Abstract
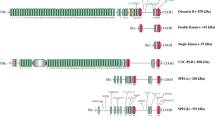
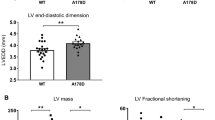
Obscurin comprises a family of giant modular proteins that play key structural and regulatory roles in striated muscles. Immunoglobulin domains 58/59 (Ig58/59) of obscurin mediate binding to essential modulators of muscle structure and function, including canonical titin, a smaller splice variant of titin, termed novex-3, and phospholamban (PLN). Importantly, missense mutations localized within the obscurin-Ig58/59 region that affect binding to titins and/or PLN have been linked to the development of myopathy in humans. To elucidate the pathophysiological role of this region, we generated a constitutive deletion mouse model, Obscn-ΔIg58/59, that expresses obscurin lacking Ig58/59, and determined the consequences of this manipulation on cardiac morphology and function under conditions of acute stress and through the physiological process of aging. Our studies show that young Obscn-ΔIg58/59 mice are susceptible to acute β-adrenergic stress. Moreover, sedentary Obscn-ΔIg58/59 mice develop left ventricular hypertrophy that progresses to dilation, contractile impairment, atrial enlargement, and arrhythmia as a function of aging with males being more affected than females. Experiments in ventricular cardiomyocytes revealed altered Ca2+ cycling associated with changes in the expression and/or phosphorylation levels of major Ca2+ cycling proteins, including PLN, SERCA2, and RyR2. Taken together, our work demonstrates that obscurin-Ig58/59 is an essential regulatory module in the heart and its deletion leads to age- and sex-dependent cardiac remodeling, ventricular dilation, and arrhythmia due to deregulated Ca2+ cycling.






Similar content being viewed by others
References
Ackermann MA, Shriver M, Perry NA, Hu LY, Kontrogianni-Konstantopoulos A (2014) Obscurins: goliaths and Davids take over non-muscle tissues. PLoS ONE 9:e88162. https://doi.org/10.1371/journal.pone.0088162
Akaike T, Du N, Lu G, Minamisawa S, Wang Y, Ruan H (2017) A sarcoplasmic reticulum localized protein phosphatase regulates phospholamban phosphorylation and promotes ischemia reperfusion injury in the heart. JACC Basic Transl Sci 2:160–180. https://doi.org/10.1016/j.jacbts.2016.12.002
Arimura T, Matsumoto Y, Okazaki O, Hayashi T, Takahashi M, Inagaki N, Hinohara K, Ashizawa N, Yano K, Kimura A (2007) Structural analysis of obscurin gene in hypertrophic cardiomyopathy. Biochem Biophys Res Commun 362:281–287. https://doi.org/10.1016/j.bbrc.2007.07.183
Bang ML, Centner T, Fornoff F, Geach AJ, Gotthardt M, McNabb M, Witt CC, Labeit D, Gregorio CC, Granzier H, Labeit S (2001) The complete gene sequence of titin, expression of an unusual approximately 700-kDa titin isoform, and its interaction with obscurin identify a novel Z-line to I-band linking system. Circ Res 89:1065–1072. https://doi.org/10.1161/hh2301.100981
Beckendorf J, van den Hoogenhof MMG, Backs J (2018) Physiological and unappreciated roles of CaMKII in the heart. Basic Res Cardiol 113:29. https://doi.org/10.1007/s00395-018-0688-8
Blenck CL, Harvey PA, Reckelhoff JF, Leinwand LA (2016) The importance of biological sex and estrogen in rodent models of cardiovascular health and disease. Circ Res 118:1294–1312. https://doi.org/10.1161/CIRCRESAHA.116.307509
Blondelle J, Marrocco V, Clark M, Desmond P, Myers S, Nguyen J, Wright M, Bremner S, Pierantozzi E, Ward S, Esteve E, Sorrentino V, Ghassemian M, Lange S (2019) Murine obscurin and Obsl1 have functionally redundant roles in sarcolemmal integrity, sarcoplasmic reticulum organization, and muscle metabolism. Commun Biol 2:178. https://doi.org/10.1038/s42003-019-0405-7
