Abstract
Purpose
We aimed to examine the association between dietary patterns and type 2 diabetes mellitus (T2DM) while considering the potential effect modification by metabolic phenotypes (metabotypes). Additionally, we aimed to explore the association between dietary scores and prediabetes.
Methods
A total of 1460 participants (11.8% with T2DM) from the cross-sectional population-based KORA FF4 study were included. Participants, classified into three metabotype subgroups, had both their FSAm-NPS dietary index (underpinning the Nutri-Score) and ultra-processed foods (UPF) intake (using NOVA classification) calculated. Glucose tolerance status was assessed via oral glucose tolerance tests (OGTT) in non-diabetic participants and was classified according to the American Diabetes Association criteria. Logistic regression models were used for both the overall and metabotype-stratified analyses of dietary scores’ association with T2DM, and multinomial probit models for their association with prediabetes.
Results
Participants who had a diet with a higher FSAm-NPS dietary index (i.e., a lower diet quality) or a greater percentage of UPF consumption showed a positive association with T2DM. Stratified analyses demonstrated a strengthened association between UPF consumption and T2DM specifically in the metabolically most unfavorable metabotype (Odds Ratio, OR 1.92; 95% Confidence Interval, CI 1.35, 2.73). A diet with a higher FSAm-NPS dietary index was also positively associated with prediabetes (OR 1.19; 95% CI 1.04, 1.35).
Conclusion
Our study suggests different associations between poorer diet quality and T2DM across individuals exhibiting diverse metabotypes, pointing to the option for stratified dietary interventions in diabetes prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder that affects millions of people worldwide and poses a substantial burden to human health and economy [1, 2]. The global prevalence of diabetes in 2021 was estimated to be 10.5% (537 million people), projected to increase to 12.2% (783 million people) by 2045 [3]. T2DM is associated with various comorbidities, including cardiovascular disease, neuropathy, retinopathy, and kidney disease, leading to significant morbidity and mortality [4]. Prediabetes is a high-risk state for diabetes defined as blood glucose levels above normal but below diabetes thresholds [5]; up to 10% of individuals with prediabetes may develop T2DM annually [6]. Existing metabolic abnormalities during the prediabetes stage can heighten the risk of multiple comorbidities and chronic complications typically associated with diabetes [3, 6], underscoring the urgency for early intervention strategies.
Many risk factors contribute to diabetes development, including genetics and modifiable lifestyle factors such as diet, smoking, and physical activity [2]. Among them, diet plays a crucial role and some food groups or nutrients have already been linked to the T2DM [1, 2]. Given the complexity of individuals’ eating habits, dietary patterns that describe the overall diet quality may better reflect the diet’s impact on multifactorial diseases than isolated components.
In the past, many dietary indices were designed to evaluate different characteristics or patterns of habitual food consumption. More recently, dietary scores based on the Nutri-Score and the NOVA classification system [7, 8] have become available. The Nutri-Score is a front-of-pack food labeling system computed from the Modified Food Standards Agency Nutrient Profiling System (FSAm-NPS), grading the overall nutritional quality of the food [7]. On the other hand, the NOVA classification rates foods based on their degree of processing, rather than solely on nutrient content [8]. Ultra-processed foods (UPF) belong to the fourth group of the NOVA classification and have undergone the highest degree of industrial processing, potentially impacting health via multiple mechanisms [8,9,10]. Both dietary indices have been widely applied to assess diet quality and ultimately may help to reduce the diet-related burden of non-communicable diseases in society [11,12,13]. Despite several investigations demonstrating a consistent association between UPF consumption and T2DM [14, 15], as well as the high Nutri-Score diet with elevated blood glucose [16, 17], the influence of individuals’ metabolic characteristics on these linkages remains largely unexplored.
Studies suggest that personalized and metabolism-specific dietary recommendations may surpass general advice in terms of their efficacy in improving eating behavior and influencing health outcomes [18,19,20,21]. To address this, one possible solution is to stratify the population into subgroups, termed metabotypes [18], according to similarities in their metabolic profiles and develop tailored preventive measures. Building on our previous work identifying various diet-T2DM associations across metabotypes [22, 23], we aimed to investigate how metabotype subgrouping may affect the dietary score and T2DM association and the implications for disease prevention. We hypothesize that strata-specific dietary interventions could be beneficial. Additionally, we sought to explore the association between dietary patterns and prediabetes. The analysis focused on the FSAm-NPS dietary index (underpinning the Nutri-Score) and UPF intake ratio (using NOVA classification).
Methods
This study’s findings were reported by following the “Strengthening the Reporting of Observational Studies in Epidemiology-Nutritional Epidemiology (STROBE-nut)” checklist [24].
Population
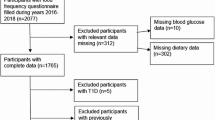
Our analysis was based on data from the German Cooperative Health Research in the Augsburg region (KORA) FF4 study (2013–2014, 2279 participants), a follow-up to the F4 (2006–2008, 3080 participants) and S4 baseline study (1999–2001, 4261 participants), conducted in a randomly selected general population in Augsburg and two surrounding counties. Study designs have been described in detail elsewhere [25]. Out of 2279 FF4 participants, subjects with missing dietary information (n = 677), type 1 diabetes (n = 3), unclear glucose tolerance status due to missing oral glucose tolerance test (OGTT) information (n = 39), or missing metabotype information (n = 24) were excluded. Participants with a diagnosis of myocardial infarction (n = 43) or stroke (n = 31) were further excluded as severe disease might change dietary behavior. A flowchart of exclusion criteria is shown in Supplementary Fig. 1. A total of 1460 participants were eventually selected for our cross-sectional analysis.
The KORA study was authorized by the Ethics Committee of the Bavarian Medical Society and conducted in accordance with the Helsinki Declaration. All study subjects provided written informed consent.
