–874. doi:10.1002/dvdy.21898
Wu M, Majewski M, Wojtkiewicz J, Vanderwinden JM, Adriaensen D, Timmermans JP (2003) Anatomical and neurochemical features of the extrinsic and intrinsic innervation of the striated muscle in the porcine esophagus: evidence for regional and species differences. Cell Tissue Res 311(3):289–297. doi:10.1007/s00441-002-0696-7
Zhao W, Dhoot GK (2000a) Both smooth and skeletal muscle precursors are present in foetal mouse oesophagus and they follow different differentiation pathways. Dev Dyn 218:587–602
Zhao W, Dhoot GK (2000b) Skeletal muscle precursors in mouse esophagus are determined during early fetal development. Dev Dyn 219:10–20
Zhou D-S, Desaki J, Komuro T (1996) Neuro-muscular junctions of longitudinal and circular muscle fibers of the guinea-pig esophagus and their relation to myenteric plexus. J Auton Nerv Syst 58:63–68
Acknowledgements
The research conducted in the authors’ laboratory has been supported by the “Johannes und Frieda Marohn-Stiftung” (Wö/04) and ELAN Fonds (04.08.07.1) to Dr. Christian Breuer and J. W., Erlangen, a travel grant to J. W. from the Chihiro and Kiyoko Yokochi Fund, Kanehara Ichiro Foundation in Tokyo, Japan and the Deutsche Forschungsgemeinschaft (NE 534/3-1).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Dr. Detlev Drenckhahn.
Rights and permissions
About this article
Cite this article
Neuhuber, W.L., Wörl, J. Enteric co-innervation of striated muscle in the esophagus: still enigmatic?. Histochem Cell Biol 146, 721–735 (2016). https://doi.org/10.1007/s00418-016-1500-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-016-1500-1