Borisov AB, Raeker MO, Kontrogianni-Konstantopoulos A, Yang K, Kurnit DM, Bloch RJ, Russell MW (2003) Rapid response of cardiac obscurin gene cluster to aortic stenosis: differential activation of Rho-GEF and MLCK and involvement in hypertrophic growth. Biochem Biophys Res Commun 310:910–918. https://doi.org/10.1016/j.bbrc.2003.09.035
Busby B, Willis CD, Ackermann MA, Kontrogianni-Konstantopoulos A, Bloch RJ (2010) Characterization and comparison of two binding sites on obscurin for small ankyrin 1. Biochemistry 49:9948–9956. https://doi.org/10.1021/bi101165p
Cerrone M, Montnach J, Lin X, Zhao YT, Zhang M, Agullo-Pascual E, Leo-Macias A, Alvarado FJ, Dolgalev I, Karathanos TV, Malkani K, Van Opbergen CJM, van Bavel JJA, Yang HQ, Vasquez C, Tester D, Fowler S, Liang F, Rothenberg E, Heguy A, Morley GE, Coetzee WA, Trayanova NA, Ackerman MJ, van Veen TAB, Valdivia HH, Delmar M (2017) Plakophilin-2 is required for transcription of genes that control calcium cycling and cardiac rhythm. Nat Commun 8:106. https://doi.org/10.1038/s41467-017-00127-0
DeGrande ST, Little SC, Nixon DJ, Wright P, Snyder J, Dun W, Murphy N, Kilic A, Higgins R, Binkley PF, Boyden PA, Carnes CA, Anderson ME, Hund TJ, Mohler PJ (2013) Molecular mechanisms underlying cardiac protein phosphatase 2A regulation in heart. J Biol Chem 288:1032–1046. https://doi.org/10.1074/jbc.M112.426957
Disertori M, Mase M, Ravelli F (2017) Myocardial fibrosis predicts ventricular tachyarrhythmias. Trends Cardiovasc Med 27:363–372. https://doi.org/10.1016/j.tcm.2017.01.011
Dobrev D, Wehrens XH (2014) Role of RyR2 phosphorylation in heart failure and arrhythmias: controversies around ryanodine receptor phosphorylation in cardiac disease. Circ Res 114:1311–1319. https://doi.org/10.1161/circresaha.114.300568(discussion 1319)
Eiringhaus J, Herting J, Schatter F, Nikolaev VO, Sprenger J, Wang Y, Kohn M, Zabel M, El-Armouche A, Hasenfuss G, Sossalla S, Fischer TH (2019) Protein kinase/phosphatase balance mediates the effects of increased late sodium current on ventricular calcium cycling. Basic Res Cardiol 114:13. https://doi.org/10.1007/s00395-019-0720-7
Erickson JR (2014) Mechanisms of CaMKII Activation in the Heart. Front Pharmacol 5:59. https://doi.org/10.3389/fphar.2014.00059
Feng Y, Cheng J, Wei B, Wang Y (2017) CaMKII inhibition reduces isoproterenol-induced ischemia and arrhythmias in hypertrophic mice. Oncotarget 8:17504–17509. https://doi.org/10.18632/oncotarget.15099
Fukuzawa A, Idowu S, Gautel M (2005) Complete human gene structure of obscurin: implications for isoform generation by differential splicing. J Muscle Res Cell Motil 26:427–434. https://doi.org/10.1007/s10974-005-9025-6
Gautel M (2011) Cytoskeletal protein kinases: titin and its relations in mechanosensing. Pflugers Arch 462:119–134. https://doi.org/10.1007/s00424-011-0946-1
Grogan A, Kontrogianni-Konstantopoulos A (2019) Unraveling obscurins in heart disease. Pflugers Arch 471:735–743. https://doi.org/10.1007/s00424-018-2191-3
Heijman J, Dewenter M, El-Armouche A, Dobrev D (2013) Function and regulation of serine/threonine phosphatases in the healthy and diseased heart. J Mol Cell Cardiol 64:90–98. https://doi.org/10.1016/j.yjmcc.2013.09.006
Hu LR, Ackermann MA, Hecker PA, Prosser BL, King B, O’Connell KA, Grogan A, Meyer LC, Berndsen CE, Wright NT, Jonathan Lederer W, Kontrogianni-Konstantopoulos A (2017) Deregulated Ca(2+) cycling underlies the development of arrhythmia and heart disease due to mutant obscurin. Sci Adv 3:e1603081. https://doi.org/10.1126/sciadv.1603081
Hu LY, Kontrogianni-Konstantopoulos A (2013) The kinase domains of obscurin interact with intercellular adhesion proteins. FASEB J 27:2001–2012. https://doi.org/10.1096/fj.12-221317