Dietary assessment and dietary indices
The participants’ usual dietary intake was assessed by means of one food frequency questionnaire (FFQ) and repeated 24-hour food lists (24HFL). During the study center visit, participants were required to complete a first 24HFL that assesses foods that were consumed over the previous day; up to two further 24HFLs were collected during the next three months. The 24HFL comprised 246 food items and was derived from the German National Cohort (NAKO) Health study [26]. The FFQ, adapted from the German version of the multilingual European Food Propensity Questionnaire [27], queries the consumption frequency of 148 food items over the past 12 months. Each participant’s usual daily food intake was determined by combining estimated consumption probability and amount, and the detailed has been described elsewhere [28]. The validity of this approach is supported by evidence showing that the combined use of multiple 24-hour recalls and FFQ data provide more accurate intake estimates as compared to either method applied alone [29]. In addition, the usual intake estimates of food items were categorized into main food groups and subgroups based on the EPIC-Soft classification system [30] and estimates of nutrient intake were derived for every participant by linking the usual intake estimates to the German Nutrient Database (Bundeslebensmittelschlüssel 3.02).
In order to indicate participants’ overall dietary quality, we calculated the FSAm-NPS dietary index [7] and the proportion of UPF intake [8] based on their daily consumption estimates.
The FSAm-NPS dietary index assesses the overall nutritional quality of an individual’s diet based on the nutrient profiling system (NPS). It is slightly adjusted to the allocation of points for specific foods (beverages, cheese, and added fats) recommended by the French High Council of Public Health as a modified version of the original Nutrient Analysis System (FSA-NPS) [7, 31]. This ensures that the FSAm-NPS score corresponds to nutritional recommendations and that the nutritional quality of products within these groups can be distinguished [31]. The FSAm-NPS score’s calculation method is described in more detail elsewhere [7, 31, 32]. Theoretically the FSAm-NPS score falls on a scale from − 15 (the healthiest option) to 40. In practice, the Nutri-Score labeling system classifies foods and beverages into five categories, from category A (indicating higher nutritional quality) to category E (indicating lower nutritional quality), based on the FSAm-NPS score [7]. We calculated the FSAm-NPS dietary index for each participant’s diet by adding the FSAm-NPS score for each food or beverage consumed, multiplying it by the amount of energy provided by this product (energy content per 100 g multiplied by the estimated daily intake assessed), and dividing it by the total amount of energy intake [33]. Increasing values of the FSAm-NPS dietary index thus indicate poorer overall diet quality.
UPF intake was estimated using the NOVA classification [8], which categorizes foods based on the level and intensity of industrial processing: (1) fresh/minimally processed foods (e.g., fruit, meat, milk); (2) processed culinary ingredients (e.g., oils, butter, sugar); (3) processed foods (e.g., canned fish, fresh unpackaged bread); and (4) ultra-processed foods (UPFs), made mostly or entirely from derived food constituents with added flavors, colors, and other additives (e.g., sugar-sweetened drinks, processed meat, and savory packaged snacks). To determine the proportion of UPF consumption of the total dietary intake for each participant, we summed the quantities (in kcal/d and gram/d) of each food group from all four categories of NOVA for the total diet and calculated the proportion (%) of UPF (in kcal/d and gram/d, respectively) of the total diet, which is called energy ratio (or weight ratio) in our study. Individuals with a higher UPF intake ratio tend to have a poorer diet quality.
Metabotypes
Aiming at metabolic homogeneity within groups, our study population was divided into three clusters (metabotypes). Further details are described elsewhere [34]. The metabotyping process used 5 parameters, i.e., fasting glucose, high density lipoprotein (HDL) cholesterol, non-HDL cholesterol, uric acid, and body mass index (BMI), to identify clusters by applying the k-means clustering algorithm. Here, we used the Euclidean distances of the three cluster centers as determined in KORA F4 to allocate the KORA FF4 participants to three metabolic clusters [34], using the parameter values measured in fasting blood samples collected during the FF4 study center visit. Among the three clusters, cluster 1 is deemed the most metabolically favorable (“healthy metabotype”), cluster 3 the least favorable, and cluster 2 an “intermediate metabotype” between the other two [34].
Measurement of covariates
Information such as age, sex, education, hypertensive status, alcohol consumption, fasting status, smoking status, and physical activity was assessed by trained medical staff through a standardized interview. The classification of the education variable was determined according to the educational system in Germany. Waist circumference was measured midway between the lowest margin of the least palpable rib and the top of the iliac crest using stretch-resistant tapes. Sitting blood pressure was measured by a trained health worker using an electronic sphygmomanometer. Measurements were conducted after a rest of at least 5 min on the right arm three times. For the analysis, the second and third measurements were averaged.
Blood samples were collected after an overnight fast of ≥ 8 h and at 2 h after ingestion of a 75 g glucose solution in the absence of stasis and stored at 4 °C until centrifugation. Serum glucose was measured with hexokinase/glucose-6-phosphat-dehydrogenase (GLUFlex; Dade Behring, USA). Triglycerides was enzymatically measured in serum (glycerine phosphate oxidase peroxidase method) (TGL Flex, Dade Behring). HDL cholesterol, low density lipoprotein (LDL) cholesterol and total cholesterol were measured in serum by enzymatic methods (CHOL Flex and AHDL Flex, Dade Behring). High-sensitivity C-reactive protein (hs C-reactive protein) was determined by nephelometry on a BN II analyzer (Siemens, Erlangen, Germany) from frozen plasma. Serum concentrations of uric acid were measured from fresh samples by the uricase method (enzymatic color test, URCA Flex®; Dade Behring). More technical details on the processing of blood samples and biomarker measurements could be found elsewhere [35, 36].
BMI was calculated as weight (kg) per height (m2) and categorized into underweight: BMI < 18.5 kg/m2; normal weight: 18.5 ≤ BMI < 25 kg/m2; overweight: 25 ≤ BMI < 30 kg/m2; obese: BMI ≥ 30 kg/m2 [37]. Hypertension was defined as current hypertension (≥ 140/90 mmHg) and/or known hypertension controlled by medication. Participants were categorized as “physically active” if they spent ≥ 1 h per week on leisure physical activity in at least one season (summer or winter); otherwise, they were “physically inactive” [38].
Glucose tolerance status
Glucose tolerance status was categorized into normal glucose tolerance, prediabetes, and T2DM, following the American Diabetes Association criteria [39]. Normal glucose tolerance was defined as fasting glucose < 5.6 mmol/L or 2-h OGTT < 7.8 mmol/L. Prediabetes was defined based on the presence of impaired glucose tolerance (2-h OGTT concentration of 7.8–11.0 mmol/L), impaired fasting glucose (fasting glucose concentration of 5.6–6.9 mmol/L), or both. Undetected diabetes was defined as fasting glucose ≥ 7.0 mmol/L or 2-h OGTT ≥ 11.1 mmol/L in individuals without prevalent, i.e. already known, diabetes. While prevalent diabetes referred to a known diagnosis of T2DM or use of anti-diabetic medication, verified by consulting their treating physician. The OGTT test was conducted in all participants without a previous physician-confirmed diagnosis of T2DM after an overnight fasting period of at least 8 h. We classified individuals as having T2DM if they fell into either the undetected or the prevalent diabetes category.
Statistical analysis
We analyzed a sample of 1460 individuals from the KORA FF4 study. Baseline characteristics were reported stratified by the tertiles of UPF intake (energy ratio). Daily intakes of major food groups and macronutrients were presented separately stratified by tertiles of dietary scores. Normally distributed continuous covariates were reported as means (with standard deviation (SD)), non-normally distributed continuous covariates as medians with the interquartile ranges. Categorical variables were presented as absolute frequencies and proportions in the form of percentages. We compared differences between groups by using the Chi-square test or Fisher’s exact test for categorical variables and analysis of variance (ANOVA) test or Kruskal-Wallis test for continuous variables.
We used multivariable logistic regression models to study the association between dietary scores and T2DM. The ratio of UPF intake was calculated per 5% increase in UPF intake, and the FSAm-NPS dietary index was scaled per 2-point increase in FSAm-NPS points. These models were constructed based on potential confounders according to previous literature. Model 1 was adjusted for age, sex, and total energy intake, while Model 2 included additional adjustments for education, physical activity, and smoking. To avoid overfitting, total energy intake was excluded in models concerning UPF intake energy ratio. Furthermore, we adjusted for metabotype to investigate its effect on this relationship in both models. To evaluate the effect modification of metabotype, we conducted likelihood-ratio tests. Due to significant interactions between metabotype and dietary scores, metabotype-stratified analyses were also employed.
As the assumptions of ordinal regression were violated, multinomial probit models were used to assess the association between dietary scores and glucose tolerance status. The choice of a multinomial probit model over a multinomial regression model was motivated by the results of the Hausman-McFadden test, which indicated a violation of the assumption of irrelevant alternatives (IIA) [40]. Glucose tolerance status was categorized into normal, prediabetes, and T2DM. The adjusted variables were consistent with the logistic regression models mentioned above.
In sensitivity analyses, we further adjusted for waist circumference and hypertension status (Model 3) and replaced waist circumference with BMI in Model 3 alternatives considering their potential mediation role [41,42,43]. To assess the impact of carbohydrates intake on results, we conducted additional analyses (Model 4), adjusting for this factor. We calculated odds ratio (OR) with 95% confidence interval (CI) from the logistic regressions and multinomial probit models. All P values correspond with two-tailed tests. P-values of < 0.05 were considered significant. All statistical analyses were performed using R V.4.1.1.
Results
Among 1460 adults (52.7% of them being women) aged 38–87 years eligible for the study, 173 (11.8%) had T2DM. The mean UPF energy ratio in participants’ diets was 38% (SD = 7%), accompanied by a weight ratio of 16% (SD = 7%) and an FSAm-NPS dietary index averaging 6.93 points (SD = 1.35). Demographic data and comorbidity prevalence by tertiles of UPF intake energy ratio were summarized in Table 1. Compared with the other two groups, participants in the highest UPF intake group were on average younger and male. They had the highest BMI, the largest waist circumference and thus were more often classified as obese. They also showed higher mean triglyceride and C-reactive protein, and lower HDL cholesterol levels than other groups. Furthermore, the highest prevalence of T2DM was observed in this group. We found that participants belonging to metabotype cluster 3 were, on average, the oldest, most likely to be male, and generally had the most unfavorable lifestyle (Supplementary Table S2).
As expected, a higher consumption of UPF (energy ratio) was characterized by higher intakes of meat, non-alcoholic beverages, carbohydrates, fats, protein, and lower intakes of vegetables and fruits (Table 2), whereas no association was evident for UPF with cereals and fats. Stratifying dietary intake by FSAm-NPS dietary index revealed a similar distribution, where the highest FSAm-NPS dietary index tertile group had the highest consumption of meat, carbohydrates, and fats, and the lowest consumption of vegetables.
The associations between UPF intake and T2DM were shown in Fig. 1. In the total study sample, a per 5% increase in UPF intake ratio was associated with elevated odds of T2DM for both energy [OR (95% CI): 1.35 (1.18, 1.55)] and weight ratios [OR (95% CI): 1.27 (1.10, 1.46)], following adjustment with model 1 (without the metabotype variable). On further adjustment (model 2), the significance persisted for the energy ratio [OR (95% CI): 1.22 (1.06, 1.42)], and weight ratio [OR (95% CI): 1.23 (1.05, 1.42)]. No association was detected between UPF intake and T2DM after including the metabotype for all models. However, significant interaction terms (p-interaction < 0.001) were found for metabotype and UPF intake in all models. In stratified analyses, significant associations were only seen in the third cluster. After adjustment using model 1, UPF intake [OR (95% CI) for energy ratio: 1.31 (1.05, 1.65); OR (95% CI) for weight ratio: 1.42 (1.10, 1.87)] was positively associated with T2DM. The association remained significant but decreased slightly for UPF intake weight ratio [OR (95% CI): 1.38 (1.07, 1.81)] after adjustment in model 2. Further adjustment in model 2 significantly attenuated the associations for energy ratio [OR (95% CI): 1.25 (0.99, 1.59)]. Regarding the FSAm-NPS dietary index, a significant positive relationship was observed between each 2-point increment of the index with prevalent T2DM in model 1 [OR (95% CI): 1.82 (1.34, 2.48)] and model 2 [OR (95% CI): 1.54 (1.11, 2.15)] only for the total sample (Fig. 1).