Huke S, Bers DM (2008) Ryanodine receptor phosphorylation at Serine 2030, 2808 and 2814 in rat cardiomyocytes. Biochem Biophys Res Commun 376:80–85. https://doi.org/10.1016/j.bbrc.2008.08.084
Kellermayer D, Smith JE 3rd, Granzier H (2017) Novex-3, the tiny titin of muscle. Biophys Rev 9:201–206. https://doi.org/10.1007/s12551-017-0261-y
Kontrogianni-Konstantopoulos A, Ackermann MA, Bowman AL, Yap SV, Bloch RJ (2009) Muscle giants: molecular scaffolds in sarcomerogenesis. Physiol Rev 89:1217–1267. https://doi.org/10.1152/physrev.00017.2009
Kontrogianni-Konstantopoulos A, Catino DH, Strong JC, Bloch RJ (2006) De novo myofibrillogenesis in C2C12 cells: evidence for the independent assembly of M bands and Z disks. Am J Physiol Cell Physiol 290:C626–C637. https://doi.org/10.1152/ajpcell.00442.2005
Kontrogianni-Konstantopoulos A, Catino DH, Strong JC, Sutter S, Borisov AB, Pumplin DW, Russell MW, Bloch RJ (2006) Obscurin modulates the assembly and organization of sarcomeres and the sarcoplasmic reticulum. FASEB J 20:2102–2111. https://doi.org/10.1096/fj.06-5761com
Kontrogianni-Konstantopoulos A, Jones EM, Van Rossum DB, Bloch RJ (2003) Obscurin is a ligand for small ankyrin 1 in skeletal muscle. Mol Biol Cell 14:1138–1148. https://doi.org/10.1091/mbc.e02-07-0411
Lange S, Ouyang K, Meyer G, Cui L, Cheng H, Lieber RL, Chen J (2009) Obscurin determines the architecture of the longitudinal sarcoplasmic reticulum. J Cell Sci 122:2640–2650. https://doi.org/10.1242/jcs.046193
Linke WA (2018) Titin Gene and protein functions in passive and active muscle. Annu Rev Physiol 80:389–411. https://doi.org/10.1146/annurev-physiol-021317-121234
Linke WA, Hamdani N (2014) Gigantic business: titin properties and function through thick and thin. Circ Res 114:1052–1068. https://doi.org/10.1161/CIRCRESAHA.114.301286
Louzao-Martinez L, Vink A, Harakalova M, Asselbergs FW, Verhaar MC, Cheng C (2016) Characteristic adaptations of the extracellular matrix in dilated cardiomyopathy. Int J Cardiol 220:634–646. https://doi.org/10.1016/j.ijcard.2016.06.253
MacDougall LK, Jones LR, Cohen P (1991) Identification of the major protein phosphatases in mammalian cardiac muscle which dephosphorylate phospholamban. Eur J Biochem 196:725–734. https://doi.org/10.1111/j.1432-1033.1991.tb15871.x
Mishra S, Sabbah HN, Jain JC, Gupta RC (2003) Reduced Ca2+-calmodulin-dependent protein kinase activity and expression in LV myocardium of dogs with heart failure. Am J Physiol Heart Circ Physiol 284:H876–H883. https://doi.org/10.1152/ajpheart.00266.2002
Murphy E, Steenbergen C (2014) Estrogen regulation of protein expression and signaling pathways in the heart. Biol Sex Differ 5:6. https://doi.org/10.1186/2042-6410-5-6
Netticadan T, Temsah RM, Kawabata K, Dhalla NS (2000) Sarcoplasmic reticulum Ca(2+)/Calmodulin-dependent protein kinase is altered in heart failure. Circ Res 86:596–605. https://doi.org/10.1161/01.res.86.5.596
Neumann J, Eschenhagen T, Jones LR, Linck B, Schmitz W, Scholz H, Zimmermann N (1997) Increased expression of cardiac phosphatases in patients with end-stage heart failure. J Mol Cell Cardiol 29:265–272. https://doi.org/10.1006/jmcc.1996.0271
Qadota H, Matsunaga Y, Bagchi P, Lange KI, Carrier KJ, Pols WV, Swartzbaugh E, Wilson KJ, Srayko M, Pallas DC, Benian GM (2018) Protein phosphatase 2A is crucial for sarcomere organization in Caenorhabditis elegans striated muscle. Mol Biol Cell 29:2084–2097. https://doi.org/10.1091/mbc.E18-03-0192
Qadota H, McGaha LA, Mercer KB, Stark TJ, Ferrara TM, Benian GM (2008) A novel protein phosphatase is a binding partner for the protein kinase domains of UNC-89 (Obscurin) in Caenorhabditis elegans. Mol Biol Cell 19:2424–2432. https://doi.org/10.1091/mbc.E08-01-0053