Associations between ultra-processed foods (UPF) intake and Food Standards Agency nutrient profiling system (FSAm-NPS) dietary index with type 2 diabetes (T2DM) in the total sample, and stratified by metabotype. Model 1 adjusted for age, sex, total energy intake. Model 2 additionally adjusted for education, physical activity, smoking. Model 3 additionally adjusted waist circumference and hypertension. For UPF intake (energy ratio), the variable total energy intake was not in models. The T2DM was defined as individuals with undetected or prevalent type 2 diabetes mellitus. The interaction between metabotype and dietary scores were found to be significant in all models (p-interaction < 0.001). UPF, ultra-processed foods; T2DM, type-2 diabetes mellitus; OR, odds ratio; CI, confidence interval. Shown are per 5% increase of UPF intake ratio and per 2-point increase of FSAm-NPS dietary index
In the multinomial probit regression analysis (Table 3), significant positive associations were only found between FSAm-NPS dietary index and prediabetes in model 1 [OR (95% CI): 1.26 (1.11, 1.42)] and model 2 [OR (95% CI): 1.19 (1.04, 1.35)].
The sensitivity analysis, wherein we further adjusted for hypertension and waist circumference (Model 3), showed no significant associations between dietary scores and T2DM, nor for prediabetes in total study sample (Table 3; Fig. 1). Additionally, we ran Model 3 with BMI substituted for waist circumference, the effect sizes and P-values remained similar (data not shown). The further inclusion of carbohydrates intake level (model 4) into the multivariable models strengthened the associations of the UPF intake with T2DM, whereas this adjustment did not significantly alter the association of the FSAm-NPS index with T2DM (Supplementary Tables S3–4).
Discussion
Our population-based study found a significant positive association between diets with poor nutritional profiles or higher UPF intake and T2DM, as indicated by the FSAm-NPS dietary index and the NOVA classification, respectively. The interaction effects between both dietary scores and metabotype were found to be significant. Specifically, in metabotype-stratified subgroups, only metabotype 3, the most metabolically unfavorable group, showed a significant association between UPF consumption and T2DM. No significant relationship was found between the FSAm-NPS dietary index and T2DM in any metabotype subgroup. Concerning glucose tolerance status, a higher FSAm-NPS dietary index, i.e., a lower diet quality, was associated with prediabetes.
Earlier research suggested that diets high in FSAm-NPS dietary index or UPF, were associated with an elevated risk of chronic diseases, specifically diabetes, hypertension, cardiovascular disease, and cancer [44,45,46,47,48]. In accordance with previous studies [16, 49], our study suggests that both diet patterns were similar in that they consisted of substantially larger amounts of meat and sugar, along with reduced vegetables and fruits. Aside from the low nutritional density, UPF are often subjected to degradation of the food matrix, chemical modification of food substances, presence of food additives, loss of micronutrients, and exposure to packaging materials [8, 10]. Due to their pervasive accessibility and affordability, these high-energy-density options may not just account for the majority of consumers’ daily caloric intake [49, 50], but potentially reduce their consumption of health promoting foods such as whole grains, fruits, and vegetables [51]. This shift in dietary habits could, in turn, heighten the risk of adverse health outcomes [9, 50, 52].
In our sensitivity analysis, adjusting for waist circumference and hypertension weakened the association between both dietary scores and T2DM in total study sample. Since the association between diet quality and T2DM is likely mediated, at least partly, by these factors it is possible that model 3 was over-adjusted. Unfortunately, due to the cross-sectional design of our study, conducting a mediation analysis to examine the potential mediating effect is ill-advised. With regards to waist circumference, the analysis demonstrated unaltered results upon substituting it with BMI in Model 3. Given that waist circumference is even more strongly associated with the risk of developing cardiovascular disease and is more frequently used as an indicator of central adiposity than BMI [37], we chose waist circumference over BMI in analysis model 3.
Association between dietary patterns and T2DM
Prior to including the metabotype variable, our study demonstrated a significant positive association between UPF intake (both weight ratio and energy ratio) and T2DM, aligning with the existing literature. The French Nutri Net-Santé cohort study demonstrated that a 10.0% increase in the consumption of UPF (in weight) escalated the risk of T2DM by 13.0% [53]. The higher intake of UPF has been shown to be positively associated with a higher risk for T2DM (hazard ratio 1.12 per 10% increment in UPF weight) in another prospective cohort study based on participants from the UK Biobank [54]. With a median 12-year follow-up, the SUN (Seguimiento Universidad de Navarra) project showed a 53% increased T2DM risk for participants in the highest tertile of UPF intake (in energy) compared to the lowest tertile [55]. Similar trends were also found in the other cohort studies in Netherlands and China [15, 56]. In an effort to bolster comparability and generalizability, we analyzed both the weight and energy ratios of UPF intake, diverging from most previous research that typically focused just on one of these aspects.
In our study, a diet characterized with a higher FSAm-NPS diet index, i.e., a nutritionally poorer diet, was significantly positively associated with diabetes. A significant association was also seen with prediabetes. Few studies have revealed the association between higher FSAm-NPS diet index (or Nutri-Score) and elevated glucose levels [16, 17], an observation that is consistent with our findings. To the best of our knowledge, the current study is the first to examine and identify the associations between FSAm-NPS diet index with both prediabetes and diabetes within a large sample. Notably, all non-diabetic subjects received an OGTT, ensuring an accurate characterization of glucose metabolism.
Our findings support and enhance previous research indicating that both dietary scores similarly impact health [57]. Of note Ferreiro et al. [58] observed that a greater proportion of UPF exists in the higher Nutri-Score categories, with the percentage escalating from 26.1% in category A to a staggering 83.7% in category E. As a practical and simple food labeling system, the Nutri-Score has already proven effectiveness in supporting informed decision-making about healthier food choices, and has been suggested by the EU to enhance customers’ diet quality [12, 13].
Incorporating the metabotype concept
In our analysis, the relationship between UPF consumption and T2DM risk was confined to metabotype 3, the most metabolically unfavorable subgroup. Extensive studies have demonstrated the role of UPF in promoting the development of T2DM [14, 15, 53,54,55,56]; however, they largely overlooked individual factors such as genetics and metabolism, which led to a one-size-fits-all approach in dietary guidelines. Our study differentiates itself by scrutinizing the varied responses—or lack thereof—to dietary patterns across multiple metabotypes, potentially enabling more precise dietary suggestions.
Some researchers argue that the efficacy of dietary interventions can be contingent on individual-specific factors; e.g., the impact of vitamin D [59], breads [60], and red wine polyphenols [61] has been found to significantly differ among various metabotypes. This notion is further substantiated by studies utilizing the decision tree method to deliver personalized dietary recommendations for specific metabolic subgroups [21, 62]. Our research group, in its prior investigations, has also explored the relationship between diet and T2DM, identifying distinct associations only in specific metabotype clusters [22, 23]. Similar to our current results, we have also previously reported stronger associations between common dietary patterns-quantified by the Alternate Healthy Eating Index and the Mediterranean Diet Score—and T2DM in metabotype 3 compared with the total study group [22]. In addition, applying the same metabotype subgrouping approach as in this study, different reactions to the OGTT or dietary fiber supplementation across metabotypes were observed in another study population [63]. Our finding suggests a potentially more pronounced impact of UPF consumption on individuals with unhealthy metabolic profile, supporting the argument for metabotype-specific dietary recommendations over generalized dietary advice [18,19,20].
Subjects in metabotype cluster 3 exhibited relatively adverse demographic characteristics, which are recognized risk factors for diabetes [1, 3], and a higher prevalence of prediabetes, T2DM, and hypertension, demonstrating the metabotype concept’s effectiveness in stratifying populations into distinct metabolic subgroups [34]. To further confirm our hypothesis, as well as deepen our understanding of the intricate interplay between dietary patterns, metabotype, and diabetes risk, it is necessary to conduct future prospective studies with larger sample size. Such an approach could help formulate tailored dietary recommendations for different metabotype subgroups and implement personalized disease prevention strategies on a population-wide scale.
Strengths and limitations
Our relatively large sample size, comprehensive dietary data collected via combined repeated 24HFLs and an FFQ, and detailed glucose tolerance status information strengthened our study. Regarding the cross-sectional study design, our findings can provide insights into potential associations and generate hypotheses on dietary recommendation, rather than establishing causality. Also, subgrouping resulted in a smaller participants number in each metabotype. The recall bias and the potential for under- or over-reporting inherent in dietary data collection cannot be entirely circumvented. Population loss during follow-up from KORA S4 to FF4 and the exclusion criteria may have introduced selection bias. Also, the definition of “metabotype” is yet to be standardized. Nevertheless, we employed an optimized metabotype as proposed by Dahal et al. [34], which reduced the previous comprehensive set of 32 [35] and 16 [23] parameters down to just 5 that are measured routinely. This approach simplifies the metabotype concept while preserving its validity, potentially facilitating its wider dissemination and application.
Conclusion
Taken together, the study suggests that poorer diet quality or higher UPF intake, evaluated by FSAm-NPS dietary index and NOVA classification, respectively, were significantly associated with T2DM. Poorer overall dietary quality is also related to prediabetes. Notably, across different metabotype subgroups, the association between UPF consumption and T2DM was only found in subjects with the metabolically most unfavorable metabotype 3. This study emphasizes the potential benefits of personalized or stratified dietary interventions as a means to address the challenge of reducing diabetes incidence. Future studies evaluating long-term diabetes outcomes are needed before a metabotype-specific dietary recommendation can be fully endorsed.
Data availability
The data underlying this article cannot be shared publicly due to data protection reasons. The data will be shared on reasonable request to the corresponding author.
Abbreviations
- BMI:
-
Body Mass Index
- CI:
-
Confidence Interval
- FFQ:
-
Food Frequency Questionnaire
- FSAm-NPS:
-
Modified Food Standards Agency Nutrient Profiling System
- HDL cholesterol:
-
High Density Lipoprotein cholesterol
- hs C-reactive protein:
-
High-Sensitivity C-reactive protein
- KORA:
-
Cooperative Health Research in the Region of Augsburg
- LDL cholesterol:
-
Low-density lipoprotein cholesterol
- NAKO:
-
German National Cohort
- OGTT:
-
Oral Glucose Tolerance Test
- OR:
-
Odds Ratio
- T2DM:
-
Type 2 Diabetes Mellitus
- UPF:
-
Ultra-Processed Foods
- 24HFL:
-
24-Hour Food Lists
References
Roglic G (2016) WHO Global report on diabetes: a summary. Int J Noncommunicable Dis 1(1):3
Bellou V, Belbasis L, Tzoulaki I, Evangelou E (2018) Risk factors for type 2 diabetes mellitus: an exposure-wide umbrella review of meta-analyses. PLoS ONE 13(3):e0194127. https://doi.org/10.1371/journal.pone.0194127
Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A et al (2022) IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract 183:109119. https://doi.org/10.1016/j.diabres.2021.109119
2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2021 (2021) Diabetes Care 44(Suppl 1):S15–s33. https://doi.org/10.2337/dc19-S002
Buysschaert M, Bergman M (2011) Definition of prediabetes. Med Clin N Am 95(2):289–297, vii. https://doi.org/10.1016/j.mcna.2010.11.002
Tabák AG, Herder C, Rathmann W, Brunner EJ, Kivimäki M (2012) Prediabetes: a high-risk state for diabetes development. Lancet (London England) 379(9833):2279–2290. https://doi.org/10.1016/s0140-6736(12)60283-9