Raeker MO, Su F, Geisler SB, Borisov AB, Kontrogianni-Konstantopoulos A, Lyons SE, Russell MW (2006) Obscurin is required for the lateral alignment of striated myofibrils in zebrafish. Dev Dyn 235:2018–2029. https://doi.org/10.1002/dvdy.20812
Rossi D, Palmio J, Evila A, Galli L, Barone V, Caldwell TA, Policke RA, Aldkheil E, Berndsen CE, Wright NT, Malfatti E, Brochier G, Pierantozzi E, Jordanova A, Guergueltcheva V, Romero NB, Hackman P, Eymard B, Udd B, Sorrentino V (2017) A novel FLNC frameshift and an OBSCN variant in a family with distal muscular dystrophy. PLoS ONE 12:e0186642. https://doi.org/10.1371/journal.pone.0186642
Russell MW, Raeker MO, Korytkowski KA, Sonneman KJ (2002) Identification, tissue expression and chromosomal localization of human Obscurin-MLCK, a member of the titin and Dbl families of myosin light chain kinases. Gene 282:237–246. https://doi.org/10.1016/s0378-1119(01)00795-8
Schulman H, Anderson ME (2010) Ca/calmodulin-dependent protein kinase II in heart failure. Drug Discov Today Dis Mech 7:e117–e122. https://doi.org/10.1016/j.ddmec.2010.07.005
Shioya T (2007) A simple technique for isolating healthy heart cells from mouse models. J Physiol Sci 57:327–335. https://doi.org/10.2170/physiolsci.RP010107
Shriver M, Stroka KM, Vitolo MI, Martin S, Huso DL, Konstantopoulos K, Kontrogianni-Konstantopoulos A (2015) Loss of giant obscurins from breast epithelium promotes epithelial-to-mesenchymal transition, tumorigenicity and metastasis. Oncogene 34:4248–4259. https://doi.org/10.1038/onc.2014.358
Simmerman HK, Collins JH, Theibert JL, Wegener AD, Jones LR (1986) Sequence analysis of phospholamban. Identification of phosphorylation sites and two major structural domains. J Biol Chem 261:13333–13341
Wang L, Geist J, Grogan A, Hu LR, Kontrogianni-Konstantopoulos A (2018) Thick filament protein network, functions, and disease association. Compr Physiol 8:631–709. https://doi.org/10.1002/cphy.c170023
Wehrens XH, Lehnart SE, Reiken SR, Marks AR (2004) Ca2 +/calmodulin-dependent protein kinase II phosphorylation regulates the cardiac ryanodine receptor. Circ Res 94:e61–e70. https://doi.org/10.1161/01.RES.0000125626.33738.E2
Young P, Ehler E, Gautel M (2001) Obscurin, a giant sarcomeric Rho guanine nucleotide exchange factor protein involved in sarcomere assembly. J Cell Biol 154:123–136. https://doi.org/10.1083/jcb.200102110
Yuan Q, Fan GC, Dong M, Altschafl B, Diwan A, Ren X, Hahn HH, Zhao W, Waggoner JR, Jones LR, Jones WK, Bers DM, Dorn GW 2nd, Wang HS, Valdivia HH, Chu G, Kranias EG (2007) Sarcoplasmic reticulum calcium overloading in junction deficiency enhances cardiac contractility but increases ventricular automaticity. Circulation 115:300–309. https://doi.org/10.1161/CIRCULATIONAHA.106.654699
Acknowledgements
The authors wish to thank Dr. Maegen Ackermann for propagating the Obscn-ΔIg58/59 mouse model and Dr. Li-Yen Rebecca Hu for propagating the Obscn-ΔIg58/59 mouse model and performing pilot studies.
Funding
This work was supported by the National Institutes of Health [Training Program in Muscle Biology, T32 AR007592-17 to A.G. and A.C., R35HL144998 to H.G, and R01AR071618 and R01AR071614 to C.W.W.]; and the American Heart Association [Grant In Aid 16GRNT31290010 to A.K.K. and AHA 19POST34450156 to H.J.].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Grogan, A., Coleman, A., Joca, H. et al. Deletion of obscurin immunoglobulin domains Ig58/59 leads to age-dependent cardiac remodeling and arrhythmia. Basic Res Cardiol 115, 60 (2020). https://doi.org/10.1007/s00395-020-00818-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-020-00818-8