Chantal J, Hercberg S (2017) WHO Development of a new front-of-pack nutrition label in France: the five-colour nutri-score. Public Health Panorama 3(04):712–725
Monteiro CA, Cannon G, Levy R, Moubarac J-C, Jaime P, Martins AP, Canella D, Louzada M, Parra D (2016) NOVA. The star shines bright. World Nutr 7(1–3):28–38
Dicken SJ, Batterham RL (2022) The role of Diet Quality in Mediating the Association between Ultra-processed Food Intake, obesity and health-related outcomes: a review of prospective cohort studies. Nutrients 14(1):23. https://doi.org/10.3390/nu14010023
Monteiro CA (2009) Nutrition and health. The issue is not food, nor nutrients, so much as processing. Public Health Nutr 12(5):729–731. https://doi.org/10.1017/s1368980009005291
Martinez-Steele E, Khandpur N, Batis C, Bes-Rastrollo M, Bonaccio M, Cediel G, Huybrechts I, Juul F, Levy RB, da Costa Louzada ML (2023) Best practices for applying the Nova food classification system. Nat Food 1–4. https://doi.org/10.1038/s43016-023-00779-w
Delhomme V (2021) Front-of-pack nutrition labelling in the European Union: a behavioural, legal and political analysis. Eur J Risk Regul 12(4):825–848. https://doi.org/10.1017/err.2021.5
Hercberg S, Touvier M, Salas-Salvado J (2021) Group of European scientists supporting the implementation of Nutri-Score in Europe. The Nutri-Score Nutrition Label: a Public Health Tool Based. on Rigorous Scientific Evidence Aiming to Improve the Nutritional Status of the Population
Delpino FM, Figueiredo LM, Bielemann RM, da Silva BGC, dos Santos FS, Mintem GC, Flores TR, Arcêncio RA, Nunes BP (2021) Ultra-processed food and risk of type 2 diabetes: a systematic review and meta-analysis of longitudinal studies. Int J Epidemiol 51(4):1120–1141. https://doi.org/10.1093/ije/dyab247
Duan M-J, Vinke PC, Navis G, Corpeleijn E, Dekker LH (2022) Ultra-processed food and incident type 2 diabetes: studying the underlying consumption patterns to unravel the health effects of this heterogeneous food category in the prospective Lifelines cohort. BMC Med 20(1):7. https://doi.org/10.1186/s12916-021-02200-4
Paper L, Ahmed M, Lee JJ, Kesse-Guyot E, Touvier M, Hercberg S, Galan P, Salanave B, Verdot C, L’Abbé MR, Deschamps V, Julia C (2023) Cross-sectional comparisons of dietary indexes underlying nutrition labels: nutri-score, Canadian ‘high in’ labels and Diabetes Canada Clinical Practices (DCCP). Eur J Nutr 62(1):261–274. https://doi.org/10.1007/s00394-022-02978-w
Khoury N, Gómez-Donoso C, Martínez M, Martínez-González M, Corella D, Fitó M, Martínez JA, Alonso-Gómez ÁM, Wärnberg J, Vioque J, Romaguera D, León-Acuña A et al (2022) Associations between the Modified Food Standard Agency Nutrient Profiling System Dietary Index and Cardiovascular Risk factors in an Elderly Population. Front Nutr 9:897089. https://doi.org/10.3389/fnut.2022.897089
Holmes E, Wilson ID, Nicholson JK (2008) Metabolic phenotyping in health and disease. Cell 134(5):714–717. https://doi.org/10.1016/j.cell.2008.08.026
Celis-Morales C, Livingstone KM, Marsaux CF, Macready AL, Fallaize R, O’Donovan CB, Woolhead C, Forster H, Walsh MC, Navas-Carretero S, San-Cristobal R, Tsirigoti L et al (2016) Effect of personalized nutrition on health-related behaviour change: evidence from the Food4Me European randomized controlled trial. Int J Epidemiol 46(2):578–588. https://doi.org/10.1093/ije/dyw186
Livingstone K, Celis-Morales C, Navas-Carretero S, San-Cristobal R, Forster H, Woolhead C, O’Donovan C, Moschonis G, Manios Y, Traczyk I, Gundersen T, Drevon C et al (2021) Personalized Nutrition Advice Reduces Intake of Discretionary Foods and beverages: findings from the Food4Me Randomized Controlled Trial. Curr Developments Nutr 5:152. https://doi.org/10.1093/cdn/nzab035_060
Zeevi D, Korem T, Zmora N, Israeli D, Rothschild D, Weinberger A, Ben-Yacov O, Lador D, Avnit-Sagi T, Lotan-Pompan M (2015) Personalized nutrition by prediction of glycemic responses. Cell 163(5):1079–1094. https://doi.org/10.1016/j.cell.2015.11.001
Wawro N, Pestoni G, Riedl A, Breuninger TA, Peters A, Rathmann W, Koenig W, Huth C, Meisinger C, Rohrmann S (2020) Association of dietary patterns and type-2 diabetes mellitus in metabolically homogeneous subgroups in the KORA FF4 study. Nutrients 12(6):1684. https://doi.org/10.3390/nu12061684
Riedl A, Wawro N, Gieger C, Meisinger C, Peters A, Rathmann W, Koenig W, Strauch K, Quante AS, Thorand B (2020) Modifying effect of metabotype on diet–diabetes associations. Eur J Nutr 59:1357–1369. https://doi.org/10.1007/s00394-019-01988-5
Lachat C, Hawwash D, Ocké MC, Berg C, Forsum E, Hörnell A, Larsson CL, Sonestedt E, Wirfält E, Åkesson A, Kolsteren P, Byrnes G et al (2016) Strengthening the reporting of Observational studies in Epidemiology - nutritional epidemiology (STROBE-nut): an extension of the STROBE statement. Nutr Bull 41(3):240–251. https://doi.org/10.1111/nbu.12217
Laxy M, Knoll G, Schunk M, Meisinger C, Huth C, Holle R (2016) Quality of Diabetes Care in Germany Improved from 2000 to 2007 to 2014, but improvements diminished since 2007. Evidence from the Population-based KORA studies. PLoS ONE 11(10):e0164704. https://doi.org/10.1371/journal.pone.0164704
Freese J, Feller S, Harttig U, Kleiser C, Linseisen J, Fischer B, Leitzmann M, Six-Merker J, Michels K, Nimptsch K (2014) Development and evaluation of a short 24-h food list as part of a blended dietary assessment strategy in large-scale cohort studies. Eur J Clin Nutr 68(3):324–329. https://doi.org/10.1038/ejcn.2013.274
Illner A-K, Harttig U, Tognon G, Palli D, Salvini S, Bower E, Amiano P, Kassik T, Metspalu A, Engeset D (2011) Feasibility of innovative dietary assessment in epidemiological studies using the approach of combining different assessment instruments. Public Health Nutr 14(6):1055–1063. https://doi.org/10.1017/S1368980010003587
Mitry P, Wawro N, Six-Merker J, Zoller D, Jourdan C, Meisinger C, Thierry S, Nothlings U, Knuppel S, Boeing H, Linseisen J (2019) Usual dietary intake estimation based on a combination of repeated 24-H food lists and a food frequency questionnaire in the KORA FF4 cross-sectional study. Front Nutr 6:145. https://doi.org/10.3389/fnut.2019.00145
Carroll RJ, Midthune D, Subar AF, Shumakovich M, Freedman LS, Thompson FE, Kipnis V (2012) Taking advantage of the strengths of 2 different dietary assessment instruments to improve intake estimates for nutritional epidemiology. Am J Epidemiol 175(4):340–347. https://doi.org/10.1093/aje/kwr317
Slimani N, Deharveng G, Charrondiere RU, van Kappel AL, Ocke MC, Welch A, Lagiou A, van Liere M, Agudo A, Pala V, Brandstetter B, Andren C et al (1999) Structure of the standardized computerized 24-h diet recall interview used as reference method in the 22 centers participating in the EPIC project. European prospective investigation into Cancer and Nutrition. Comput Methods Programs Biomed 58(3):251–266. https://doi.org/10.1016/s0169-2607(98)00088-1
Publique HCS (2015) Opinion on information regarding the nutritional quality of foodstuffs. HCSP, Paris
Julia C, Kesse-Guyot E, Touvier M, Mejean C, Fezeu L, Hercberg S (2014) Application of the British Food Standards Agency nutrient profiling system in a French food composition database. Br J Nutr 112(10):1699–1705. https://doi.org/10.1017/S0007114514002761
Julia C, Méjean C, Touvier M, Péneau S, Lassale C, Ducrot P, Hercberg S, Kesse-Guyot E (2016) Validation of the FSA nutrient profiling system dietary index in French adults-findings from SUVIMAX study. Eur J Nutr 55(5):1901–1910. https://doi.org/10.1007/s00394-015-1006-y
Dahal C, Wawro N, Meisinger C, Breuninger TA, Thorand B, Rathmann W, Koenig W, Hauner H, Peters A, Linseisen J (2022) Optimized metabotype definition based on a Limited Number of Standard Clinical Parameters in the Population-based KORA Study. Life (Basel) 12(10). https://doi.org/10.3390/life12101460
Riedl A, Wawro N, Gieger C, Meisinger C, Peters A, Roden M, Kronenberg F, Herder C, Rathmann W, Völzke H (2018) Identification of Comprehensive Metabotypes Associated with Cardiometabolic diseases in the Population-based KORA Study. Mol Nutr Food Res 62(16):1800117. https://doi.org/10.1002/mnfr.201800117
Breuninger TA, Wawro N, Meisinger C, Artati A, Adamski J, Peters A, Grallert H, Linseisen J (2019) Associations between fecal bile acids, neutral sterols, and serum lipids in the KORA FF4 study. Atherosclerosis 288:1–8. https://doi.org/10.1016/j.atherosclerosis.2019.06.911
WHO (2000) Obesity: preventing and managing the global epidemic
Rabel M, Meisinger C, Peters A, Holle R, Laxy M (2017) The longitudinal association between change in physical activity, weight, and health-related quality of life: results from the population-based KORA S4/F4/FF4 cohort study. PLoS ONE 12(9):e0185205. https://doi.org/10.1371/journal.pone.0185205
ADA (2013) Diagnosis and classification of diabetes Mellitus. Diabetes Care 37(Supplement1):S81–S90. https://doi.org/10.2337/dc14-S081
Dow JK, Endersby JW (2004) Multinomial probit and multinomial logit: a comparison of choice models for voting research. Electoral Stud 23(1):107–122. https://doi.org/10.1016/S0261-3794(03)00040-4
Ley SH, Pan A, Li Y, Manson JE, Willett WC, Sun Q, Hu FB (2016) Changes in overall Diet Quality and subsequent type 2 diabetes risk: three U.S. prospective cohorts. Diabetes Care 39(11):2011–2018. https://doi.org/10.2337/dc16-0574
Byles JE, Forder PM, Holder C, Jackson JK, MacDonald-Wicks LK, McEvoy MA, Oldmeadow C, Patterson AJ (2020) Better diet quality scores are associated with a lower risk of hypertension and non-fatal CVD in middle-aged Australian women over 15 years of follow-up. Public Health Nutr 23(5):882–893. https://doi.org/10.1017/S1368980019002842
Pate RR, Taverno Ross SE, Liese AD, Dowda M (2015) Associations among physical activity, diet quality, and weight status in US adults. Med Sci Sports Exerc 47(4):743–750. https://doi.org/10.1249/MSS.0000000000000456
Bakre S, Shea B, Langheier J, Hu EA (2022) Blood pressure control in individuals with hypertension who used a Digital, Personalized Nutrition platform: longitudinal study. JMIR Form Res 6(3):e35503. https://doi.org/10.2196/35503
Andreeva VA, Egnell M, Touvier M, Galan P, Julia C, Hercberg S (2021) International evidence for the effectiveness of the front-of-package nutrition label called nutri-score. Cent Eur J Public Health 29(1):76–79. https://doi.org/10.21101/cejph.a6239
Lane MM, Davis JA, Beattie S, Gómez-Donoso C, Loughman A, O’Neil A, Jacka F, Berk M, Page R, Marx W, Rocks T (2021) Ultraprocessed food and chronic noncommunicable diseases: a systematic review and meta-analysis of 43 observational studies. Obes Reviews: Official J Int Association Study Obes 22(3):e13146. https://doi.org/10.1111/obr.13146
Jardim MZ, Costa BVL, Pessoa MC, Duarte CK (2021) Ultra-processed foods increase noncommunicable chronic disease risk. Nutrition research. (New York NY) 95:19–34. https://doi.org/10.1016/j.nutres.2021.08.006
Cordova R, Viallon V, Fontvieille E, Peruchet-Noray L, Jansana A, Wagner KH, Kyrø C, Tjønneland A, Katzke V, Bajracharya R, Schulze MB, Masala G et al (2023) Consumption of ultra-processed foods and risk of multimorbidity of cancer and cardiometabolic diseases: a multinational cohort study. Lancet Reg Health Europe 35:100771. https://doi.org/10.1016/j.lanepe.2023.100771
Martini D, Godos J, Bonaccio M, Vitaglione P, Grosso G (2021) Ultra-processed Foods and Nutritional Dietary Profile: a Meta-analysis of nationally Representative Samples. Nutrients 13(10):3390. https://doi.org/10.3390/nu13103390
Rauber F, Louzada M, Martinez Steele E, Rezende LFM, Millett C, Monteiro CA, Levy RB (2019) Ultra-processed foods and excessive free sugar intake in the UK: a nationally representative cross-sectional study. BMJ open 9(10):e027546. https://doi.org/10.1136/bmjopen-2018-027546
Popkin BM, Adair LS, Ng SW (2012) Global nutrition transition and the pandemic of obesity in developing countries. Nutr Rev 70(1):3–21. https://doi.org/10.1111/j.1753-4887.2011.00456.x
Canhada SL, Luft VC, Giatti L, Duncan BB, Chor D, Fonseca MdJMd, Matos SMA, Molina MCB, Barreto SM, Levy RB, Schmidt MI (2020) Ultra-processed foods, incident overweight and obesity, and longitudinal changes in weight and waist circumference: the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil). Public Health Nutr 23(6):1076–1086. https://doi.org/10.1017/S1368980019002854
Srour B, Fezeu LK, Kesse-Guyot E, Allès B, Debras C, Druesne-Pecollo N, Chazelas E, Deschasaux M, Hercberg S, Galan P, Monteiro CA, Julia C et al (2020) Ultraprocessed Food Consumption and Risk of type 2 diabetes among participants of the NutriNet-Santé prospective cohort. JAMA Intern Med 180(2):283–291. https://doi.org/10.1001/jamainternmed.2019.5942
Levy RB, Rauber F, Chang K, Louzada MLC, Monteiro CA, Millett C, Vamos EP (2021) Ultra-processed food consumption and type 2 diabetes incidence: a prospective cohort study. Clin Nutr 40(5):3608–3614. https://doi.org/10.1016/j.clnu.2020.12.018
Llavero-Valero M, Escalada-San Martín J, Martínez-González MA, Basterra-Gortari FJ, de la Fuente-Arrillaga C, Bes-Rastrollo M (2021) Ultra-processed foods and type-2 diabetes risk in the SUN project: a prospective cohort study. Clin Nutr 40(5):2817–2824. https://doi.org/10.1016/j.clnu.2021.03.039
Li M, Shi Z (2022) Association between ultra-processed food consumption and diabetes in Chinese Adults—Results from the China Health and Nutrition Survey. Nutrients 14(20):4241. https://doi.org/10.3390/nu14204241
Bonaccio M, Di Castelnuovo A, Ruggiero E, Costanzo S, Grosso G, De Curtis A, Cerletti C, Donati MB, de Gaetano G, Iacoviello L, Moli-sani Study I (2022) Joint association of food nutritional profile by nutri-score front-of-pack label and ultra-processed food intake with mortality: Moli-Sani prospective cohort study. BMJ (Clinical Res ed) 378:e070688. https://doi.org/10.1136/bmj-2022-070688
Romero Ferreiro C, Lora Pablos D, de la Gómez A (2021) Two dimensions of Nutritional Value: Nutri-score and NOVA. Nutrients 13(8). https://doi.org/10.3390/nu13082783
O’Sullivan A, Gibney MJ, Connor AO, Mion B, Kaluskar S, Cashman KD, Flynn A, Shanahan F, Brennan L (2011) Biochemical and metabolomic phenotyping in the identification of a vitamin D responsive metabotype for markers of the metabolic syndrome. Mol Nutr Food Res 55(5):679–690. https://doi.org/10.1002/mnfr.201000458
Moazzami AA, Shrestha A, Morrison DA, Poutanen K, Mykkänen H (2014) Metabolomics Reveals Differences in Postprandial Responses to Breads and Fasting Metabolic Characteristics Associated with Postprandial insulin demand in Postmenopausal Women. J Nutr 144(6):807–814. https://doi.org/10.3945/jn.113.188912
Vázquez-Fresno R, Llorach R, Perera A, Mandal R, Feliz M, Tinahones FJ, Wishart DS, Andres-Lacueva C (2016) Clinical phenotype clustering in cardiovascular risk patients for the identification of responsive metabotypes after red wine polyphenol intake. J Nutr Biochem 28:114–120. https://doi.org/10.1016/j.jnutbio.2015.10.002
O’Donovan CB, Walsh MC, Woolhead C, Forster H, Celis-Morales C, Fallaize R, Macready AL, Marsaux CF, Navas-Carretero S, San-Cristobal SR (2017) Metabotyping for the development of tailored dietary advice solutions in a European population: the Food4Me study. Br J Nutr 118(8):561–569. https://doi.org/10.1017/S0007114517002069
Dahal C, Wawro N, Meisinger C, Brandl B, Skurk T, Volkert D, Hauner H, Linseisen J (2022) Evaluation of the metabotype concept after intervention with oral glucose tolerance test and dietary fiber-enriched food: an enable study. Nutrition, metabolism, and cardiovascular diseases. NMCD 32(10):2399–2409. https://doi.org/10.1016/j.numecd.2022.06.007
Acknowledgements
The contribution of the participants of the KORA FF4 study is very much acknowledged.
Funding
The KORA study was initiated and financed by the Helmholtz Zentrum München – German Research Center for Environmental Health, which is funded by the German Federal Ministry of Education and Research (BMBF) and by the State of Bavaria. Dietary data collection in the KORA FF4 study was done in cooperation with the Chair of Epidemiology, University Hospital Augsburg.
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
ZD conducted the statistical analysis, interpreted the data, and drafted and revised the manuscript; JL designed the study, and supervised the work; TB established the dietary scores in KORA FF4 and introduced ZD; NW and DF supervised the statistical analysis; CM contributed to the data interpretation and the revision of the draft manuscript; AP and MH were responsible for the design and conduct of the KORA FF4 study; all authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
The KORA FF4 study was conducted according to the guidelines laid down in the Declaration of Helsinki, including written informed consent of all participants. All study methods involving human subjects were approved by the ethics committee of the Bavarian Chamber of Physicians, Munich (protocol code EC No. 06068, 25 October 2012).
Consent to participate
All participants provided written informed consent.
Conflict of interest
The authors declare that there are no conflicts of interest to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, Z., Wawro, N., Freuer, D. et al. Differential association of dietary scores with the risk of type 2 diabetes by metabotype. Eur J Nutr (2024). https://doi.org/10.1007/s00394-024-03411-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00394-024-03411-0